Abstract
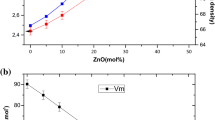
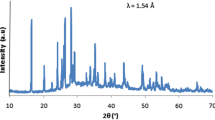
The glass system xZnO-10B2O3-(100 − x)P2O5 was prepared by cooling using the melt-quenching technique in the composition series x from 30 to 60 mol %. The density, thermal expansion coefficient, glass transition temperature, and softening temperature of the glass system were determined. Structural characterization was performed by using a combination of IR and Raman spectroscopy and 11B/31P solid state NMR spectroscopy data. In particular, variations in the phosphate network structure upon the addition of ZnO were investigated. Data analysis indicated that increasing the ZnO content and decreasing the P2O5 content increased the extent of cross-linking between the phosphate and borate units in the glass network; this was because incorporation of ZnO polyhedra into the structural network of ZnO increased the network dimensionality. These structural changes were confirmed by Raman and IR spectroscopy.
Similar content being viewed by others
References
Kim, Y.S., Lee, K.H., Kim, T.H., Jung, Y.J., and Ryu, B.K., Effect of ZnO addition on the photoluminescence of a novel phosphate glass, J. Korean Phys. Soc., 2008, vol. 53, no. 1, pp. 232–235.
Ivanda, M., Babocsi, K., Dem, C., Schmitt, M., Montana, M., and Kiefer, W., Low-wave-number Raman scattering from CdSxSe1 − x quantum dots embedded in a glass matrix, Phys. Rev. B: Condens. Matter, 2003, vol. 67, no. 23, pp. 15–22.
Maly, P. and Miyoshi, T., Effect of photodarkening on photoluminescence dynamics in CdS-doped glass, J. Lumin., 2000, vol. 90, no. 3, pp. 129–134.
Hussin, R., Holland, D., and Dupree, R., A MAS NMR structural study of cadmium phosphate glasses, J. Non-Cryst. Solids, 2002, vol. 298, no. 1, pp. 32–42.
Martin, S.W., Review of the structures of phosphate glasses, Eur. J. Solid State Inorg. Chem., 1991, vol. 28, no. 1, pp. 163–205.
Marino, A.E., Arrasmith, S.R., Gregg, L.L., Jacobs, S.D., Chen, G., and Duc, Y., Durable phosphate glasses with lower transition temperatures, J. Non-Cryst. Solids, 2001, vol. 289, no. 1, pp. 37–41.
Yu, X., Day, D.E., Long, G.J., and Brow, R.K., Properties and structure of sodium-iron phosphate glasses, J. Non-Cryst. Solids, 1991, vol. 215, no. 1, pp. 21–31.
Takebe, H., Baba, Y., and Kuwabara, M., Dissolution behavior of ZnO-P2O5 glasses in water, J. Non-Cryst. Solids, 2006, vol. 352, no. 28, pp. 3088–3094.
Samuneva, B., Tzvetkova, P., Gugov, I., and Dimitrov, V., Structural studies of phosphate glasses, J. Mater. Sci. Lett., 1996, vol. 15, no. 24, pp. 2180–2183.
Gongyi Guo and Yuli Chen, Thermal analysis and infrared measurements of a lead-barium-aluminum phosphate glass, J. Non-Cryst. Solids, 1996, vol. 201, no. 3, pp. 262–266.
Furdanowicz, W. and Klein, L.C., Viscosities and durabilities of lead phosphate glasses, Glass Technol., 1986, vol. 24, no. 4, pp. 198–201.
Westman, A.E.R. and Crowther, J., Constitution of soluble phosphate glasses, J. Am. Ceram. Soc., 1954, vol. 37, no. 9, pp. 420–427.
Van Wazer, J.R., Structure and properties of the condensed phosphates: II. A theory of the molecular structure of sodium phosphate glasses, J. Am. Chem. Soc., 1950, vol. 72, no. 2, pp. 644–647.
Westman, A.E.R. and Gartaganis, P.A., Constitution of sodium, potassium, and lithium phosphate glasses, J. Am. Ceram. Soc., 1957, vol. 40, no. 9, pp. 293–299.
Ducel, J.F., Videau, J.J., and Couzi, M., Structural study of borophosphate glassed by Raman and infrared spectroscopy, Phys. Chem. Glasses, 1993, vol. 34, no. 5, pp. 212–218.
Lee, S.W., Ryoo, K.S., Kim, J.E., Lee, J.H., Kim, C.D., and Hong, K.S., Structure and radiative properties of aluminophosphate glasses, J. Mater. Sci., 1994, vol. 29, no. 17, pp. 4577–4582.
Koo, J.M., Bae, B.S., and Na, H.K., Raman spectroscopy of copper phosphate glasses, J. Non-Cryst. Solids, 1997, vol. 212, no. 2, pp. 173–179.
Hudgens, J.J. and Martin, S.W., Glass transition and infrared spectra of low-alkali, anhydrous lithium phosphate glasses, J. Am. Ceram. Soc., 1993, vol. 76, no. 7, pp. 1691–1696.
Meyer, K., Characterization of the structure of binary zinc ultraphosphate glasses by infrared and Raman spectroscopy, J. Non-Cryst. Solids, 1997, vol. 209, no. 3, pp. 227–239.
Lee, S.W., Hong, K.S., and Condrate, R.A., Raman microprobe investigation of oxygen and carbon dioxide dissolution from bubbles in silicate glasses containing antimony oxide, J. Mater. Sci., 1991, vol. 26, no. 15, pp. 4250–4254.
Kingery, W.D., Bowen, H.K., and Uhlmann, D.R., Introduction to Ceramics, New York: Wiley, 1976.
Scholze, H., Glass: Nature, Structure, and Properties, London: Springer-Verlag, 2011.
Ushakov, D.F., Baskova, N.F., and Tarlakov, Yu.P., Glass formation in the system ZnO-B2O3-P2O5, Fiz. Khim. Stekla, 1975, vol. 1, no. 1, pp. 151–156.
Hoppe, U., A structural model for phosphate glasses, J. Non-Cryst. Solids, 1996, vol. 195, no. 1, pp. 138–147.
Clinton, J.M. and Coffeen, W.W., Low-melting glasses in the system B2O3-ZnO-CaO-P2O5, Am. Ceram. Soc. Bull., 1984, vol. 63, no. 11, pp. 1401–1404.
Brow, R.K. and Tallant, D.R., Structural design of sealing glasses, J. Non-Cryst. Solids, 1997, vol. 222, no. 1, pp. 396–406.
Martin, S.W., Review of the structures of phosphate glasses, Eur. J. Solid State Inorg. Chem., 1991, vol. 28, no. 17, pp. 163–205.
Bansal, N.P. and Doremus, R.H., Handbook of Glass Properties, Orlando, Florida, United States: Academic, 1986.
Rosenthal, A.B. and Garofalini, S.H., Structural role of zinc oxide in silica and soda-silica glasses, J. Am. Ceram. Soc., 1987, vol. 70, no. 11, pp. 821–826.
Nakamoto, K., Infrared and Raman Spectra of Inorganic and Coordination Compounds, New York: Wiley, 1986.
Greaves, G.N., EXAFS and the structure of glass, J. Non-Cryst. Solids, 1985, vol. 71, no. 1, pp. 203–217.
Byun, J.-O., Kim, B.-H., Hong, K.-S., Jung, H.-J., Lee, S.-W., and Izyneev, A.A., Properties and structure of (45 − x)RO · xNa2O · 2.5Al2O3 · 52.5P2O5 (R = Mg, Ca, Sr, Ba) glasses, J. Non-Cryst. Solids, 1995, vol. 190, no. 1, pp. 288–295.
Ducel, J.F. and Videau, J.J., Structural study of borophosphate glasses by Raman and infrared spectroscopy, Phys. Chem. Glasses, 1994, vol. 34, no. 5, pp. 212–218.
Brow, R.K., Review: The structure of simple phosphate glasses, J. Non-Cryst. Solids, 2000, vol. 263, no. 1, pp. 1–28.
Brow, R.K., An XPS study of oxygen bonding in zinc phosphate and zinc borophosphate glasses, J. Non-Cryst. Solids, 1996, vol. 194, no. 3, pp. 267–273.
Ray, N.H., Study of the coordination of B in K borophosphate glasses by Raman spectroscopy, Phys. Chem. Glasses, 1975, vol. 16, no. 4, pp. 75–80.
Dumbaugh, W.H., Infrared transmitting glasses, Opt. Eng., 1985, vol. 24, no. 2, pp. 257–262.
Kim, D.H., Hwang, C.H., Gwoo, D.G., Kim, T.H., Kim, N.J., and Ryu, B.K., Effect of titanium on structure of sodium borophosphate glasses, Electron. Mater. Lett., 2011, vol. 7, no. 4, pp. 343–347.
Pei Chen, Shengchun Li, Wenjie Qiao, and Yaogang Li, Structure and crystallization of ZnO-B2O3-P2O5 glasses, Glass Phys. Chem., 2011, vol. 37, no. 1, pp. 29–33.
Koudelka, L. and Mosner, P., Borophosphate glasses of the ZnO-B2O3-P2O5 system, Mater. Lett., 2000, vol. 42, no. 3, pp. 194–199.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Kim, YS., Choi, WG. & Ryu, BK. Effect of ZnO content change on the structure and properties of zinc borophosphate glasses. Glass Phys Chem 40, 408–414 (2014). https://doi.org/10.1134/S1087659614040142
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1087659614040142