Abstract
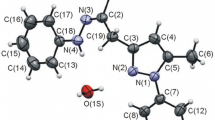
Monophenylhydrazones were prepared from three monochloro derivatives of arylaliphatic 1,5-diketones, and some their transformations were investigated. The monophenylhydrazones formed regiospecifically, and the direction of the reaction was governed by the substituent attached to C3 atom in the initial chlorodiketones. Formerly unknown products of transformations suffered by monophenylhydrazones obtained were described: 2-amino-1,3-diphenyl-3-(2-phenylindolyl-3)-propan-1-one, 3-benzoyl-2,4,6-triphenyl-2,3,4,5-tetrahydropyridazine, and 3-methyl-1,5-diphenyl-5-phenylazopent-4-en-1-one.
Similar content being viewed by others
References
Kharchenko, V.G., Chalaya, S.N., Litvinov, O.V., Yudovich, L.M., and Promonenkov, V.K., Zh. Org. Khim., 1984, vol. 20, p. 1208; Litvinov, O.V., Kolyagin, N.T., Chalaya, S.N., Kharchenko, V.G., Yanovskii, A.I., and Struchkov, M.T., Zh. Org. Khim., 1989, vol. 25, p. 34.
Moskovkina, T.V. and Vysotskii, V.I., Zh. Org. Khim., 1991, vol. 27, p. 833; Moskovkina, T.V. and Vysotskii, V.I., Zh. Org. Khim., 1994, vol. 30, p. 996.
Grandberg, I.I., Izv. Akad. Nauk SSSR, Ser. Khim., 1966, p. 1682;, Grandberg, I.I., Afonina, N.I., and Zuyanova, T.I., Khim. Geterotsikl. Soedin., 1968, p. 1038.
Grandberg, I.I. and Zuyanova, T.I., Khim. Geterotsikl. Soedin., 1970, p. 750.
Rochus, J., Wolf, M., Beier, N., Kluxen, F., and Fittschen, C., German Patent 59484, 2000; Chem. Abstr., 2000, vol. 133, 301117a; Shibata, T., Tamaki, K., Tanazawa, K., and Kurakata, S., Japan Patent 163862, 2001; Chem. Abstr., 2001, vol. 135, 46189c.
Kitaev, Yu.P. and Buzykin, B.I., Gidrazony (Hydrazones), Moscow: Nauka, 1974, p. 370.
Author information
Authors and Affiliations
Additional information
Original Russian Text © T.V. Moskovkina, A.I. Kalinovskii, 2007, published in Zhurnal Organicheskoi Khimii, 2007, Vol. 43, No. 2, pp. 222–227.
Rights and permissions
About this article
Cite this article
Moskovkina, T.V., Kalinovskii, A.I. Transformations of stereoisomeric 2-chloro-3-R-pentane-1,5-diones in reaction with phenylhydrazine. Russ J Org Chem 43, 214–219 (2007). https://doi.org/10.1134/S1070428007020108
Received:
Issue Date:
DOI: https://doi.org/10.1134/S1070428007020108