Abstract
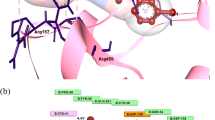
A new series of coupled benzofuran derivatives that contain the trifluoromethyl group and pyrazole moiety is synthesized for evaluation of their anti-inflammatory and analgesic activities. Their synthesis proceeds via cyclocondensation of 2-hydroxy-4-(trifluoromethyl)benzaldehyde with chloroacetone with formation of 2-acetylbenzofuran, which upon treatment with different aldehydes via the Claisen-Schmidt condensation gives the corresponding chalcones. Cyclization of chalcones with hydrazine hydrate leads to pyrazoline derivatives, that exhibit significant anti-inflammatory and analgesic activities. Docking modelling of selected active compounds are correlated well with their anti-inflammatory tests data.
Similar content being viewed by others
References
Farag, A.A., Khalifa, E.M., Sadik, N.A., Abbas, S.Y., Al-Sehemi, A.G., and Ammar, Y.A., Med. Chem. Res., 2013, vol. 22, p. 440. doi https://doi.org/10.1007/s00044-012-0046-6
Sharma, S., Srivastava, V.K. and Kumar, A., Eur. J. Med. Chem., 2002, vol. 37, p. 689. doi https://doi.org/10.1016/S0223-5234(02)01340-5
Aggarwal, R., Bansal, A., Rozas, I., Kelly, B., Kaushik, P., and Kaushik, D., Eur. J. Med. Chem., 2013, vol. 70, p. 350. doi https://doi.org/10.1016/j.ejmech.2013.09.052
Kumar, R.S., Arif, I.A., Ahamed, A., and Idhayadhulla, A., Saudi J. Biolo. Sci. 2016, vol. 23, p. 614. doi https://doi.org/10.1016/j.sjbs.2015.07.005
Karrouchi, K., Radi, S., Ramli, Y., Taoufik, J., Mabkhot, Y., and Al-aizari, F., Molecules 2018, vol. 23, p. 134. doi https://doi.org/10.3390/molecules23010134
Lipman, A.G., Internat. J. Pharm. Pract, 1993, vol. 2, p. 124. doi https://doi.org/10.1111/j.2042-7174.1993.tb00740.x.
Hu, Z.-F., Chen, L.-L., Qi, J., Wang, Y.-H., Zhang, H., and Yu, B.-Y. Fitoterapia, 2011, vol. 82, p. 190. doi https://doi.org/10.1016/j.fitote.2010.09.002.
Diaz, P., Phatak, S.S., Xu, J., Fronczek, F. R., Astruc-Diaz, F., Thompson, C.M., Cavasotto, C.N., and Naguib, M., Chem. Med. Chem.: Chem. Enabl. Drug Discov., 2009, vol. 4, p. 1615. doi https://doi.org/10.1002/cmdc.200900226
Shakya, A.K., Kamal, M., Balaramnavar, V.M., Bardaweel, S.K., Naik, R.R., Saxena, A.K., and Siddiqui, H., Acta Pharma, 2016, vol. 66, p. 353. doi https://doi.org/10.1515/acph-2016-0023
Rindhe, S., Rode, M., and Karale, B., Indian J. Pharm. Sci., 2010, vol. 72, p. 231. doi https://doi.org/10.4103/0250-474X.65022
Ebiike, H., Masubuchi, M., Liu, P., Kawasaki, K.-I., Morikami, K., Sogabe, S., Hayase, M., Fujii, T., Sakata, K., Shindoh, H., Shiratoria, Y., Aoki, Y., Ohtsuka, T., and Shimma, N., Bioorg. Med.C hem. Lett., 2002, vol. 12, p. 607. doi https://doi.org/10.1016/S0960-894X(01)00808-3
Usachev, B.I., Obydennov, D.L., Röschenthaler, G.-V., and Sosnovskikh, V.Y., Org. Lett., 2008, vol. 10, p. 2857. doi https://doi.org/10.1021/ol800992z
Usachev, B.I., Obydennov, D.L., Röschenthaler, G.-V., and Sosnovskikh, V.Y., J. Fluorine Chem., 2012, vol. 137, p. 22. doi https://doi.org/10.1016/j.jfluchem.2012.01.006
Ewies, E.F., El-Shehry, M.F., and Boulos, L.S., Int. J. ChemTech. Res., 2014–2015, vol. 7, no. 5, p. 2506
Gouhar, R.S., Ewies, E.F., El-Shehry, M.F., Shaheen, M.N., and Ibrahim, E.M.M., J. Heterocycl. Chem., 2018, vol. 55, p. 2368, doi https://doi.org/10.1002/jhet.3301
El-Shehry, M.F., Abu-Zied, K.M., Ewies, E.F., Awad, S.M., and Mohram, M.E., Der Pharma Chem., 2013, 5(5), p. 318
Farag, A.A., El Shehry, M.F., Abbas, S.Y., Abd-Alrahman, S.N., Atrees, A.A., Albasheer, H.Z., and Ammar, Y.A., Z. Naturforsch. B, 2015, vol. 70, p. 519. doi https://doi.org/10.1515/znb-2015-0009
Wang, Z., Claisen-Schmidt Condensation. Comprehensive Organic Name Reactions and Reagents, Hoboken, NJ: John Wiley & Sons, 2010. doi https://doi.org/10.1002/9780470638859
Nalamachu, S. and Wortmann, R., Postgraduate Med., 2014, vol. 126, p. 92. doi https://doi.org/10.3810/pgm.2014.07.2787
Winter, C.A., Risley, E.A., and Nuss, G.W., Exper.Biol. Med., 1962, vol. 111, p. 544. doi https://doi.org/10.3181/00379727-111-27849
Matthews, J., Berry, G., and Armitage, P., Statistical Methods in Medical Research, Oxford: Blackwell Science, 2002, 4 ed.
Eddy, N.B. and Leimbach, D., J. Pharma. Exp. Ther., 1953, vol. 107, p. 385. PubMed: 13035677.
Halgren, T.A., J. Comp. Chem. 1996, vol. 17, p. 490. doi https://doi.org/10.1002/(SICI)1096-987X(199604)17:5/6<490:: AID-JCC1>3.0.CO,2-P
Morris, G.M., Goodsell, D.S., Halliday, R.S., Huey, R., Hart, W.E., Belew, R.K., and Olson, A.J., J. Comp. Chem., 1998, vol. 19, p. 1639. doi https://doi.org/10.1002/(SICI)1096-987X(19981115)19:14<1639::AID-JCC10>3.0.CO,2-B
Solis, F.J. and Wets, R.J.-B., Math. Oper. Res., 1981, vol. 6, p. 19. doi 0364-765X/81/0601/0019$01.25
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
El Shehry, M.F., Ewies, E.F. & Zayed, E.M. Synthesis of New Pyrazole Derivatives, Their Anti-Inflammatory and Analgesic Activities, and Molecular Docking Studies. Russ J Gen Chem 89, 492–498 (2019). https://doi.org/10.1134/S1070363219030216
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363219030216