Abstract
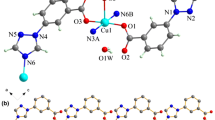
A new coordination polymer of terbium tartrate [Tb(H2O)3(C4H5O6)(C4H4O6)] has been synthesized and crystallizes in the polar space group P41 with cell constants a = 6.0415(9), b = 6.0415(9), c = 36.516(7) Å, V = 1332.8(4) Å3, Z = 4. The terbium(III) ion of title complex is nine-coordinate through oxygen donors. Four different coordination modes of tartrate occur. This Tb(III) complex exhibits a characteristic luminescence in the visible region upon excitation at 353 nm. The temperature-dependent magnetic properties of the Tb(III) complex were investigated in the temperature range of 2–300 K. Title compound exhibits significant ferroelectric properties at room temperature (remnant polarization 2P r = 0.160 μC cm−2, coercive field 2E c = 44.5 kV cm−1, saturation of the spontaneous polarization P s = 0.176 μC cm−2).
Similar content being viewed by others
References
Torres, M.E., Peraza, J., Yanes, A.C., et al., J. Phys. Chem. Solids, 2002, vol. 63, p. 491.
Fousek, J., Cross, L.E., and Seely, K., Ferroelectrics, 1970, vol. 1, p. 63.
Torres, M.E., Peraza, J., Yanes, A.C., et al., J. Appl. Phys., 1998, vol. 84, p. 5729.
Medrano, C., Gunter, P., and Arend, H., Phys. Status Solidi, B, 1997, vol. 143, p. 749.
Watanabe, O., Noritake, T., Hirose, Y., et al., J. Mater. Chem., 1993, vol. 3, p. 1053.
Zyss, J., Masse, R., Beucher-Bagieu, M., et al., Adv. Mater., 1993, vol. 5, p. 120.
Ovchinnikova, G.I., Gavrilova, N.D., and Lotonov, A.M., Izv. Akad. Nauk., Ser. Fiz., 1997, vol. 61, p. 2431.
Arora, S.K., Patel, V., Chudasama, B., et al., J. Cryst. Growth, 2005, vol. 275, p. e657.
Suryanarayana, K. and Dharmaprakash, S.M., Mater. Lett., 2000, vol. 42, p. 92.
Arora, S.K., Patel, V., Kothari, A., et al., Cryst. Growth. Des., 2004, vol. 4, p. 343.
Suryanarayana, K. and Dharmaprakash, S.M., Mater. Chem. Phys., 2003, vol. 77, p. 179.
Tanase, S. and Reedijk, J., Coord. Chem. Rev., 2006, vol. 250, p. 2501.
Handa, S., Nagawa, K., Sohtome, Y., et al., Angew. Chem. Int. Ed., 2008, vol. 47, p. 3230.
Xia, J., Zhao, B., Wang, H.S., et al., Inorg. Chem., 2007, vol. 46, p. 3450.
Manna, S.C., Zangrando, E., Ribas, J., et al., Polyhedron, 2006, vol. 25, p. 1779.
Athar, M., Li, G.H., Shi, Z., et al., Solid State Sci., 2008, vol. 10, p. 1853.
Yan, P., Xing, J.C., Li, G.M., et al., J. Coord. Chem., 2009, vol. 62, p. 2095.
Sheldrick, G.M., SHELXS-97, Program for Crystal Stucture Refinement and SHELXL-97, Program for Crystal Stucture Solution, Göttingen (Germany): Univ. of Göttingen, 1997.
Wu, C.D., Zhan, X.P., Lu, C.Z., et al., Acta Crystallogr., E, 2002, vol. 58, p. m228.
Hawrhorne, F.C., Borys, I., and Ferguson, R.B., Acta Crystallogr., C, 1983, vol. 39, p. 540.
Nakamoto, K., Infrared and Raman Spectra of Inorganic and Coordination Compounds, Pt. B, New York: Interscience-Wiley, 2009.
Perry, T.T. and Kurtz, S.K., J. Appl. Phys., 1968, vol. 39, p. 3798.
Halasyamani, P.S. and Poeppelmeier, K.R., Chem. Mater., 1998, vol. 10, p. 2753.
Solans, X., Gonzalez-Silgo, C., and Ruiz-Pérez, C., J. Solid State Chem., 1997, vol. 131, p. 350.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Xu, W., Chang, H.S., Liu, W. et al. Synthesis, crystal structure, and properties of a new lanthanide tartrate coordination polymer. Russ J Coord Chem 40, 251–256 (2014). https://doi.org/10.1134/S1070328414030105
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070328414030105