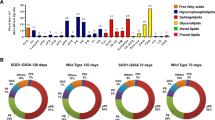
Abstract
Changes in the expression of genes for enzymes of sphingolipid metabolism in transgenic FUS mice (FUS (1-359)) simulating amyotrophic lateral sclerosis (ALS) were found. The profiles of molecular species of sphingomyelins (SM) and ceramides (CER) in the structure of the brain and in the spinal cord were analyzed. During the development of ALS, changes in the spectrum of molecular types of CER were noted only in the spinal cord, where almost all CER decreased in relation to the control after two and three months of the course of the disease. After four months of the development of the pathology, an increase in the expression of the acid ceramidase gene was observed, as a result of which sphingosine can be formed, which has pronounced proapoptotic properties. Analysis of gene expression of CER and galactosylceramide (GalCER) metabolism enzymes indicates the possibility of GalCER functioning as a source for maintaining CER levels at the terminal stage of ALS. Thus, the metabolism of sphingolipids is a promising field of study of the processes associated with ALS, both in the context of the search for potential diagnostic markers and effective drugs, and for explaining the pathogenesis of ALS.








Similar content being viewed by others
REFERENCES
Talbott, E.O., Malek, A.M., and Lacomis, D., Handb. Clin. Neurol., 2016, vol. 138, pp. 225–238. https://doi.org/10.1016/B978-0-12-802973-2.00013-6
Arthur, K.C., Calvo, A., Price, T.R., Geiger, J.T., Chio, A., and Traynor, B.J., Nat. Commun., 2016, vol. 7, p. 12408. https://doi.org/10.1038/ncomms12408
Chia, R., Chiò, A., and Traynor, B.J., Lancet Neurol., 2018, vol. 17, pp. 94–102. https://doi.org/10.1016/S1474-4422(17)30401-5
Zou, Z.Y., Zhou, Z.R., Che, C.H., Liu, C.Y., He, R.L., and Huang, H.P.J., Neurol. Neurosurg. Psychiatry, 2017, vol. 88, pp. 540–549. https://doi.org/10.1136/jnnp-2016-315018
Shi, P., Gal, J., Kwinter, D.M., Liu, X., and Zhu, H., Biochim. Biophys. Acta, 2010, vol. 1802, pp. 45–51. https://doi.org/10.1016/j.bbadis.2009.08.012
Peters, O.M., Ghasemi, M., and Brown, R.H., Jr., J. Clin. Invest., 2015, vol. 125, pp. 1767–1779. https://doi.org/10.1172/JCI71601
Van Damme, P., Dewil м., robberecht w., van den bosch l, Neurodegener. Dis., 2005, vol. 2, pp. 147–159. https://doi.org/10.1159/000089620
Chitnis, T. and Weiner, H.L., J. Clin. Invest., 2017, vol. 127, pp. 3577–3587. https://doi.org/10.1172/JCI90609
Hardiman, O., Al-Chalabi, A., Chio, A., Corr, E.M., Logroscino, G., Robberecht, W., Shaw, P.J., Simmons, Z., and Berg, L.H., Nat. Rev. Dis. Primers, 2017, vol. 3, p. 17085. https://doi.org/10.1038/nrdp.2017.85
van Damme, P., Robberecht, W., and Van Den, BoschL., Dis. Model Mech., 2017, vol. 10, pp. 537–549. https://doi.org/10.1242/dmm.029058
Van Den, BoschL., J. Biomed. Biotechnol., 2011, vol. 2011, p. 348765. https://doi.org/10.1155/2011/348765
Naumann, M., Pal, A., Goswami, A., Lojewski, X., Japtok, J., Vehlow, A., Naujock, M., Gunther, R., Jin, M., Stanslowsky, N., Reinhardt, P., Sterneckert, J., Frickenhaus, M., Pan-Montojo, F., Storkebaum, E., Poser, I., Freischmidt, A., Weishaupt, J.H., Holzmann, K., Troost, D., Ludolph, A.C., Boeckers, T.M., Liebau, S., Petri, S., Cordes, N., Hyman, A.A., Wegner, F., Grill, S.W., Weis, J., Storch, A., and Hermann, A., Nat. Commun., 2018, vol. 9, p. 335. https://doi.org/10.1038/s41467-017-02299-1
Vance, C., Rogelj, B., Hortobagyi, T., De Vos, K.J., Nishimura, A.L., Sreedharan, J., Hu, X., Smith, B., Ruddy, D., Wright, P., Ganesalingam, J., Williams, K.L., Tripathi, V., Al-Saraj, S., Al-Chalabi, A., Leigh, P.N., Blair, I.P., Nicholson, G., de Belleroche, J., Gallo, J.M., Miller, C.C., and Shaw, C.E., Science, 2009, vol. 323, pp. 1208–1211. https://doi.org/10.1126/science.1165942
Kwiatkowski, T.J., Jr., Bosco, D.A., Leclerc, A.L., Tamrazian, E., Vanderburg, C.R., Russ, C., Davis, A., Gilchrist, J., Kasarskis, E.J., Munsat, T., Valdmanis, P., Rouleau, G.A., Hosler, B.A., Cortelli, P., de Jong, P.J., Yoshinaga, Y., Haines, J.L., Pericak-Vance, M.A., Yan, J., Ticozzi, N., Siddique, T., McKenna-Yasek, D., Sapp, P.C., Horvitz, H.R., Landers, J.E., and Brown, R.H., Jr., Science, 2009, vol. 323, pp. 1205–1208. https://doi.org/10.1126/science.1166066
Conlon, E.G. and Manley, J.L., Genes Dev., 2017, vol. 31, pp. 1509–1528. https://doi.org/10.1101/gad.304055.117
Skvortsova, V.I., Mal’tsev, A.V., Grigor’ev, V.V., Ovchinnikov, R.K., Roman, A.Yu., Efimova, A.D., and Kovrazhkina, E.A., Mol. Biol. (Moscow), 2017, vol. 51, pp. 341–351. https://doi.org/10.7868/S0026898417020094
Arima, H., Omura, T., Hayasaka, T., Masaki, N., Hanada, M., Xu, D., Banno, T., Kobayashi, K., Takeuchi, H., Kadomatsu, K., Matsuyama, Y., and Setou, M., Neuroscience, 2015, vol. 297, pp. 127–136. https://doi.org/10.1016/j.neuroscience.2015.03.060
Fitzgerald, K.C., O’Reilly, E.J., Falcone, G.J., McCullough, M.L., Park, Y., Kolonel, L.N., and Ascherio, A., JAMA Neurol., 2014, vol. 71, pp. 1102–1110. https://doi.org/10.1001/jamaneurol.2014.1214
Blasco, H., Veyrat-Durebex, C., Bocca, C., Patin, F., Vourc’h, P., Kouassi Nzoughet, J., Lenaers, G., Andres, C.R., Simard, G., Corcia, P., and Reynier, P., Sci. Rep., 2017, vol. 7, p. 17652. https://doi.org/10.1038/s41598-017-17389-9
Dodge, J.C., Treleaven, C.M., Pacheco, J., Cooper, S., Bao, C., Abraham, M., Cromwell, M., Sardi, S.P., Chuang, W.L., Sidman, R.L., Cheng, S.H., and Shihabuddin, L.S., Proc. Natl. Acad. Sci. U. S. A., 2015, vol. 112, pp. 8100–8105. https://doi.org/10.1042/bst0320144
van Echten-Deckert, G. and Herget, T., Biochim. Biophys. Acta, 2006, vol. 1758, pp. 1978–1994. https://doi.org/10.1016/j.bbamem.2006.06.009
Piccinini, M., Scandroglio, F., Prioni, S., Buccinnà, B., Loberto, N., Aureli, M., Chigorno, V., Lupino, E., DeMarco, G., Lomartire, A., Rinaudo, M.T., Sonnino, S., and Prinetti, A., Mol. Neurobiol., 2010, vol. 41, pp. 314–340. https://doi.org/10.1007/s12035-009-8096-6
Hannun, Y.A. and Obeid, L.M., Nat. Rev. Mol. Cell Biol., 2008, vol. 9, pp. 139–150. https://doi.org/10.1038/nrm2329
Hannun, Y.A. and Obeid, L.M., Nat. Rev. Mol. Cell Biol., 2018, vol. 19, pp. 175–191. https://doi.org/10.1038/nrm.2017.107
Nganga, R., Oleinik, N., and Ogretmen, B., Adv. Cancer Res., 2018, vol. 140, pp. 1–25. https://doi.org/10.1016/bs.acr.2018.04.007
Chaurasia, B. and Summers, S.A., Annu. Rev. Physiol., 2021, vol. 83, pp. 303–330. https://doi.org/10.1146/annurev-physiol-031620-093815
Zhang, X., Matsuda, M., Yaegashi, N., Nabe, T., and Kitatani, K., Cells, 2020, vol. 9, p. 627. https://doi.org/10.3390/cells9030627
Mullen, T.D., Hannun, Y.A., and Obeid, L.M., Biochem. J., 2012, vol. 441, pp. 789–802. https://doi.org/10.1042/bj20111626
Zelnik, I.D., Ventura, A.E., Kim, J.L., Silva, L.C., and Futerman, A.H., Biochim. Biophys. Acta, Mol. Cell Biol. Lipids, 2020, vol. 1865, p. 158489. https://doi.org/10.1016/j.bbalip.2019.06.015
Futerman, A.H. and Hannun, Y.A., EMBO Rep., vol. 5, pp. 777–782. https://doi.org/10.1038/sj.embor.7400208
Cutler, R.G., Pedersen, W.A., Camandola, S., Rothstein, J.D., and Mattson, M.P., Ann. Neurol., 2002, vol. 52, pp. 448–457.
Potenza, R.L., De Simone, R., Armida, M., Mazziotti, V., Pèzzola, A., Popoli, P., and Minghetti, L., Neurotherapeutics, 2016, vol. 13, pp. 918–927. https://doi.org/10.1007/s13311-016-0462-2
Henriques, A., Croixmarie, V., Priestman, D.A., Rosenbohm, A., Dirrig-Grosch, S., D’Ambra, E., Huebecker, M., Hussain, G., Boursier-Neyret, C., Echaniz-Laguna, A., Ludolph, A.C., Platt, F.M., Walther, B., Spedding, M., Loeffler, J.P., and Gonzalez De Aguilar, J.L., Hum. Mol. Genet., 2015, vol. 24, pp. 7390–7405. https://doi.org/10.1093/hmg/ddv439
Deikin, A.V., Kovrazhkina, E.A., Ovchinnikov, R.K., Bronovitskii, E.V., Razinskaya, O.D., Smirnov, A.P., Ermolkevich, T.G., Elyakov, A.B., Popov, A.N., Fedorov, E.N., Lytkina, O.A., Kukharskii, M.S., Tarasova, T.V., Shelkovnikova, T.A., Ustyugov, A.A., Ninkina, N.N., Gol’dman, I.L., Sadchikov, E.R., Bachurin, S.O., and Skvortsova, V.I., Zh. Nevrol. Psikhiatr. im. S.S. Korsakova, 2014, vol. 114, pp. 63–70.
Giusto, N.M., Roque, M.E., Ilincheta, De., and Boschero, M.G., Lipids, 1992, vol. 27, pp. 835–839. https://doi.org/10.1007/BF02535859
Di Pardo, A., Basit, A., Armirotti, A., Amico, E., Castaldo, S., Pepe, G., Marracino, F., Buttari, F., Digilio, A.F., and Maglione, V., Front. Neurosci., 2017, vol. 11, p. 698. https://doi.org/10.3389/fnins.2017.00698
Lin, G., Wang, L., Marcogliese, P.C., and Bellen, H.J., Trends Endocrinol. Metab., 2019, vol. 30, pp. 106–117. https://doi.org/10.1016/j.tem.2018.11.003
Czubowicz, K., Jesko, H., Wencel, P., Lukiw, W.J., and Strosznajder, R.P., Mol. Neurobiol., 2019, vol. 56, pp. 5436–5455. https://doi.org/10.1007/s12035-018-1448-3
Brugg, B., Michel, P.P., Agid, Y., and Ruberg, M., J. Neurochem., 1996, vol. 66, pp. 733–739. https://doi.org/10.1046/j.1471-4159.1996.66020733.x
France-Lanord, V., Brugg, B., Michel, P.P., Agid, Y., and Ruberg, M., J. Neurochem., 1997, vol. 69, pp. 1612–1621. https://doi.org/10.1046/j.1471-4159.1997.69041612.x
Bras, J., Singleton, A., Cookson, M.R., and Hardy, J., FEBS J., 2008, vol. 275, pp. 5767–5773. https://doi.org/10.1111/j.1742-4658.2008.06709.x
Gutner, U.A., Shupik, M.A., Maloshitskaya, O.A., Sokolov, S.A., Rezvykh, A.P., Funikov, S.Yu., Lebedev, A.T., Ustyugov, A.A., and Alesenko, A.V., Biochemistry (Moscow), 2019, vol. 84, pp. 1166–1176. https://doi.org/10.1134/S0006297919100055
Filippov, V., Song, M.A., Zhang, K., Vinters, H.V., Tung, S., Kirsch, W.M., Yang, J., and Duerksen-Hughes, P.J., J. Alzheimer’s Dis., 2012, vol. 29, pp. 537–547. https://doi.org/10.3233/JAD-2011-111202
Siskind, L.J., Kolesnick, R.N., and Colombini, M., J. Biol. Chem., 2002, vol. 277, pp. 26796–267803. https://doi.org/10.1074/jbc.M200754200
Yonutas, H.M., Pandya, J.D., and Sullivan, P.G., J. Bioenerg. Biomembr., 2015, vol. 47, pp. 149–154. https://doi.org/10.1007/s10863-014-9593-5
Brown, M.R., Sullivan, P.G., and Geddes, J.W., J. Biol. Chem., 2006, vol. 281, pp. 11658–11668. https://doi.org/10.1074/jbc.M510303200
Brown, M.R., Geddes, J.W., and Sullivan, P.G., J. Bioenerg. Biomembr., 2004, vol. 36, pp. 401–406. https://doi.org/10.1023/B:JOBB.0000041775.10388.23
Sullivan, P.G., Rabchevsky, A.G., Keller, J.N., Lovell, M., Sodhi, A., Hart, R.P., and Scheff, S.W., J. Comp. Neurol., 2004, vol. 474, pp. 524–534. https://doi.org/10.1002/cne.20130
Bienias, K., Fiedorowicz, A., Sadowska, A., Prokopiuk, S., and Car, H., Pharmacol. Rep., vol. 668, pp. 570–581. https://doi.org/10.1016/j.pharep.2015.12.008
Berry, J.D., Paganoni, S., Atassi, N., Macklin, E.A., Goyal, N., Rivner, M., Simpson, E., Appel, S., Grasso, D.L., Mejia, N.I., Mateen, F., Gill, A., Vieira, F., Tassinari, V., and Perrin, S., Muscle Nerve, 2017, vol. 56, pp. 1077–1084. https://doi.org/10.1002/mus.25733
Pharaoh, G., Sataranatarajan, K., Street, K., Hill, S., Gregston, J., Ahn, B., Kinter, C., Kinter, M., and Van Remmen, H., Front. Neurosci., 2019, vol. 13, p. 487. https://doi.org/10.3389/fnins.2019.00487
Funikov, S.Y., Rezvykh, A.P., Mazin, P.V., Morozov, A.V., Maltsev, A.V., Chicheva, M.M., Vikhareva, E.A., Evgen’ev, M.B., and Ustyugov, A.A., Neurogenetics, 2018, vol. 19, pp. 189–204. https://doi.org/10.1007/s10048-018-0553-9
Krasnov, G.S., Dmitriev, A.A., Kudryavtseva, A.V., Shargunov, A.V., Karpov, D.S., Uroshlev, L.A., Melnikova, N.V., Blinov, V.M., Poverennaya, E.V., Archacov, A.I., Lisitsa, A.V., and Ponomarenko, E.A., J. Proteome Res., 2015, vol. 14, pp. 3729–3737. https://doi.org/10.1021/acs.jproteome.5b00490
Bolger, A.M., Lohse, M., and Usadel, B., Bioinformatics, 2014, vol. 30, pp. 2114–2120. https://doi.org/10.1093/bioinformatics/btu170
Dobin, A., Davis, C.A., Schlesinger, F., Drenkow, J., Zaleski, C., Jha, S., Batut, P., Chaisson, M., and Gingeras, T.R., Bioinformatics, 2013, vol. 29, pp. 15–21. https://doi.org/10.1093/bioinfor-matics/bts635
Anders, S., Pyl, P.T., and Huber, W., Bioinformatics, 2015, vol. 31, pp. 166–169. https://doi.org/10.1101/002824
Robinson, M.D., McCarthy, D.J., and Smyth, G.K., Bioinformatics, 2010, vol. 26, pp. 139–140. https://doi.org/10.1093/bioinformatics/btp616
R Core Team, R Language Definition, R Foundation for Statistical Computing, Vienna, Austria, 2000.
Thissen, D., Steinberg, L., and Kuang, D., J. Educ. Behav. Stat., 2002, vol. 27, pp. 77–83. https://doi.org/10.3102/10769986027001077
Wickham, H., Wiley Interdiscip. Rev., Comput. Stat., 2011, vol. 3, pp. 180–185. https://doi.org/10.1002/wics.147
Bligh, T.G. and Dyer, W.J., Can. J. Phys., 1959, vol. 37, pp. 911–917. https://doi.org/10.1139/o59-099
Sullards, M.C., Methods Enzymol., 2000, vol. 312, pp. 32–45. https://doi.org/10.1016/S0076-6879(00)12898-8
ACKNOWLEDGMENTS
Maintenance of the animals was provided by the Program for Support of Bioresource Collections of the Institute of Physiologically Active Substances of the Russian Academy of Sciences and carried out on the equipment of the Center for Collective Use of the Institute of Physiologically Active Substances of the Russian Academy of Sciences.
The analysis of high-throughput sequencing data was carried out on the equipment of the Genome Shared Use Center of the Engelhardt Institute of Molecular Biology, RAS (http://www.eimb.ru/rus/ckp/ccu_genome_c.php).
Funding
The study of neurodegenerative processes in model animals of the FUS(1-359) line was carried out with the support of the Russian Science Foundation (project no. 19-13-00378).
The animals were kept within the framework of the state assignment of the Institute of Physiologically Active Substances of the Russian Academy of Sciences no. 0090-2017-0019.
Studies of the properties of lipids in neuropathology were carried out within the framework of the state assignment of the Institute of Physiologically Active Substances of the Russian Academy of Sciences (topic no. 44.4, state registration number 01201253310).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of InterestsThe authors declare no conflicts of interest.
All applicable international, national, and institutional guidelines for the care and use of animals have been followed.
Additional information
Translated by N. Onishchenko
Abbreviations: ALS, amyotrophic lateral sclerosis; GalCER, galactosylceramide; SphL, sphingolipids; SM, sphingomyelin; CER, ceramide; 3-KSR, 3-ketodihydrosphyngoreductase; Asah, ceramidase; Asah1, acid ceramidase; aSMase, acid sphingomyelinase; CerS, ceramide synthase; DES, dihydroceramide desaturase; FUS(1-359), FUS transgenic mice; GALC, galactosylceramidase; nSMase, neutral sphingomyelinase; SMase, sphingomyelinase; SMS, sphingomyelinase synthase; SOD1, superoxide dismutase; SPT, serine palmitoyl transferase.
Corresponding author; phone: +7 (905) 588-50-66.
Rights and permissions
About this article
Cite this article
Shupik, M.A., Gutner, U.A., Ustyugov, A.A. et al. Changes in the Metabolism of Sphingomyelin and Ceramide in the Brain Structures and Spinal Cord of Transgenic Mice (FUS(1-359)) Modeling Amyotrophic Lateral Sclerosis. Russ J Bioorg Chem 48, 178–189 (2022). https://doi.org/10.1134/S1068162022010137
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162022010137