Abstract
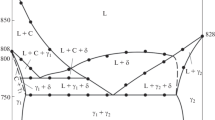
The PbSb2Se4–Pb5Bi6Se14 section of the Sb2Se3–PbSe–Bi2Se3 quasi-ternary system was studied by integrated physicochemical analysis methods, and the state diagram of this section was constructed. The PbSb2Se4–Pb5Bi6Se14 section was found to be a partially quasi-binary section of the Sb2Se3–PbSe–Bi2Se3 quasi-ternary system. A region of solid solutions based on PbSb2Se4 at room temperature was detected at 10 mol % Pb5Bi6Se14. At a ratio between the initial components of 1: 1, congruently melting quaternary compound Pb6Sb2Bi6Se18 forms. Its single crystals were produced by the method of chemical transport reactions. Pb6Sb2Bi6Se18 crystallizes in the rhombic system with the unit cell parameters a = 14.43 Å, b = 21.42 Å, c = 3.90 Å, V0 = 1205 Å3, Z = 4, and space group Pnnm. The thermodynamic functions of Pb6Sb2Bi6Se18 were calculated: the standard entropy S2980 = 1472.8 J mol–1 K–1, the enthalpy ΔH2980 =–1215.8 kJ mol–1, and the Gibbs free energy of formation ΔG2980 =–1193.6 kJ mol–1.
Similar content being viewed by others
References
M. G. Konatridis, Acc. Chem. Res., 38, 361 (2005).
A. R. West, Solid State Chemistry and Its Applications, 2nd ed. (Wiley, New York, 2014).
L. D. Ivanova, L. I. Petrova, Yu. V. Granatkina, et al., Inorg. Mater. 52, 248 (2016).
R. D. Kurbanova, A. N. Mamedov, A. M. Alidzhanov, and S. G. Agdamskaya, Inorg. Mater, 38, 652 (2002).
S. M. Asadov, A. N. Mamedov, and S. A. Kulieva, Inorg. Mater. 38, 876 (2016).
M. Caputo, M. Panighel, S. Lisi, et al., Nano Lett. 16, 3409 (2016).
L. Viti, D. Coquillat, A. Politano, et al., Nano Lett. 16, 80 (2016).
N. A. Tulina, I. Yu. Borisenko, I. M. Shmytko, et al., Phys. Lett. A. 376A (45), 3398 (2012).
Z. Y. Wang, H. D. Li, X. Guo, et al., J. Cryst. Growth 334, 96 (2011).
E. I. Elagina, in Proceedings of the IV Conference on Semiconductor Materials: Problems of Metallurgy and Physics of Semiconductors (AN SSSR, Moscow, 1961), p. 153 [in Russian].
K. A. Agaev, A. G. Talybov, and S. A. Semiletov, Kristallografiya 11, 736 (1966).
K. A. Agaev and S. A. Semiletov, Kristallografiya 13, 258 (1968).
R. M. Imamov, S. A. Semiletov, and Z. G. Pinsker, Kristallografiya 15, 287 (1970).
Y. Zhang, A. P. Wilkinson, P. L. Lee, et al., J. Appl. Crystallogr. 38, 433 (2005).
L. E. Shelimova, O. G. Karpinskii, and V. S. Zemskov, Inorg. Mater. 44, 927 (2008).
L. E. Shelimova, O. G. Karpinskii, P. P. Konstantinov, et al., Inorg. Mater. 46, 120 (2010).
L. E. Shelimova, V. N. Tomashik, and V. I. Grytsiv, State Diagrams in Semiconductor Materials Science (Systems Based on Chalcogenides Si, Ge, Sn, and Pb): A Reference Book, Ed. by V. S. Zemskov (Nauka, Moscow, 1991) [in Russian].
A. Skowron, F. W. Boswel, J. M. Corbett, and N. J. Taylor, J. Solid State Chem. 112, 251 (1994).
A. Skowron and I. D. Brown, Acta Crystallogr. C 46, 2287 (1990).
M. Emirdag-Eanes and J. W. Kolis, Z. Anorg. Allg. Chem. 628, 10 (2002).
V. V. Nagaev, V. P. Orlovskaya, and I. V. Gananaev, Izv. Akad. Nauk SSSR, Neorg. Mater. 20, 937 (1984).
Extended Abstracts of the VI International Conference on Crystal Growth: Growth from Gas Phase (Nauka, Moscow, 1980), Vol. 1 [in Russian].
Semiconductors, Ed. by N. B. Hannay (Reinhold, New York, 1960).
A. N. Mamedov, Thermodynamics of Systems with Nonmolecular Compounds: Calculation and Approximation of Thermodynamic Functions and Phase Diagrams (LAP Lambert Academic Publishing, Germany, 2015) [in Russian].
PURE 4.4. SGTE Pure Elements (Unary) Database (Scientific Group Thermodata Europe 1991–2006).
Physicochemical Properties of Semiconductor Substances: A Handbook, Ed. by A. V. Novoselova and V. B. Lazarev (Nauka, Moscow, 1978) [in Russian].
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © G.R. Gurbanov, Sh.G. Mamedov, M.B. Adygezalova, A.N. Mamedov, 2017, published in Zhurnal Neorganicheskoi Khimii, 2017, Vol. 62, No. 12, pp. 1655–1660.
Rights and permissions
About this article
Cite this article
Gurbanov, G.R., Mamedov, S.G., Adygezalova, M.B. et al. The PbSb2Se4–Pb5Bi6Se14 section of the Sb2Se3–PbSe–Bi2Se3 quasi-ternary system. Russ. J. Inorg. Chem. 62, 1659–1664 (2017). https://doi.org/10.1134/S0036023617120099
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023617120099