Abstract
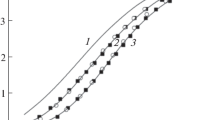
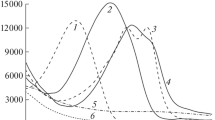
The substitution equilibria AuCl −2 + iNH +4 = Au(NH3)iCl2 − i + iCl− + iH+, β *i . were studied pH-metrically at 25°C and I = 1 mol/L (NaCl) in aqueous solution. It was found that logβ *1 = −5.10±0.15 and logβ *2 = −10.25±0.10. For equilibrium AuNH3Clsolid = AuNH3Cl, log K s = −3.1±0.3. Taking into account the protonation constants of ammonia (log K H = 9.40), the obtained results show that for equilibria AuCl −2 + iNH3 = Au(NH3)iCl2 − i + iCl−, logβ1 = 4.3±0.2, and logβ2 = 8.55±0.15. The standard potentials E 1/00 of AuNH3Cl0 and Au(NH3) +2 species are equal to 0.90±0.02 and 0.64±0.01 V, respectively.
Similar content being viewed by others
References
H. Skibsted and J. Bjerrum, Acta Chem. Scand. 28(7), 764 (1974).
V. I. Belevantsev, I. V. Mironov, and B. I. Peshchevitskii, Zh. Neorg. Khim., No. 1, 53 (1982).
J. Berglund and L. I. Elding, Inorg. Chem. 34(2), 513 (1995).
I. V. Mironov and V. I. Belevantsev, Zh. Neorg. Khim. 50(7), 1210 (2005) [Russ. J. Inorg. Chem. 50 (7), 1115 (2005)].
J. W. Mellor, A Comprehensive Treatise on Inorganic and Theoretical Chemistry (Longmans, Green and Co, London, 1947), Vol. 3, p. 589.
V. I. Belevantsev and B. I. Peshchevitskii, Investigations of Complex Solution Equilibria (Nauka, Novosibirsk, 1978) [in Russian].
M. Maeda, G. Nakagawa, and G. Biederman, J. Phys. Chem. 87(1), 121 (1983).
J. Bjerrum, Metal Ammine Formation in Aqueous Solution (Haase, Copenhagen, 1957; Inostrannaya Literatura, Moscow, 1961).
V. I. Belevantsev, B. I. Peshchevitskii, and S. V. Zemskov, Izv. Sib. Otd. Akad. Nauk SSSR, Ser. Khim. Nauk No. 4 (2), 24 (1976).
Author information
Authors and Affiliations
Additional information
Original Russian Text © I.V. Mironov, 2007, published in Zhurnal Neorganicheskoi Khimii, 2007, Vol. 52, No. 6, pp. 1032–1034.
Rights and permissions
About this article
Cite this article
Mironov, I.V. Stability of diammine and chloroammine gold(I) complexes in aqueous solution. Russ. J. Inorg. Chem. 52, 960–962 (2007). https://doi.org/10.1134/S003602360706023X
Received:
Issue Date:
DOI: https://doi.org/10.1134/S003602360706023X