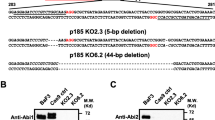
Abstract
In the present study, we have applied the siRNA approach to reduce the expression of AML1-ETO and RUNX1(K83N) oncogenes, which are frequently found in leukemic cells. We have designed small hairpin RNAs (shRNA) for targeting AML1-ETO oncogene and a region close to the 5′-untranslated region of mRNA for the mutant RUNX1(K83N) oncogene and expressed the shRNAs in lentiviral vectors. We report a stable reduction in expression of oncogenes following the introduction of shRNAs into cells.
Similar content being viewed by others
Abbreviations
- AML:
-
Acute Myeloid Leykosis
- FAB:
-
Franco-American-British classification of leucosis
- HSC:
-
Hemopoietic Stem Cell
- siRNA:
-
small interfering RNA
- shPHK:
-
small hairpin interfering RNA.
References
Weinberg R.A. 2007. The Biology of Cancer. NY: Garland Science.
Jemal A., Siegel R., Ward E., Hao Y., Xu J., Thun M.J. 2009. Cancer statistics. Ca. Cancer. J. Clin. 59, 225–249.
Peterson L.F., Zhang D.E. 2004. The 8;21 translocation in leukemogenesis. Oncogene. 23, 4255–4562.
Imai Y., Kurokawa M., Izutsu K., et al. 2000. Mutations of the AML1 gene in myelodysplastic syndrome and their functional implications in leukemogenesis. Blood. 96, 3154–3160.
Osato M., Asou N., Abdalla E., et al. 1999. Biallelic and heterozygous point mutations in the runt domain of the AML1/PEBP2B gene associated with myeloblastic leukemia. Blood. 93, 1817–1824.
Michaud J., Wu F., Osato M., et al. 2002. In vitro analyses of known and novel RUNX1/AML1 mutations in dominant familial platelet disorder with predisposition to acute myelogenous leukemia: implications for mechanisms of pathogenesis. Blood. 99, 1364–1372.
Cammenga J., Niebuhr B., Horn S., Bergholz U., Putz G., Buchholz F., Löhler J., Stocking C. 2007. RUNX1 DNA-binding mutants, associated with minimally differentiated acute myelogenous leukemia, disrupt myeloid differentiation. Cancer Res. 67, 537–545.
Wang Y.Y., Zhou G.B., Yin T., et al. 2005. AML1-ETO and C-KIT mutation/overexpression in t(8;21) leukemia: Implication in stepwise leukemogenesis and response to Gleevec. Proc. Natl. Acad. Sci. USA. 102, 1104–1109.
Peterson L.F., Boyapati A., Ahn E.Y., Biggs J.R., Okumura A.J., Lo M.C., Yan M., Zhang D.E. 2007. Acute myeloid leukemia with the 8q22;21q22 translocation: Secondary mutational events and alternative t(8;21) transcripts. Blood. 110, 799–805.
Bantounas I., Phylactou L.A., Uney J.B. 2004. RNA interference and the use of small interfering RNA to study gene function in mammalian systems. J. Mol. Endocrinol. 33, 545–557.
Vilgelm A.E., Chumakov S.P., Prassolov V.S. 2006. RNA interference: Biology and prospects of application in biomedicine and biotechnology. Mol. Biol. 40, 339–354.
Elbashir S.M., Harborth J., Lendeckel W., Yalcin A., Weber K., Tuschl T. 2001. Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature. 411, 494–498.
Caplen N.J., Parrish S., Imani F., Fire A., Morgan R.A. 2001. Specific inhibition of gene expression by small double-stranded RNAs in invertebrate and vertebrate systems. Proc. Natl. Acad. Sci. USA. 98, 9742–9747.
Fang Z.H., Dong C.L., Chen Z., Zhou B., Liu N., Lan H.F., Liang L., Liao W.B., Zhang L., Han Z.C. 2009. Transcriptional regulation of survivin by c-Myc in BCR/ABL-transformed cells: Implications in anti-leukaemic strategy. J. Cell Mol. Med. 13, 2039–2052.
Heidenreich O. 2009. Targeting oncogenes with siRNAs. Methods Mol. Biol. 487, 221–242.
Thomas M., Greil J., Heidenreich O. 2006. Targeting leukemic fusion proteins with small interfering RNAs: Recent advances and therapeutic potentials. Acta Pharmacol. Sin. Mar. 27, 273–281.
Nechipurenko O., Rossi L., Moore B. 2007. Comparison of approaches for rational siRNA design leading to a new efficient and transparent method. Nucleic Acids Res. 35, 1–10.
Volkov A.A., Kruglova N.S., Meschaninova M.I., Venyaminova A.G., Zenkova M.A., Vlassov V.V., Chernolovskaya E.L. 2009. Selective protection of nuclease-sensitive sites in siRNA prolongs silencing effect. Oligonucleotides. 19, 191–202.
Fagard M., Vaucheret H. 2000. Systemic silencing signals. Plant Mol. Biol. 43, 285–293.
Singer O., Verma I.M. 2008. Applications of lentiviral vectors for shRNA delivery and transgenesis. Curr. Gene. Ther. 8, 483–488.
Manjunath N., Wu H., Subramanya S., Shankar P. 2009. Lentiviral delivery of short hairpin RNAs. Adv. Drug Delivery Rev. 61, 732–745.
Schwieger M., Löhler J., Friel J., Scheller M., Horak I., Stocking C. 2002. AML1-ETO inhibits maturation of multiple lymphohematopoietic lineages and induces myeloblast transformation in synergy with ICSBP deficiency. J. Exp. Med. 196, 1227–1240.
Kravchenko J.E., Ilyinskaya G.V., Komarov P.G., Agapova L.S., Kochetkov D.V., Strom E., Frolova E.I., Kovriga I., Gudkov A.V., Feinstein E., Chumakov P.M. 2008. Small-molecule RETRA suppresses mutant p53-bearing cancer cells through a p73-dependent salvage pathway. Proc. Natl. Acad. Sci. USA. 105, 6302–6307.
Bergemann J., Kuhlcke K., Fehse B., Ratz I., Ostertag W., Lother H. 1995. Excision of specific DNA-sequences from integrated retroviral vectors via site-specific recombination. Nucleic Acids Res. 21, 4451–4456.
Spirin P.V., Baskaran D., Rubtsov P.M., Zenkova M.A., Vlasov V.V., Chernolovskaya E.L., Prassolov V. S. 2009. A comparison of target gene silencing using syntheti-cally modified siRNA and shRNA that express recombinant lentiviral vectors. Acta Naturae. 2, 86–90.
Takenokuchi M., Yashida C., Takeuchi K., Nakamachi Y., Mukai M., Kondo S., Kumagai S., Saigo K., Murayama T., Koizumi T., Tatsumi E. 2004. Quantitative nested reverse transcriptase PCR vs. real-time PCR for measuring AML1/ETO (MTG8) transcripts. Clin. Lab. Haem. 26, 107–114
Koldehoff M., Kordelas L., Beelen D.W., Elmaagacli A.H. 2010. Small interfering RNA against BCR-ABL transcripts sensitize mutated T315I cells to nilotinib. Haematologica. 95, 388–397.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © P.V. Spirin, D. Baskaran, N.N. Orlova, A.V. Rulina, N.A. Nikitenko, E.L. Chernolovskaya, M.A. Zenkova, V.V. Vlassov, P.M. Rubtsov, P.M. Chumakov, C. Stocking, V.S. Prassolov, 2010, published in Molekulyarnaya Biologiya, 2010, Vol. 44, No. 5, pp. 876–888.
Rights and permissions
About this article
Cite this article
Spirin, P.V., Baskaran, D., Orlova, N.N. et al. Downregulation of activated leukemic oncogenes AML1-ETO and RUNX1(K83N) expression with RNA-interference. Mol Biol 44, 776–786 (2010). https://doi.org/10.1134/S0026893310050146
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0026893310050146