Abstract
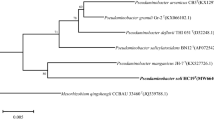
An isolate of prosthecate bacteria was isolated from the takyr soil of a rice field on the Akdala massif (Kazakhstan) and designated as strain 22T. Strain 22T was represented by gram-negative, aerobic, motile, oxidase and catalase-positive bacteria. Cells of this strain formed numerous prosthecae extending in different directions from the cell surface. Bacteria had a dimorphic life cycle, with two different morphological types of cells. The prosthecae may be short (i.e., less than 1.0 µm long) or long (i.e., more than 2.0 µm long). Cells reproduced by budding and grew within the pH range of 6.5–8.5 and at 28–30°C. Major cellular fatty acids were C18:1 ω7, C16:0, C19:1 branched, and C16:1 ω7. Phylogenetic analysis based on the 16S rRNA gene sequence showed that strain 22T shared the highest gene sequence similarity with Prosthecomicrobium hirschii 16T (99.9%) and formed a common branch on the phylogenomic tree with P. hirschii 16T and Siculibacillus lacustris SA-279T. This branch was only distantly related to the phylogenetic lineage defined by the type species of the genus Prosthecomicrobium, P. pneumaticum ATCC 23633T, thus indicating a polyphyletic nature of the genus Prosthecomicrobium. The average nucleotide identity (ANI) and in silico DNA–DNA hybridization (dDDH) values between strain 22T and the most closely related species P. hirschii 16T were 93.6 and 47.6%, respectively, and were below the threshold accepted for species demarcation. Based on the results of phylogenetic analysis, strains 22T and 16T should be assigned to the Ancalomicrobiaceae family, with introduction of a novel genus Prosthecodimorpha gen. nov. The G + C content of the genome of strain 22T was 61.9%. Strain 22T represented a novel species in a novel genus, for which the name Prosthecodimorpha staleyi gen. nov., sp. nov. is proposed, with the type strain 22T (=VKM B-3576T = UQM 41461T = DSMZ 113594T).



Similar content being viewed by others
REFERENCES
Atlas, R.M., Handbook of Microbiological Media, Taylor and Francis Group, Ed., LLC, 2010, p. 2043.
Auch, A.F., Jan, M., Klenk, H.P., and Göker, M., Digital DNA-DNA hybridization for microbial species delineation by means of genome-to-genome sequence comparison, Stand. Genomic. Sci. 2010, vol. 2, pp. 117–134. https://doi.org/10.4056/sigs.531120
Bankevich, A., Nurk, S., Antipov, D., Gurevich, A.A., Dvorkin, M., Kulikov, A.S., Lesin, V.M., Nikolenko, S.I., Pham, S., Prjibelski, A.D., Pyshkin, A.V., Sirotkin, A.V., Vyahhi, N., Tesler, G., Alekseyev, M.A., and Pevzner, P.A., SPAdes: a new genome assembly algorithm and its applications to single-cell sequencing, J. Comput. Bi-ol., 2012, vol. 19, pp. 455–477. https://doi.org/10.1089/cmb.2012.0021
Bolger, A.M., Lohse, M., and Usadel, B., Trimmomatic: a flexible trimmer for illumina sequence data, Bioinformatics, 2014, vol. 30, pp. 2114–2120. https://doi.org/10.1093/bioinformatics/btu170
Boone, D.R., Castenholz, R.W., and Garrity, G.M., Taxonomic outline of the Archaea and Bacteria, in Bergey’s Manual of Systematic Bacteriology: The Archaea and the Deeply Branching and Phototrophic Bacteria, Whit-man, W.B., Ed., N.Y.: Springer, 2001b, vol. 1, pp. 155–166.
Camacho, C., Coulouris, G., Avagyan, V., Ma, N., Papadopoulos, J., Bealer, K., and Madden, T.L., BLAST+: architecture and applications. BMC, Bioinformatics, 2009, vol. 10, p. 421. https://doi.org/10.3389/fgene.2021.822986
Chaumeil, P.A., Mussig, A.J., Hugenholtz, P., and Parks, D.H., GTDB-Tk: a toolkit to classify genomes with the Genome Taxonomy Database, Bioinformatics, 2020, vol. 36, pp. 1925–1927. https://doi.org/10.1093/bioinformatics/btz848
Chun, J., Oren, A., Ventosa, A., Christensen, H., Arahal, D.R., Costa, M.S., Rooney, A.P., Yi, H., Xu, X.-W., Meyer, S., and Trujillo, M.E., Proposed minimal standards for the use of genome data for the taxonomy of prokaryotes, Int. J. Syst. Evol. Microbiol., 2018, vol. 68, pp. 461–466. https://doi.org/10.1099/ijsem.0.002516
Dahal, R.H., Chaudhary, D.K., and Kim, J., Pinisolibacter ravus gen. nov., sp. nov., isolated from pine forest soil and allocation of the genera Ancalomicrobium and Pinisolibacter to the family Ancalomicrobiaceae fam. nov., and emendation of the genus Ancalomicrobium Staley, 1968, Int. J. Syst. Evo.l Microbiol., 2018, vol. 68, pp. 1955−1962. https://doi.org/10.1099/ijsem.0.002772
Edgar, R.C., Muscle: multiple sequence alignment with high accuracy and high throughput, Nucleic. Acids Res., 2004, vol. 32, pp. 1792−1797. https://doi.org/10.1093/nar/gkh340
Hirch, P., Bernhard, M., Cohen, S.S., Ensign, J.C., Jannasch, H.W., Koch, A.I., Marshall, K.C., Matin, A., Poindexter, J.S., Rittenberg, S.C., Smith, D.C., and Veldkamp, H., Life under conditions of low nutrient concentrations, in Strategies of Microbial Life in Extreme Environment, Shillo, M., Ed., Dahlem Konferenzen, 1979, pp. 357−372.
Hoang, D.T., Chernomor, O., Von Haeseler, A., Minh, B.Q., and Vinh, L.S., UFBoot2: improving the ultrafast bootstrap approximation, Mol. Biol. Evol., 2017, vol. 35, pp. 518–522. https://doi.org/10.1093/molbev/msx281
Hoang, D.T., Vinh, L.S., Flouri, T., Stamatakis, A., von Haeseler, A., and Minh, B.Q., MPBoot: fast phylogenetic maximum parsimony tree inference and bootstrap approximation, BMC Evol. Biol., 2018, vol. 18, p. 11. https://doi.org/10.1186/s12862-018-1131-3
Hordt, A., Lopez, M.G., Meier-Kolthoff, J.P., Schleuning, M., Weinhold, L.-M., Tindall, B.J., Gronow, S., Kyrpides, N.C., Woyke, T., and Goker, M., Analysis of 1,000+ type-strain genomes substantially improves taxonomic classification of Alphaproteobacteria, Front. Microbiol., 2020, vol. 11, p. 1−112. https://doi.org/10.3389/fmicb.2020.00468
Jain, C., Rodriguez, R.L.-M., Phillippy, A.M., Konstantinidis, K.T., and Aluru, S., High throughput ANI analysis of 90K prokaryotic genomes reveals clear species boundaries, Nat. Commun., 2018, vol. 9, p. 5114. https://doi.org/10.1038/s41467-018-07641-9
Kalyaanamoorthy, S., Minh, B.Q., Wong, T.K.F., Haeseler, A., and Jermiin, L.S., Model Finder: fast model selection for accurate phylogenetic estimates, Nat. Methods., 2017, vol. 14, pp. 587–589. https://doi.org/10.1038/nmeth.4285
Kumar, S., Stecher, G., and Tamura, K., MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets, Mol. Biol. Evol., 2016, vol. 33, pp. 1870–1874. https://doi.org/10.1093/molbev/msw054
Lane, D.J., 16S/23S rRNA sequencing in nucleic acid techniques in bacterial systematics, in Nucleic Acid Techniques in Bacterial Systematics, Stackebrandt, E. and Goodfel-low, M., Eds., Chichester: Wiley, 1991, pp. 115−175.
Lee, K.-B., Liu, C.-T., Anzai, Y., Kim, H., Aono, T., and Oyaizu, H., The hierarchical system of the ‘Alphaproteobacteria’: description of Hyphomonadaceae fam. nov., Xanthobacteraceae fam. nov. and Erythrobacteraceae fam. nov., Int. J. Syst. Evol. Microbiol., 2005, vol. 55, pp. 1907–1919. https://doi.org/10.1099/ijs.0.63663-0
Minnikin, D.E., O’Donnell, A.G., Goodfellow, M., Alderson, G., Athalye, M.A., Alderson, G., Athalye, M., Schaal, A., and Parlett, J.H., An integrated procedure for the extraction of bacterial isoprenoid quinones and polar lipids, J. Microbiol. Methods, 1984, vol. 2, pp. 233−241.
Nguyen, L.-T., Schmidt, H.A., Haeseler, A., and Minh, B.Q., IQ-TREE: a fast and effective stochastic algorithm for estimating maximumlikelihood phylogenies, Mol. Biol. Evol., 2015, vol. 32, pp. 268–274. https://doi.org/10.1093/molbev/msu300
Park, S., Jung, Y.T., Kim, S., and Yoon, J.H., Devosia confluentis sp. nov., isolated from the junction between the ocean and a freshwater lake, and reclassification of two Vasilyevaea species as Devosia enhydra comb. nov. and Devosia mishustinii comb. nov., Int. J. Syst. Evol. Microbiol., 2016, vol. 66, pp. 3935−3941. https://doi.org/10.1099/ijsem.0.001291
Poindexter, J.S., Oligotrophy. Fast and famine existence, Advans. Microbiol. Ecol., 1981, vol. 5, pp. 63‒89.
Schlesner, H., Kath, T., Fischer, A., and Stackebrandt, E., Studies on the phylogenetic position of Prosthecomicrobium pneumaticum, Prosthecomicrobium enhydrum, Ancalomicrobium adetum, and various Prosthecomicrobium-like bacteria, Syst. Appl. Microbiol., 1989, vol. 12, pp. 150−155. https://doi.org/10.1016/S0723-2020(89)80006-2
Semenov, A.M. and Vasilyeva, L.V., Morphological and physiological characteristics of the oligotrophic prosthecobacterium Prosthecomicrobium hirschii grown under conditions of batch and continuous cultivation, Mikrobiologiya, 1986, vol. 55, pp., 248−252.
Slobodkina, G.B., Merkel, A.Y., Novikov, A.A., Bonch-Osmolovskaya, E.A., and Slobodkin, A.I., Pelomicrobium methylotrophicum gen. nov., sp. nov. a moderately thermophilic, facultatively anaerobic, lithoautotrophic and methylotrophic bacterium isolated from a terrestrial mud volcano, Extremophiles, 2020, vol. 24, pp. 177−185. https://doi.org/10.1007/s00792-019-01145-0
Staley, J.T., Prosthecomicrobium and Ancalomicrobium: new prosthecate freshwater bacteria, J. Bacteriol., 1968, vol. 95, pp. 1921−1942. https://doi.org/10.1128/JB.95.5.1921-1942.1968
Staley, J.T., Prosthecomicrobium hirschii, a new species in a redefined genus, Int. J. Syst. Bacteriol., 1984, vol. 34, pp. 304−308. https://doi.org/10.1099/00207713-34-3-304
Tatusova, T., DiCuccio M., Badretdin, A., Chetvernin, V., Nawrocki, E.P., Zaslavsky, L., Lomsadze, A., Kim, D., Pruitt, K.D., Borodovsky, M., and Ostell, J., NCBI prokaryotic genome annotation pipeline, Nucleic. Acids Res. 2016, vol. 44, pp. 6614–6624. https://doi.org/10.1093/nar/gkw569
Vasilyeva, L.V., Oligotrofic microorganisms as components of biogeocoenosis, in Soil Microorganisms as Components of Biogeocoenosis, Mishustin, E.N., Ed., Moscow, Nauka, 1984, pp. 232‒241.
Vasilyeva, L.V., Prostecobactheria in soils of rice fields, in Increase in Efficiency of Soils of Rice Fields, Mishustin, E.N., Ed., Moscow, Science, 1985, pp. 125‒132.
Vasil’eva, L.V., Semenov, A.M., and Giniyatullina, A.I., A new species of soil bacteria of the genus Prosthecomicrobium, Microbiology, 1991, vol. 60, no. 2, pp. 243‒250.
Vasilyeva, L.V and Zavarzin, G.A., Dissipotrophs in the microbial community, Microbiologiya, 1995, vol. 64, no.2, pp. 239‒244.
Yee, B., Oertli, G.E., Fuerst, J.A., and Staley, J.T., Reclassification of the polyphyletic genus Prosthecomicrobium to form two novel genera, Vasilyevaea gen. nov. and Bauldia gen. nov. with four new combinations: Vasilyevaea enhydra comb. nov., Vasilyevaea mishustinii comb. nov., Bauldia consociata comb. nov. and Bauldia litoralis comb. nov., Int. J. Syst. Evol. Microbiol., 2010, vol. 60, pp. 2960−2966. https://doi.org/10.1099/ijs.0.018234-0
ACKNOWLEDGMENTS
Bioinformatic analysis was performed using computing resources at the SciBear OU (https://sci-bear.com/).
Funding
The work was supported by the Ministry of Science and Higher Education of the Russian Federation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest. This article does not contain any studies involving animals or human participants performed by any of the authors.
Rights and permissions
About this article
Cite this article
Vasilyeva, L., Grouzdev, D., Koziaeva, V. et al. Prosthecodimorpha staleyi gen. nov., sp. nov., Novel Prosthecate Bacteria within the Family Ancalomicrobiaceae and Reclassification of the Polyphyletic Genus Prosthecomicrobium. Microbiology 91, 479–488 (2022). https://doi.org/10.1134/S0026261722601105
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0026261722601105