Abstract
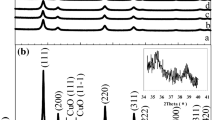
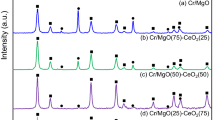
The effect of the microstructure of titanium dioxide on the structure, thermal stability, and catalytic properties of supported CuO/TiO2 and CuO/(CeO2-TiO2) catalysts in CO oxidation was studied. The formation of a nanocrystalline structure was found in the CuO/TiO2 catalysts calcined at 500°C. This nanocrystalline structure consisted of aggregated fine anatase particles about 10 nm in size and interblock boundaries between them, in which Cu2+ ions were stabilized. Heat treatment of this catalyst at 700°C led to a change in its microstructure with the formation of fine CuO particles 2.5–3 nm in size, which were strongly bound to the surface of TiO2 (anatase) with a regular well-ordered crystal structure. In the CuO/(CeO2-TiO2) catalysts, the nanocrystalline structure of anatase was thermally more stable than in the CuO/TiO2 catalyst, and it persisted up to 700°C. The study of the catalytic properties of the resulting catalysts showed that the CuO/(CeO2-TiO2) catalysts with the nanocrystalline structure of anatase were characterized by the high-est activity in CO oxidation to CO2.
Similar content being viewed by others
References
Centi, G., Nigro, C., Perathoner, S., and Stella, G., Catal. Today, 1993, vol. 17, p. 159.
Centi, G., Perathoner, S., Kartheuser, B., Hodnett, B.K., and Stella, G., Catal. Today, 1993, vol. 17, p. 103.
Centi, G., Perathoner, S., Kartheuser, B., Rohan, D., and Hodnett, B.K., Appl. Catal., B, 1992, vol. 1, p. 129.
Centri, G. and Perathoner, S., Appl. Catal., A, 1995, vol. 132, p. 179.
Komova, O.V., Simakov, A.V., Tzykoza, L.T., Sasonova, N.N., Ushakov, A.V., Barannik, G.B., and Ismagilov, Z.R., React. Kinet. Catal. Lett., 1995, vol. 54, p. 361.
Schoenmarker-Stolk, M.C., Verwijs, J.M., and Scholten, J.F., Appl. Catal., A, 1987, vol. 30, p. 339.
Taylor, K.C., Catal. Rev. Sci. Eng., 1993, vol. 35, p. 457.
Larsson, P.O. and Andersson, A., J. Catal., 1998, vol. 179, p. 72.
Larsson, P.O., Andersson, A., Wallenberg, B., and Svensson, B., J. Catal., 1996, vol. 163, p. 279.
Guo, X.-Z., Huang, J., Wang, S.-R., Wang, Y.-M., Zang, B.-L., and Wu, S.-H., J. Dispersion Sci. Technol., 2009, vol. 30, p. 1114.
Huang, J., Wang, Sh., Zhao, Y., Wang, X., Wang, Sh., Wu, Sh., Zhang, Sh., and Huang, W., Catal. Commun., 2006, no. 7, p. 1029.
Tsodikov, M.V., Trusova, Ye.A., Slivinskii, Ye.V., Hernandez, G.G., Kochubey, D.I., Lipovich, V.G., and Navio, J.A., Stud. Surf. Sci. Catal., 1998, vol. 127, p. 679.
Trusova, E.A., Tsodikov, M.V., Slivinskii, E.V., Hernandez, G.G., Bukhtenko, O.V., Zhdanova, T.N., Kochubey, D.I., and Navio, J.A., Mendeleev Commun., 1998, no. 3, p. 102.
Chen, C.-S., You, J.-H., Lin, J.-H., and Chen, Y.Y., Catal. Commun., 2008, vol. 9, p. 2381.
Arana, J., Rodriguez, C.F., Diaz, O.G., Melian, J.A.H., and Pena, J.P., Catal. Today, 2005, vol. 101, p. 261.
Chiang, K., Amal, R., and Tran, T., Adv. Environ. Res., 2002, vol. 6, p. 471.
Komova, O.V., Simakov, A.V., Rogov, V.A., Kochubei, D.I., Odegova, G.V., Kriventsov, V.V., Paukshtis, E.A., Ushakov, V.A., Sasonova, N.N., and Nikoro, T.A., J. Mol. Catal. A: Chem., 2000, vol. 161, nos. 1–2, p. 191.
Boccuzzi, F., Chiorino, A., Martra, G., Gargano, M., Ravasio, N., and Carrozzini, B., J. Catal., 1997, vol. 165, no. 2, p. 129.
Boccuzzi, F., Chiorino, A., Martra, G., Gargano, M., Ravasio, N., and Carrozzini, B., J. Catal., 1997, vol. 165, no. 2, p. 140.
Yu, X.-F., Wu, N.-Z., Xie, Y.-C., and Tang, Y.-Q., J. Mater. Chem., 2000, vol. 10, p. 1629.
Zhu, H., Wu, Y., Zhao, X., Wan, H., Yang, L., Hong, J., Yu, Q., Dong, L., Chen, Y., Jian, C., Wei, J., and Xu, P., J. Mol. Catal. A: Chem., 2006, vol. 243, no. 1, p. 24.
Boccuzzi, F., Coluccia, S., Martra, G., and Ravasio, N., J. Catal., 1999, vol. 184, no. 2, p. 316.
Zenkovets, G.A., Shutilov, A.A., Gavrilov, V.Yu., Tsybulya, S.V., and Kryukova, G.N., Kinet. Catal., 2007, vol. 48, no. 5, p. 742.
Shutilov, A.A., Zenkovets, G.A., Gavrilov, V.Yu., and Tsybulya, S.V., Kinet. Catal., 2011, vol. 52, no. 1, p. 111.
Zenkovets, G.A., Gavrilov, V.Yu., Shutilov, A.A., and Tsybulya, S.V., Kinet. Catal., 2009, vol. 50, no. 5, p. 760.
Guinier, A., Theorie et technique de la radiocristallographie, Paris: Dunot, 1956.
Sherwood, P.M.A., in Practical Surface Analysis by Auger and X-ray Photoelectron Spectroscopy, Briggs, D. and Seah, M.P., Eds., Chichester: Wiley, 1983, p. 555.
Barret, E.P., Joyner, L.G., and Hallenda, P.H., J. Am. Chem. Soc., 1951, vol. 73, no. 1, p. 373.
Leonov, A.I., Vysokotemperaturnaya khimiya kislorodnye soedinenii tseriya (High-Temperature Chemistry of Oxygen-Containing Cerium Compounds), Leningrad: Nauka, 1970.
Leonov, A.I., Piryutko, M.M., and Keller, E.K., Izv. Akad. Nauk SSSR, 1966, no. 5, p. 787.
Sinha, A.K. and Suzuki, K., J. Phys. Chem. B, 2001, vol. 109, no. 5, p. 1708.
Handbook of X-Ray Photoelectron Spectroscopy, Moulder, J.F., Stickle, W.F., Sobol, P.E., et al., Eds., Eden Prairie, Minn.: PerkinElmer Corp., Physical electronics Division, 1992.
Wagner, C.D., Naumkin, A.V., Kraut-Vass, A., Allison, J.W., Powell, C.J., and Rumble, C.J., NIST X-Ray Photoelectron Spectroscopy Database, Version 3.5, National Institute of Standards and Technology, Gaithersburg, 2003. http://srdata.nist.gov/xps
Mason, M.G., Phys. Rev., 1983, vol. 27, p. 748.
Zang, J., Li, M.-J., Feng, Z.-C., Chen, J., and Li, C., J. Phys. Chem. B, 2006, vol. 110, p. 927.
Zou, Zh.-Q., Meng, M., Guo, L.-H., and Zha, Yu.-Q., J. Hazard. Mater., 2009, vol. 163, p. 835.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.A. Shutilov, G.A. Zenkovets, S.V. Tsybulya, V.Yu. Gavrilov, G.N. Kryukova, 2012, published in Kinetika i Kataliz, 2012, Vol. 53, No. 3, pp. 424–434.
Rights and permissions
About this article
Cite this article
Shutilov, A.A., Zenkovets, G.A., Tsybulya, S.V. et al. Effect of the microstructure of the supported catalysts CuO/TiO2 and CuO/(CeO2-TiO2) on their catalytic properties in carbon monoxide oxidation. Kinet Catal 53, 409–418 (2012). https://doi.org/10.1134/S0023158412030111
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0023158412030111