Abstract
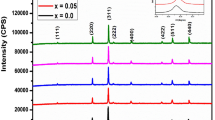
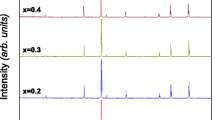
We have studied the cation distribution over the tetrahedral and octahedral sites in the spinel structure of nanocrystalline Ni x Zn1 − x Fe2O4 ferrites prepared by spray pyrolysis. 57Fe Mössbauer spectroscopy data for the ferrites demonstrate that, depending on the composition of the materials, the tetrahedral site may accommodate only Fe3+ (inverse spinel, x ≥ 0.4) or both Fe3+ and Zn2+ cations (mixed spinel, x = 0 and 0.2), which accounts for the fact that the composition dependence of the unit-cell parameter for the ferrites deviates from Vegard’s law.
Similar content being viewed by others
References
Gubin, S.P., Koksharov, Yu.A., Khomutov, G.B., and Yurkov, G.Yu., Magnetic Nanoparticles: Preparation, Structure, and Properties, Usp. Khim., 2005, vol. 74, no. 6, pp. 539–574.
Wei, Q.-M., Li, J.-B., Chen, Y.-J., and Han, Y.-S, Cation Distribution in NixMn1 − x Fe2O4 Ferrites, Mater. Chem. Phys., 2002, vol. 74, pp. 340–343.
Manova, E., Kunev, B., Paneva, D., et al., Mechanochemical Synthesis and Magnetic Properties of Nano-Dimensional Cobalt Ferrite, Chem. Mater., 2004, vol. 16, pp. 5689–5696.
Morrison, S.A., Gahill, C.L., Carpenter, E.E., et al., Magnetic and Structural Properties of Nickel Zinc Oxide Nanoparticles Synthesized at Room Temperature, J. Appl. Phys., 2004, vol. 95, pp. 6392–6395.
Jacob, J. and Abdul Khadar, M., Investigation of Mixed Spinel Structure of Nanostructured Nickel Ferrite, J. Appl. Phys., 2010, vol. 107, paper 114 310.
Manova, E., Estournès, C., Paneva, D., et al., Mossbauer Study of Nanodimensional Nickel Ferrite-Mechanochemical Synthesis and Catalytic Properties, Hyperfine Interact., 2005, vol. 165, pp. 215–220.
Lee, P.Y., Ishizaka, K., Suematsu, H., et al., Magnetic and Gas Sensing Property of Nanosized NiFe2O4 Powders Synthesized by Pulsed Wire Discharge, J. Nanoparticle Res., 2006, vol. 8, pp. 29–35.
Kazin, A.P., Rumyantseva, M.N., Prusakov, V.E., et al., Microstructure and Gas-Sensing Properties of Nanocrystalline NiFe2O4 Prepared by Spray Pyrolysis, Inorg. Mater., 2010, vol. 46, no. 11, pp. 1254–1259.
Tucic, A., Brankovic, G., Marinkovic, Z., and Mancic, L., Synthesis of Thin Films by the Pyrosol Process, Chem. Ind., 2002, vol. 56, no. 9, pp. 375–380.
Shannon, R.D., Revised Effective Ionic Radii and Systematic Studies of Interatomic Distances in Halides and Chalcogenides, Acta Crystallogr., Sect. A: Cryst. Phys., Diffr., Theor. Gen. Crystallogr., 1976, vol. 32, no. 5, pp. 751–767.
Greenwood, N.N. and Webb, T.G., Mössbauer Spectroscopy, London: Chapman and Hall, 1971.
Schiessl, W., Potzel, W., Karzel, H., et al., Magnetic Properties of the ZnFe2O4 Spinel, Phys. Rev. B: Condens. Matter Mater. Phys., 1996, vol. 53, no. 14, pp. 9143–9152.
Suzdalev, I.P., Buravtsev, V.N., Imshennik, V.K., et al., Magnetic Properties of Ultrafine Ferrihydrite Clusters Studied by Mössbauer Spectroscopy and Thermodynamic Analysis, Z. Phys. D, 1996, vol. 37, pp. 55–61.
Ma, Y.G., Jin, M.Z., Liu, M.L., et al., Effect of High Pressure on Mössbauer Spectra of NiFe2O4 Ultrafine Particles with Different Grain Sizes, Mater. Chem. Phys., 2000, vol. 65, pp. 79–84.
Leung, K., Evans, B.J., and Morrish, A.H., Low-Temperature Mössbauer Study of a Nickel-Zinc Ferrite ZnxNi1 − x Fe2O4, Phys. Rev. B: Solid State, 1973, vol. 8, pp. 29–43.
Yangkyu Aiin, Eun Jung Choi, Sehum Kim, et al., Magnetization and Mössbauer Study of Nanosize ZnFe2O4 Particles Synthesized by Using a Microemulsion Method, J. Korean Phys. Soc., 2002, vol. 41, pp. 123–128.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.P. Kazin, M.N. Rumyantseva, V.E. Prusakov, I.P. Suzdalev, A.M. Gaskov, 2012, published in Neorganicheskie Materialy, 2012, Vol. 48, No. 5, pp. 611–616.
Rights and permissions
About this article
Cite this article
Kazin, A.P., Rumyantseva, M.N., Prusakov, V.E. et al. Cation distribution in nanocrystalline Ni x Zn1 − x Fe2O4 spinel ferrites. Inorg Mater 48, 525–530 (2012). https://doi.org/10.1134/S002016851205007X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S002016851205007X