Abstract
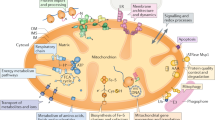
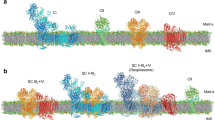
Topogenesis of mitochondrial proteins includes their synthesis in cytosol and mitochondria, their translocation across the outer and inner membranes, sorting to various mitochondrial compartments, and assembly of different protein complexes. These complexes are involved in transport functions, electron transfer through the respiratory chain, generation of transmembrane electrochemical potential, oxidative phosphorylation of ADP into ATP, etc. To perform these functions, a special stringent control is required over formation of submitochondrial structures and the mitochondrion as a whole. Such control is expected to rigorously eliminate not only misfolded proteins but also incorrectly incorporated subunits and is realized in mitochondria by means of numerous proteases with different functions and localizations. In the case of more complicated protein formations, e.g. supercomplexes, the protein quality is assessed by their ability to realize the integral function of the respiratory chain and, thus, ensure the stability of the whole system. Considering supercomplexes of the mitochondrial respiratory chain, the present review clearly demonstrates that this control is realized by means of various (mainly vacuolar) proteases with different functions and localizations. The contemporary experimental data also confirm the author’s original idea that the general mechanism of assembly of subcellular structures is based on the “selection by performance criterion” and “stabilization by functioning”.
Similar content being viewed by others
References
Zorov, D. B., Isaev, N. K., Plotnikov, E. Yu., Zorova, L. D., Stelmashook, E. V., Vasileva, A. K., Arkhangelskaya, A. A., and Khrjapenkova, T. G. (2007) Biochemistry (Moscow), 72, 1115–1126.
Suen, D. G., Norris, K. L., and Youle, R. J. (2008) Genes Dev., 22, 1577–1590.
Neupert, W., and Herrmann, J. M. (2007) Annu. Rev. Biochem., 76, 723–749.
Boekema, E. J., and Braun, H.-P. (2007) J. Biol. Chem., 282, 1–4.
Luzikov, V. N. (1980) Regulation of Mitochondria Formation [in Russian], Nauka, Moscow.
Shwerzmann, K., Cruz-Orive, L. M., Eggman, R., Sanger, A., and Weibel, E. R. (1986) J. Cell Biol., 102, 97–103.
Wallace, D. C. (2005) Ann. Rev. Genet., 39, 359–407.
Poyton, R. O., and McEwen, J. E. (1996) Annu. Rev. Biochem., 65, 563–607.
Fontanesi, F., Soto, I. C., and Barrientos, A. (2008) IUBMB Life, 60, 557–568.
Tsukihara, T., Aoyama, H., Yamashita, E., Tomizaki, T., Yamaguchi, H., Shinzawa-Itoh, K., Nakashima, R., Yaono, R., and Yoshikawa, S. (1996) Science, 272, 1136–1144.
Capaldi, R. A., Malatesta, F., and Darley-Usmar, V. M. (1983) Biochim. Biophys. Acta, 726, 135–148.
Church, C., Goehring, B., Forsha, D., Wazny, P., and Poyton, R. O. (2005) J. Biol. Chem., 280, 1854–1863.
Zara, V., Palmisano, I., Conte, L., and Trumpower, B. L. (2004) Eur. J. Biochem., 271, 1209–1218.
Cruciat, C.-M., Hell, K., Folsch, H., Neupert, W., and Stuart, R. A. (1999) EMBO J., 18, 5226–5233.
Sazanov, L. A., Peak-Chew, S. Y., Fearnley, I. M., and Walker, J. E. (2000) Biochemistry, 39, 7229–7235.
Grivennikova, V. G., and Vinogradov, A. D. (2003) Usp. Biol. Khim., 43, 19–58.
Duarte, M., Sousa, R., and Videira, A. (1995) Genetics, 139, 1211–1221.
Boyer, P. D. (1997) Annu. Rev. Biochem., 66, 717–749.
Weber, J. (2007) Trends Biochem. Sci., 32, 53–56.
Lau, W. C., Baker, L. A., and Rubinstein, J. L. (2008) J. Mol. Biol., 382, 1256–1264.
Devenish, R. J., Prescott, M., and Rodgers, A. J. (2008) Int. Rev. Cell Mol. Biol., 267, 1–58.
Tzagoloff, A., Barrientos, A., Neupert, W., and Herrmann, J. M. (2004) J. Biol. Chem., 279, 19775–19780.
Wang, Z. G., White, P. S., and Ackerman, S. H. (2001) J. Biol. Chem., 276, 30773–30778.
Green, D. E., and Hechter, O. (1965) Proc. Natl. Acad. Sci. USA, 53, 318–325.
Schagger, H., and Pfeiffer, K. (2000) EMBO J., 19, 1777–1783.
Cruciat, C.-M., Brunner, S., Baumann, F., Neupert, W., and Stuart, R. A. (2000) J. Biol. Chem., 275, 18093–18098.
Schafer, E., Seelert, H., Reifschneider, N. H., Krause, F., Dencher, N. A., and Vonck, J. (2006) J. Biol. Chem., 281, 15370–15375.
Schagger, H. (2001) IUBMB Life, 52, 119–128.
Luzikov, V. N. (1973) Subcell. Biochem., 2, 1–31.
Ko, Y. H., Delannoy, M., Hullihen, J., Chiu, W., and Pedersen, P. L. (2003) J. Biol. Chem., 278, 12305–12309.
Chen, C., Ko, Y., Delannoy, M., Ludtke, S. J., Chiu, W., and Pedersen, P. L. (2004) J. Biol. Chem., 279, 31761–31768.
Shutt, T. E., and Gray, M. W. (2006) Trends Gen., 22, 90–95.
Ryan, M. T., and Hoogenraad, N. J. (2007) Annu. Rev. Biochem., 76, 701–722.
Scarpulla, R. C. (2008) Physiol. Rev., 88, 611–638.
Wu, Z., Puigserver, P., Andersson, U., Zhang, G., Adelmant, G., Mootha, V., Troy, A., Cinti, S., Lowell, B., Scarpulla, R. C., and Spiegelman, B. M. (1999) Cell, 98, 115–124.
Arny, Z., Wagner, B. K., Ma, Y., Chinsomboom, J., Laznik, D., and Spiegelman, B. M. (2008) Proc. Natl. Acad. Sci. USA, 105, 4721–4725.
Kutik, S., Guiarad, B., Meyer, H., Wiedemann, N., and Pfanner, N. (2007) J. Biol. Chem., 179, 585–591.
Neupert, W. (1997) Annu. Rev. Biochem., 66, 863–917.
Van Dyck, L., and Langer, T. (1999) Cell Mol. Life Sci., 56, 825–842.
Young, L., Leonhard, K., Tatsuta, T., Trowsdale, J., and Langer, T. (2001) Science, 291, 2135–2138.
Steglich, G., Neupert, W., and Langer, T. (1999) Mol. Cell. Biol., 19, 3435–3442.
Langer, T. (2000) Trends Biochem. Sci., 25, 247–251.
Kaser, M., Kambacheld, M., Kisters-Woike, B., and Langer, T. (2003) J. Biol. Chem., 278, 46414–46423.
Kambacheld, M., Augustin, S., Tatsuta, T., Muller, S., and Langer, T. (2005) J. Biol. Chem., 280, 20132–20139.
Wheeldon, L. W., Dianoux, A. C., Bof, M., and Vignais, P. V. (1974) Eur. J. Biochem., 46, 189–199.
Kalnov, S. L., Novikova, L. A., Zubatov, A. S., and Luzikov, V. N. (1979) Biochem. J., 182, 195–202.
Kalnov, S. L., Novikova, L. A., Zubatov, A. S., and Luzikov, V. N. (1979) FEBS Lett., 101, 355–358.
Suzuki, C. K., Rep, M., van Dijl, J. M., Suda, K., Grivell, L. A., and Schatz, G. (1997) Trends Biochem. Sci., 22, 118–123.
Guelin, E., Rep, M., and Grivell, L. A. (1996) FEBS Lett., 381, 42–46.
Savel’ev, A. S., Novikova, L. A., Kovaleva, I. E., Luzikov, V. N., Neupert, W., and Langer, T. (1998) J. Biol. Chem., 273, 20596–20602.
Weber, E. R., Hanekamp, T., and Thorsness, P. E. (1996) Mol. Biol. Cell., 7, 307–317.
Pearce, D. A., and Sherman, F. (1997) J. Biol. Chem., 272, 31829–31836.
Wagner, I., Arlt, H., van Dyck, L., Langer, T., and Neupert, W. (1994) EMBO J., 13, 5135–5145.
Kovaleva, I. E., Novikova, L. A., Nazarov, P. A., Grivennikov, S. I., and Luzikov, V. N. (2003) Eur. J. Biochem., 270, 222–229.
Stuart, R. A., Cyr, D. M., Craig, E. A., and Neupert, W. (1994) Trends Biochem. Sci., 19, 87–92.
Rottgers, K., Zufall, N., Guiard, B., and Voos, W. (2002) J. Biol. Chem., 277, 45829–45837.
Von Janovsky, B., Major, T., Knapp, K., and Koos, W. (2006) J. Mol. Biol., 31, 799–807.
Arlt, H., Tauer, R., Feldmann, H., Neupert, W., and Langer, T. (1996) Cell, 85, 875–885.
Leonhard, K., Sriegler, A., Neupert, W., and Langer, T. (1999) Nature, 398, 348–351.
Graef, M., Seewald, G., and Langer, T. (2007) Mol. Cell. Biol., 27, 2476–2485.
Arlt, H., Steglich, G., Perryman, R., Guiard, B., Neupert, W., and Langer, T. (1998) EMBO J., 17, 4837–4847.
Augustin, S., Nolden, M., Muller, S., Hardt, O., Arnold, I., and Langer, T. (2005) J. Biol. Chem., 280, 2691–2699.
Arnold, I., Wagner-Ecker, M., Ansorge, W., and Langer, T. (2006) Gene, 367, 74–88.
Dubaquie, Y., Looser, R., Funfschilling, U., Jeno, P., and Rospert, S. (1998) EMBO J., 17, 5868–5876.
Voisine, C., Schilke, B., Ohlson, M., Beinert, H., Marszalek, J., and Craig, E. A. (2000) Mol. Cell. Biol., 20, 3677–3684.
Schmidt, S., Strub, A., Rottgers, K., Zufall, N., and Voos, W. (2001) J. Mol. Biol., 313, 13–26.
Rep, M., van Dijl, J. M., Suda, K., Schatz, G., Grivell, L. A., and Suzuki, C. K. (1996) Science, 274, 103–106.
Rep, M., Nooy, J., Guelin, E., and Grivell, L. A. (1996) Curr. Genet., 30, 206–211.
Galkin, A. V., Tsoi, T. V., and Luzikov, V. N. (1980) Biochem. J., 190, 145–156.
Luzikov, V. N., Zubatov, A. S., and Rainina, E. I. (1973) J. Bioenerg. Biomembr., 5, 129–149.
Parry, G. (1978) Subcell. Biochem., 5, 261–325.
Baba, M., Takeshige, K., Baba, N., and Ohsumi, Y. (1994) J. Cell Biol., 124, 903–913.
Ziegler, D. M., Linnane, A. W., Green, D. E., Dass, C. M., and Ris, H. (1958) Biochim. Biophys. Acta, 28, 524–538.
Luzikov, V. N. (1985) Mitochondrial Biogenesis and Breakdown, Plenum Publisher Corporation, New York-London.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V. N. Luzikov, 2009, published in Uspekhi Biologicheskoi Khimii, 2009, Vol. 49, pp. 77–106.
The Editorial Board is deeply grateful to Academician A. A. Bogdanov for editing this review.
Rights and permissions
About this article
Cite this article
Luzikov, V.N. Principles of control over formation of structures responsible for respiratory functions of mitochondria. Biochemistry Moscow 74, 1443–1456 (2009). https://doi.org/10.1134/S0006297909130021
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006297909130021