-
PDF
- Split View
-
Views
-
Cite
Cite
Chia-Tai Shiu, Tse-Min Lee, Ultraviolet-B-induced oxidative stress and responses of the ascorbate–glutathione cycle in a marine macroalga Ulva fasciata, Journal of Experimental Botany, Volume 56, Issue 421, November 2005, Pages 2851–2865, https://doi.org/10.1093/jxb/eri277
Close - Share Icon Share
Abstract
The regulation of the antioxidant defence system by ultraviolet-B (UV-B) was determined in a marine macroalga Ulva fasciata Delile exposed to low (0.5, 1 W m−2), medium (2.5, 5 W m−2), and high (10, 20 W m−2) UV-B irradiance. UV-B ≥2.5 W m−2 increased H2O2 contents that are positively correlated with lipid peroxidation and total peroxide contents. Inhibition of the UV-B-induced H2O2 increase by a specific
Introduction
The stratospheric ozone layer is vital to life on Earth because it is the principal agent absorbing the ultraviolet radiation in the Earth's atmosphere. Since 1990, the depletion of the stratosphere ozone layer due to anthropogenic and natural destruction is leading to increasing levels of solar ultraviolet-B (UV-B: 280–320 nm) radiation reaching the Earth's surface, especially in Arctic, Antarctic, and temperate regions (Seckmeyer and McKenzie, 1992; Kerr and McElroy, 1993; Russell et al., 1996), where UV-B and UV-A (315–400 nm) are capable of penetrating the water column to an ecologically significant depth (Calkins and Thordardottir, 1982; Smith et al., 1992). Ambient UV-B irradiance at low latitudes is also high due to the high solar angle and a relatively low stratospheric ozone amount; the current UV-B flux at low latitudes is higher than any likely UV-B flux at higher latitudes even with further depletion of stratospheric ozone. High ultraviolet radiation can penetrate more deeply into the oligotrophic tropical water column because of the relatively low amount of dissolved and particulate organic matter (Baker et al., 1980). It would be expected that tropical seaweeds potentially encounter higher UV-B radiation than those occurring in temperate and Polar regions even under the normal ozone layer (Häder, 1993).
Exposure to UV-B leads to the generation of reactive oxygen species (ROS). In general, oxidative stress results from the disruption of cellular homeostasis of ROS production from the excitation of O2 to form singlet oxygen
The over-production of ROS and the induction of oxidative stress by UV-B radiation have been observed in microalgae such as Chlorella vulgaris (Malanga et al., 1997), cyanobacteria (Foyer et al., 1994; He et al., 2002), a diatom (Rijstenbil, 2002), Euglena gracilis (Tschiersch and Ohmann, 1993), and a symbiotic dinoflagellate (Lesser, 1996). An increase in the activities of ROS scavenging enzymes (such as SOD, APX, and CAT), and in the concentrations of antioxidants detoxifying accumulated ROS, was observed in algae exposed to oxidative stress (Butow et al., 1994; Malanga and Puntarulo, 1995; Malanga et al., 1997; Collén and Davison, 1999b; Aguilera et al., 2002; Rijstenbil, 2002). It is known that the antioxidant defence mechanism against ROS is pivotal for algal survival under stressful conditions; higher antioxidant contents and antioxidant enzyme activities are associated with higher stress tolerance in algae (Butow et al., 1994; Collén and Davison, 1999a, b). The comparison of antioxidant enzyme activity among Arctic marine macroalgae to UV radiation showed that algal tolerance to oxidative stress was correlated with high SOD, APX, and CAT activities (Aguilera et al., 2002).
As far as is known, the mechanisms in the regulation of the antioxidant defence system in marine macroalgae under UV-B stress are still under debate. This study has been conducted to examine the regulation of the antioxidant defence system by UV-B in the tropical chlorophyte Ulva fasciata Delile by exposure of thallus segments (1 cm in length) to varying UV-B doses. The growth rate and the reduction of 2,3,5-triphenyltetrazolium chloride (TTC) as cell viability (Chang et al., 1999) were determined as the parameters to elucidate the UV-B tolerance limit. The extent of lipid peroxidation estimated by a thiobarbituric acid reacting substance (TBARS) and the contents of peroxide served to indicate the degree of oxidative damage. The contents of both thallus and seawater H2O2 were quantified as the representatives of ROS generation by U. fasciata. The dynamics of water-soluble antioxidants and ROS detoxifying enzyme activities were assessed. An attempt was also made to investigate whether
Materials and methods
Algal materials, treatments and seawater samples
Ulva fasciata at 15–25 cm plant height was collected between August and November 2004 from the high intertidal regions at Hsitzu Bay, Kaohsiung, Taiwan. After harvesting, whole algae were extensively washed with natural seawater to remove any attached sand and the rhizoidal portions were removed to avoid microbial contamination in the following culture. Thalli were preincubated at 25 °C for 14 d in 35‰ nutrient-enriched artificial seawater (ASW) containing 403.5 mM NaCl, 10 mM KCl, 10 mM CaCl2, 30 mM MgSO4, 10 mM TRIS–HCl (pH 8.0), N-
To elucidate the involvement of H2O2 in the UV-B-induced antioxidant defence system, thallus segments (0.7 g w. wt.) after incubation as mentioned above were first cultured in a Petri dish (internal diameter: 10 cm) containing 30 ml of 10 mM SB, 100 μM Tiron, or 100 μM DTMU (prepared in 35‰ nutrient-enriched ASW) for 1 h in darkness and then transferred to 2.5 W m−2 UV-B radiation for 12 h under 150 μmol photons m−2 s−1 PAR provided by 40 W cool-fluorescent lamps (FL40D, China Electric Apparatus Ltd., Taiwan, Republic of China).
After treatments, thallus segments were sampled and divided into three parts: the first part was immediately used for the TTC reduction ability assay, the second part for the determination of lipid peroxidation and the contents of peroxide, H2O2 and water-soluble antioxidants was fixed in liquid nitrogen and kept in a freezer at −20 °C until analysis, and the third part was fixed in liquid nitrogen and lyophilized at −40 °C for the enzyme assay.
For the determination of seawater H2O2 and peroxide contents, seawater samples were collected daily and assayed immediately. In this study, each Petri dish was considered as a replicate in all the statistical analysis and there were three replicates for each treatment.
Determination of TTC reduction ability
To determine the cellular activity, freshly sampled thallus segments of approximately 0.05 g w. wt. were incubated at 25 °C in 1.5 ml of 50 mM potassium phosphate buffer (pH 7.4) containing 0.8% (w/v) 2,3,5-triphenyltetrazolium chloride (TTC) and 35‰ ASW under darkness for 16 h (Chang et al., 1999). After washing three times with 10 ml of 35‰ ASW, intracellular insoluble formanzan was extracted twice with 5 ml of 95% ethanol at 80 °C for 20 min and the ethanol extracts were combined. After making up to 10 ml, the absorbance of combined ethanol extract was determined at A530. The A530 values of UV-B-treated thallus segments were calculated as the percentage of the control, that is, the UV-B-free treatment.
Determination of chlorophyll a and carotenoid contents
Determination of lipid peroxidation, peroxide and H2O2 contents
Thallus segments of approximately 0.1 g w. wt. were ground in liquid nitrogen and then 1 ml of 5% (w/v) trichloroacetic acid (TCA) was added. After centrifugation at 12 000 g for 10 min at 4 °C, the supernatant was collected as a TCA extract for the determination of lipid peroxidation, peroxide, and H2O2.
The levels of lipid peroxidation were determined from the thiobarbituric acid reacting substance (TBARS) contents resulting from the thiobarbituric acid (TBA) reaction as described by Health and Packer (1968). The TBARS contents were calculated based on A532–A600 with the extinction coefficient of 155 mM−1 cm−1.
Thallus and seawater peroxide contents were determined by the absorbance at A480 and extinction coefficient of 13.93 μM−1 cm−1 according to Sagisaka (1976). Because seawater was changed every day, the contents of peroxide in seawater were determined every day and they were summed as cumulative seawater peroxide over 4 d.
Thallus H2O2 contents were determined based on the decomposition of H2O2 by peroxidase as described by Okuda et al. (1991). KOH (4 M) of 11.51 μl was added to 0.2 ml supernatant to adjust the pH to 7.5 and the mixture was centrifuged at 12 000 g for 1 min at 4 °C. The supernatant was collected and applied to a 1 ml column of Amberlite IRA-410, and residual H2O2 was washed out by 800 μl of distilled water. The contents of H2O2 in the eluate were determined within 10 min. For the determination of H2O2 in the eluate, 0.4 ml of 12.5 mM 3-dimethylaminobenzoic acid (DMAB), 0.4 ml of 10 mM 3-methyl-2-benzothiazoline hydrazone (MBTH), and finally 0.02 ml of 0.25 unit ml−1 horsedish peroxidase (Sigma, USA) was added and then the reaction was measured at 590 nm for 3 min. The amount of thallus H2O2 was estimated from the H2O2 standard curve that had been determined as described above.
Seawater H2O2 contents were also determined following the same method for thallus H2O2 determination. One ml of the sampled seawater was immediately passed through a 1 ml column of Amberlite IRA-410 and then the residual H2O2 in the column was eluted by applying 800 μl Milli-Q water. Eluates were combined and adjusted to pH 7.0 with 4 M KOH. Seawater H2O2 contents were calculated based on the calculation from a series of H2O2 concentrations prepared in nutrient-enriched ASW. H2O2 contents in seawater were determined daily and they were summed as cumulative seawater H2O2 over 4 d.
Determination of ascorbate and glutathione contents
Thallus segments of approximately 0.1 g w. wt. were ground in liquid nitrogen and then 1 ml of 5% (w/v) trichloroacetic acid (TCA) was added. After centrifugation at 12 000 g for 10 min at 4 °C, the supernatant was collected as a TCA extract for the determination of water-soluble antioxidant contents.
The measurement of total ascorbate and reduced ascorbate (AsA) contents were modified from the method of Hodges et al. (1996). Total ascorbate contents were determined in a 1 ml mixture containing 200 μl TCA extract, 50 mM potassium phosphate buffer (pH 7.4), 3 mM EDTA, and 1 mM dithiothreitol (DTT). The mixture was incubated at 25 °C for 10 min, 100 μl of N-ethylmaleimide was added, and then 400 μl of 0.61 M TCA, 400 μl of 0.8 M orthophosphoric acid, and 400 μl of α,α′-bipyridyl were added. Finally, 200 μl of FeCl3 was added and the mixture was incubated in a 55 °C water bath for 10 min and the absorbance was detected at A525. AsA contents were determined by adding distilled water instead of DTT and N-ethylmaleimide and then followed the same method as above. Total AsA and AsA contents were estimated from the standard curve of 0–40 nmol L-AsA determined by the above methods. DHA contents were calculated by the subtraction of AsA from total AsA.
Total glutathione contents were determined by the absorbance at 570 nm according to the method of Griffiths (1980). The contents of glutathione (reduced form) were estimated from the standard curve of 0–20 nmol glutathione. After the removal of glutathione by 2-vinylpyridine derivatization, glutathione disulphide contents were determined, and the glutathione contents were calculated by the subtraction of glutathione disulphide contents from total glutathione contents.
Determination of antioxidant enzyme activity
Lyophilized thallus discs of approximately 0.005 g dry wt. (d. wt.) were first homogenized in liquid nitrogen and 0.6 ml of 0.1 M sodium phosphate buffer (pH 6.8) containing 1% (w/v) polyvinylpolypyrrolidone (PVPP) and 0.25% (v/v) Triton X-100 was then added. After centrifugation at 12 000 g for 10 min at 4 °C, the supernatant was used for enzyme activity assay of SOD, CAT, POX, GR, MDHAR, and DHAR. For the determination of APX, 0.005 g d. wt. of lyophilized thallus discs was homogenized in liquid nitrogen and 0.6 ml of extraction buffer (0.1 M sodium phosphate buffer (pH 6.8) containing 1% (w/v) PVPP, 0.25% (v/v) Triton X-100, and 0.5 mM L-ascorbate) was added. After centrifugation at 12 000 g for 10 min at 4 °C, the supernatant was used for the APX assay. The soluble protein contents were determined by the Coomassie blue dye binding method (Bradford, 1976) with bovine serum albumin as the standard curve.
The CAT activity was measured at A420 for H2O2 decomposition rate using the extinction coefficient of 40 mM−1 cm−1 according to Kato and Shimizu (1987). Guaiacol POX activity was determined by the formation rate of tetraguaiacol detected at A470 with the extinction coefficient of 26.6 mM−1 cm−1 according to Kato and Shimizu (1987). SOD activity was determined by the inhibition of photochemical inhibition of nitro blue tetrazolium according to Giannopolitis and Ries (1977). APX activity was determined at A290 for DHA according to the extinction coefficient of 2.8 mM−1 cm−1 (Nakano and Asada, 1981). GR activity was monitored by A340 for β-NADPH oxidization as GSSG reduction according to Schaedle and Bassham (1977).
Both MDHAR and DHAR activities were determined according to Hou and Lin (1997). DHAR activity was measured by monitoring the absorbance at 265 nm for 3 min in the reaction mixture that consisted of enzyme extract, 50 mM Na-phosphate buffer (pH 7.0), 0.3 mM glutathione, 0.06 mM Na2EDTA and 0.2 mM DHA. MDHAR activity was measured by the monitoring the absorbance at 340 nm for 3 min in 50 mM Na-phosphate buffer (pH 7.6), 0.1 mM β-NADPH and 0.1 unit AsA oxidase (Sigma, MO, USA) and 2.5 mM AsA. Non-enzymatic reduction of DHA or MDHA in phosphate buffer was measured in a separate cuvette at the same time.
Chemicals and statistical analysis
Chemicals were purchased from Merck (Germany) or Sigma (USA). The data were analysed by SAS (SAS version 8.1, NC, USA). The present results were the mean of three replicates with a Petri dish as a replicate. The effects of UV-B dose on TTC reduction ability, TBARS contents, peroxide contents, H2O2 contents, water-soluble antioxidant contents, and enzyme activities, and the effects of ROS scavengers were analysed by 1-way analysis of variance (ANOVA). The difference among means was analysed by Duncan's new multiple range test following by significant ANOVA at P <0.05.
Results
Growth rate, TTC reduction ability, and the contents of chlorophyll a, carotenoid, TBARS, peroxide, and H2O2
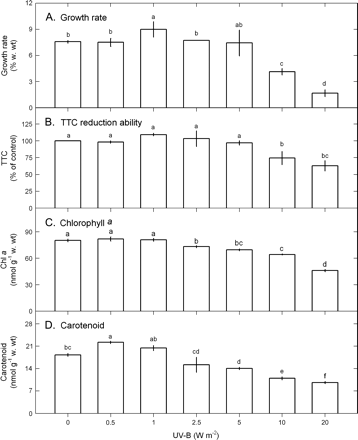
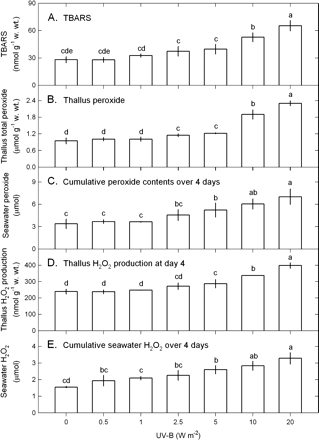
The tolerance limit of U. fasciata to UV-B exposure was evaluated by the changes of growth rate, TTC reduction ability, and chlorophyll a and carotenoid contents 4 d after exposure to varying UV-B doses. The data in Fig. 1 show that growth rate, TTC reduction ability, chlorophyll a contents, and carotenoid contents were all affected by UV-B (1-way ANOVA, P <0.05). Growth rate (Fig. 1A) and TTC reduction ability (Fig. 1B) were not affected by UV-B ranging from 0.5–5 W m−2 and were significantly decreased by UV-B ≥10 W m−2. Chlorophyll a (Fig. 1C) and carotenoid (Fig. 1D) contents were not influenced by the low UV-B dose ranging from 0.5–1 W m−2 and slightly declined at 2.5–5 W m−2 UV-B followed by a marked drop at 10–20 W m−2 UV-B. The changes in lipid peroxidation and total peroxide production were in contrast to pigments. Compared with the control, TBARS contents remained unchanged on exposure to UV-B ranging from 0.5–1 W m−2 and then slightly rose at 2.5–5 W m−2 UV-B followed by a marked increase at 10–20 W m−2 (Fig. 2A). Both thallus total peroxide contents (day 4) (Fig. 2B) and cumulative seawater peroxide contents (over 4 d) (Fig. 2C) were not affected by 0.5–1 W m−2 UV-B followed by a slight increase at 2.5–5 W m−2 and a marked increase at 10–20 W m−2.
Changes in growth rate (A), TTC reduction ability (B), chlorophyll a (C), and carotenoid (D) in Ulva fasciata in response to varying UV-B doses. Data are present as means ±SD (n=3) and different letters indicate significant difference among treatments.
Changes in the contents of TBARS (A), total peroxide in thallus (B) and seawater (C), and H2O2 in thallus (D) and seawater (E) in Ulva fasciata in response to varying UV-B doses. Data are present as means ±SD (n=3) and different letters indicate significant difference among treatments.
Next, the effects of UV-B on H2O2 production by thallus segments was investigated. The preliminary experiments found a large quantity of H2O2 detected in seawater after incubation of U. fasciata thallus segments. It seems to suggest that H2O2 produced by thallus segments could be excreted into seawater. Therefore, in addition to H2O2 in thallus segments, the changes in H2O2 contents in seawater were also quantified. The data in Fig. 2 show that thallus H2O2 contents (day 4) and cumulative seawater H2O2 contents (over 4 d) were affected by UV-B (1-way ANOVA, P <0.05). Compared with the control, thallus H2O2 contents were not affected by UV-B ranging from 0.5–1 W m−2 and then slightly rose at 2.5–5 W m−2 UV-B followed by a significant increase at 10–20 W m−2 (Fig. 2D). Cumulative seawater H2O2 contents over 4 d showed a similar pattern (Fig. 2E). The amount of thallus H2O2 contents at day 4 and cumulative seawater H2O2 contents over 4 d appears to be a dose-response, indicating that the generation of H2O2 by thallus segments is positive depending on UV-B dose. In addition, the higher H2O2 contents in seawater than those in the thallus segments reflect that most of H2O2 produced by thallus segments could be excreted into seawater, and it was found that the release of H2O2 was increased by increasing UV-B radiation. Thus, the defence system of U. fasciata is working against UV-B-induced H2O2 accumulation and oxidative stress that can be alleviated at 0.5–1 W m−2 but could not be overcome when UV-B was ≥2.5 W m−2, thus leading to oxidative damage, especially at 10–20 W m−2.
Ascorbate and glutathione contents
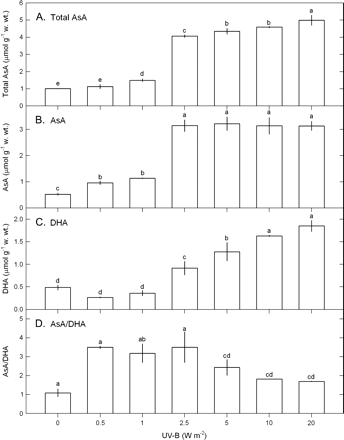
Ascorbate and glutathione contents were determined to test whether the water-soluble antioxidants are involved in the defence system against UV-B-induced oxidative stress in U. fasciata. The contents of total AsA, AsA, and DHA and the ratios of AsA/DHA were significantly influenced by UV-B (ANOVA, P <0.05) (Fig. 3). Total AsA (Fig. 3A) and AsA (Fig. 3B) contents increased with increasing UV-B (0.5–20 W m−2) while DHA contents (Fig. 3C) increased when UV-B was ≥2.5 W m−2. To explore the regeneration rate of AsA, the ratio of AsA/DHA was calculated. The data in Fig. 3D show that, upon exposure to UV-B, the ratio of AsA/DHA increased with the maximum occurring in a range from 0.5–2.5 W m−2 followed by a drop when UV-B was ≥5 W m−2.
The contents of total AsA (A), AsA (B), DHA (C), and the ratios of AsA/DHA (D), in Ulva fasciata in response to varying UV-B doses. Data are present as means ±SD (n=3) and different letters indicate significant difference among treatments.
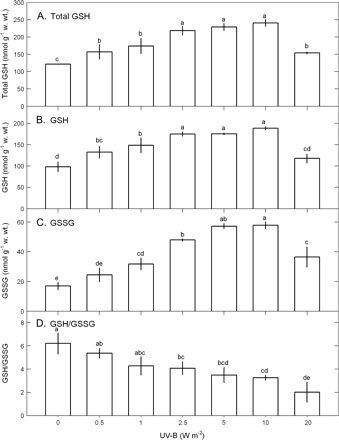
Total glutathione, glutathione, and glutathione disulphide contents and glutathione/glutathione disulphide ratios were affected by UV-B (ANOVA, P <0.05) (Fig. 4). The contents of total glutathione (Fig. 4A), glutathione (Fig. 4B), and glutathione disulphide (Fig. 4C) were increased with increasing UV-B dose, with the maximum occurring in a range of 5–10 W m−2 followed by a drop at 20 W m−2. Glutathione/glutathione disulphide ratio as an indicator of glutathione regeneration rate decreased with increasing UV-B dose because the magnitude of glutathione disulphide increments was higher than that of glutathione increments (Fig. 4D).
The contents of total glutathione (A), glutathione (B), glutathione disulphide (C), and the ratios of glutathione/glutathione disulphide (D) in Ulva fasciata in response to varying UV-B doses. Data are present as means ±SD (n=3) and different letters indicate significant difference among treatments.
Enzyme activities
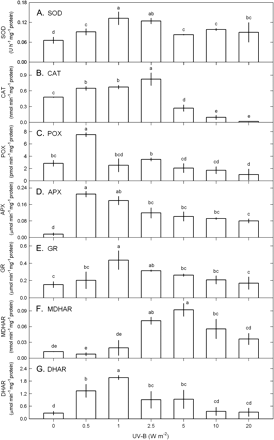
UV-B significantly affected SOD activity (ANOVA, P <0.05) that increased as UV-B increased from 0.5–2.5 W m−2 and subsequently decreased upon exposure to a higher UV-B dose, that is, 10–20 W m−2 (Fig. 5A).
The activities of SOD (A), CAT (B), POX (C), APX (D), GR (E), MDHAR (F), and DHAR (G) in Ulva fasciata in response to varying UV-B doses. Data are present as means ±SD (n=3) and different letters indicate significant difference among treatments.
Both CAT and POX activities were affected by UV-B (P <0.05); the CAT activity increased as UV-B increased from 0.5–2.5 W m−2 and then decreased to the level lower than the control when UV-B was ≥5 W m−2 (Fig. 5B), while the POX activity only increased at 0.5 W m−2 (Fig. 5C).
The activities of enzymes in the ascorbate–glutathione cycle were affected by UV-B (P <0.05) (Fig. 5). Upon exposure to UV-B, both APX (Fig. 5D) and GR (Fig. 5E) activities increased fast with the maximum occurring at 0.5–1 W m−2 and then decreased gradually as UV-B increased from 2.5 to 20 W m−2, but they were still higher than the control. MDHAR activity increased when UV-B was ≥2.5 W m−2; the maximum occurring at 5 W m−2 and then a decrease at 10–20 W m−2, they were still higher than the control (Fig. 5F). Compared with the response of MDHAR under high UV-B doses (2.5–20 W m−2 UV-B), DHAR responded to low UV-B doses ranging from 0.5–5 W m−2 with the maximum at 1 W m−2 (Fig. 5G).
Effects of SB, Tiron, and DMTU on UV-B-induced oxidative stress, antioxidant contents, and enzyme activities
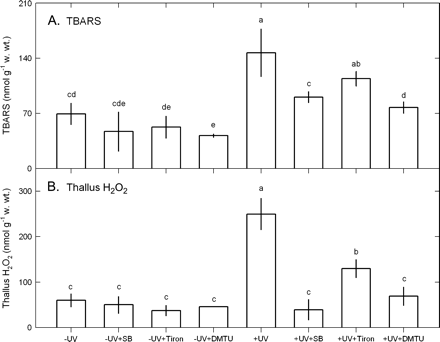
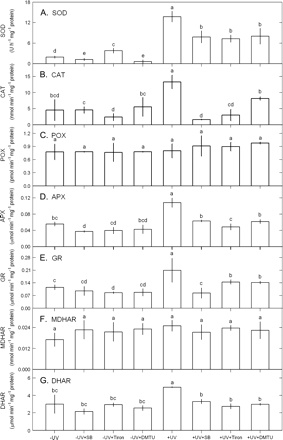
The short-term exposure to 2.5 W m−2 UV-B for 12 h increased the contents of TBARS and thallus H2O2 (Fig. 6), the activities of SOD, CAT, APX, GR, and DHAR (Fig. 7), and the contents of AsA and glutathione. However, the activities of POX and MDHAR were not induced by this short-term UV-B exposure.
Effects of 10 mM SB, 100 μM Tiron, or 100 μM DTMU on the contents of TBARS (A) and thallus H2O2 (B) in Ulva fasciata on exposure to UV-B (2.5 W m−2). Data are present as means ±SD (n=3) and different letters indicate significant difference among treatments.
Effects of 10 mM SB, 100 μM Tiron, or 100 μM DTMU on the activities of SOD (A), CAT (B), POX (C), APX (D), GR (E), DHAR (F), and MDHAR (G) in Ulva fasciata on exposure to UV-B (2.5 W m−2). Data are present as means ±SD (n=3) and different letters indicate significant difference among treatments.
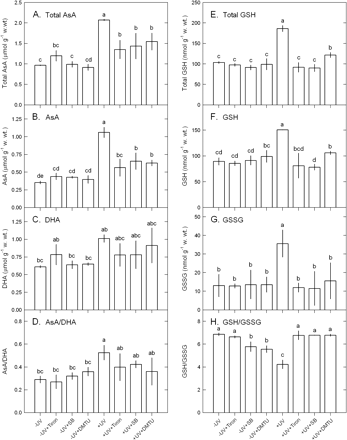
The UV-B-induced increase in the contents of TBARS and thallus H2O2 was inhibited by SB, Tiron, and DMTU (Fig. 6). Increased SOD, CAT, APX, GR, and DHAR activities by UV-B exposure were also depressed by SB, Tiron, and DMTU (Fig. 7). However, POX and MDHAR activities were not affected (Fig. 7). The UV-B-induced increase in total AsA, AsA, and DHA contents and AsA/DHA ratios were inhibited by SB, Tiron, and DMTU (Fig. 8). The UV-B-induced increase in the contents of total glutathione, glutathione, and glutathione disulphide was also inhibited by SB, Tiron, and DMTU, and the decrease in the ratios of glutathione/glutathione disulphide in UV-B-treated thalli was reversed by SB, Tiron, and DMTU (Fig. 8).
Effects of 10 mM SB, 100 μM Tiron, or 100 μM DTMU on the contents of total AsA (A), AsA (B), and DHA (C), the ratios of AsA/DHA ratio (D), the contents of total glutathione (E), glutathione (F), and glutathione disulphide (G), and the ratios of glutathione/glutathione disulphide (H) in Ulva fasciata on exposure to UV-B (2.5 W m−2). Data are present as means ±SD (n=3) and different letters indicate significant difference among treatments.
Discussion
This study was aimed at a better understanding of the regulation of the antioxidant defence system in a tropical intertidal chlorophyte U. fasciata in response to varying UV-B doses. Using growth rate, TTC reduction ability, photosynthesis pigments, total peroxide, and lipid peroxidation as indicators, the UV-B tolerance limit in U. fasciata has been identified. Increased TBARS and total peroxide contents but decreased chlorophyll a and carotenoid contents in medium (2.5–5 W m−2) and high (10–20 W m−2) UV-B doses suggest the occurrence of oxidative damage when UV-B was ≥2.5 W m−2. However, the oxidative damage was mild in medium UV-B doses as indicated by a relatively small increase in lipid peroxidation and total peroxide production and a slight decrease in the chlorophyll a and carotenoid contents. In addition, both growth rate and TTC reduction ability were only decreased by high UV-B doses. This means that the defence and repairing systems are operating normally in low (0.5–1 W m−2) and medium UV-B radiation doses with 5 W m−2 as the tolerance limit. The limit of U. fasciata to UV-B radiation is similar to the current maximum summer Taiwan exposure (4.08±0.53 W m−2). It is reasonable for this high intertidal chlorophyte, in which significant UV-B exposure will occur when the alga is submerged. Ecophysiological studies have suggested that the physiological responses of macroalgae to UV-B are associated with their zonation pattern; for example, the sensitivity of photosynthetic electron transport activity to UV-B is correlated with the vertical distribution pattern of the alga (Dring et al., 1996; Bischof et al., 1998). A survey of the UV tolerance of marine rhodophytes from tropical habitats showed that UV exposure caused less pronounced effects in intertidally occurring species than in subtidal species (van de Poll et al., 2003). Arctic marine macroalgal species from eulittoral and upper sublittoral regions also showed higher antioxidant enzyme activities than species from lower sublittoral regions upon exposure to UV (Aguilera et al., 2002). It would be expected that the antioxidant defence system has evolved to protect high intertidal U. fasciata from UV-B-induced oxidative stress.
Increasing H2O2 contents in thallus segments and seawater with increasing UV-B doses reflect the stimulation of H2O2 generation in U. fasciata by UV-B. Although the contents of
The excessive production of
The detection of a large quantity of H2O2 in seawater after incubation of U. fasciata thallus segments and the parallel increase of cumulative seawater H2O2 contents and thallus segment H2O2 contents in response to increasing UV-B doses seem to suggest that H2O2 overproduced by UV-B-treated thalli could be excreted into seawater. The ability to excrete accumulated H2O2 into the surrounding medium for preventing H2O2 toxicity has been reported in the chlorophyte Ulva rigida C. Ag. (Collén et al., 1995), the brown macroalga Fucus sp. (Collén and Davison, 1999a, b), and the economically important carrageenophyte Kappaphycus alvarezii (Doty) Doty (a red macroalga) (Barros et al., 2003). The excretion of H2O2 into the surrounding water is considered one of the mechanisms for algae to avoid harmful accumulation of cellular H2O2 (Collén et al., 1995; Barros et al., 2003). Alternatively, H2O2 in the medium of UV-B-treated U. fasciata may be produced at the external surface of the plant cells. Studies have shown that ROS such as
Evidence shows that the availability of antioxidants and the activities of antioxidant enzymes in the ascorbate–glutathione cycle are involved in H2O2 scavenging in U. fasciata under UV-B radiation. Upon exposure to low UV-B flux (0.5–1 W m−2), a significant AsA regeneration, as indicated by a marked increase in AsA/DHA ratio, suggests that a rising AsA regeneration rate is crucial for the proper scavenging of accumulated ROS in U. fasciata under low UV-B doses. Compared with low UV-B doses, marked increases in the contents of AsA, DHA, and total AsA, as well as high ratios of AsA/DHA in response to medium UV-B radiation (2.5–5 W m−2 ), indicate that an increasing AsA pool in combination with the rapid regeneration of DHA to AsA are attributable to oxidative defence in response to moderate UV-B radiation. The ability of AsA regeneration is inhibited by high UV-B flux (10–20 W m−2) as indicated by a significant drop in AsA/DHA ratios when UV-B increased to 10–20 W m−2. Glutathione also responds to UV-B stress as one component in defence system. Similar to ascorbate, the pools of glutathione and their regeneration rates are increased for detoxifying ROS under UV-B conditions. Although glutathione/GSSG ratios were decreased by UV-B, the data that the GR activity of UV-B-treated U. fasciata was higher or remained unchanged compared with the control reflect that glutathione regeneration was not inhibited by UV-B, instead glutathione was consumed faster than its regeneration. The reverse of the UV-B-induced glutathione/glutathione disulphide ratio decrease by ROS scavengers (SB, Tiron, and DMTU) supports this view. In intertidal brown and red macroalgae (Collén and Davison, 1999a, b, c), higher antioxidant levels are positively associated with their stress tolerance. Instead of keeping a high quantity of antioxidants under normal condition, the antioxidant defence ability in U. fasciata is achieved by increasing the amounts of AsA and glutathione as well as their regeneration rate under UV-B stress.
Increased APX activity functions to keep the balance of cellular H2O2 components in U. fasciata in response to low UV-B flux. Both CAT and POX are also responsible for the removal of excess H2O2 under low UV-B flux, as indicated by increasing CAT activity with increasing UV-B radiation from 0.5–2.5 W m−2 and increased guaiacol POX activity at 0.5 W m−2. As the substrate of APX reaction, the availability of AsA is becoming essential for U. fasciata under UV-B stress. Evidence shows that the recycling of AsA is mediated by MDHAR and DHAR in the generation of GR. DHAR is responsible for AsA regeneration at low UV-B doses while MDHAR operates at medium UV-B doses. Because glutathione is used as an electron donor for reducing DHA to generate AsA by DHAR and there was the same pattern of DHAR and GR upon exposure to varying UV-B doses, the decline of glutathione regeneration due to diminished GR activities when UV-B was ≥2.5 W m−2 explains why AsA was not regenerated via the DHAR route under medium and high UV-B flux. Although AsA and glutathione pools are increasing as the UV-B flux increases, the extent of UV-B-induced APX and GR activity increase under medium and high UV-B radiation was less than that under low UV-B flux. It is why the oxidative damage (lipid peroxidation and total peroxide production) was significant under medium and high UV-B radiation.
H2O2 has been known as the signal for the induction of the antioxidant defence system in plants in response to biotic and abiotic stresses (Neil et al., 2002a, b). Current results suggest that the induction of the antioxidant defence system in U. fasciata is mediated by
In conclusion, the excretion of H2O2 as well as the availability of antioxidants and the activation of SOD, CAT, guaiacol POX, and reactive oxygen scavenging enzymes in the ascorbate–glutathione cycle serve as the defence system against oxidative stress occurring in U. fasciata upon exposure to UV-B. UV-B disrupts the balance between the production and removal of H2O2 and subsequently accumulated H2O2 initiates the signalling responses leading to the induction of enzymatic antioxidant defence systems to overcome ROS production in U. fasciata.
We gratefully thank two anonymous reviewers for constructive comments and notions that improved the manuscript. Financial support from the National Science Council (NSC93-2311-B-110-004) and the Council of Agriculture (grant No. 93AS-2.4.1-FB-e1(3), 94AS-9.4.4-FB-e1(6)), Executive Yuan, Taiwan, Republic of China, to Tse-Min Lee is acknowledged.
References
Aguilera J, Dummermuth A, Karsten U, Schriek R, Wiencke C.
Alscher RG, Erturk N, Heath LS.
Arnon DI.
Asada K.
Asada K, Takahashi M.
Baker KS, Smith RC, Green AES.
Barros MP, Pedersen M, Colepicolo P, Snoeijs P.
Beckett RP, Minibayeva FV, Vylegzhanina NN, Tolpysheva T.
Bischof K, Hanelt D, Wiencke C.
Bischof K, Hanelt D, Wiencke C.
Bradford MM.
Bolwell GP.
Bolwell GP, Davies DR, Gerrish C, Auh C-K, Murphy TM.
Butow B, Wynne D, Tel-Or E.
Caldwell MM.
Calkins J, Thordardottir T.
Chang WC, Chen MH, Lee TM.
Collén J, Davison IR.
Collén J, Davison IR.
Collén J, Davison IR.
Collén J, Del Rio MJ, Garcia-Reina G, Pedersen M.
Davis KJA.
Dring MJ, Wagner A, Boeskov J, Lüning K.
Foyer CH, Lelandais M, Kunert KJ.
Giannopolitis CN, Ries SK.
Griffiths OW.
Grobe CW, Murphy TM.
Häder DP.
He YY, Klisch M, Hader DP.
Health RL, Packer G.
Hodges DM, Andrews CJ, Johnson DA, Hamilton RI.
Hou WC, Lin YH.
Hung KT, Kao CH.
Karpinski S, Reynolds H, Karpinska B, Wingsle G, Creissen G, Mullineaus P.
Kato M, Shimizu S.
Kerr JB, McElroy CT.
Lesser MP.
Lesser MP, Shick JM.
Levine A, Tenhaken R, Dixon R, Lamb C.
Malanga G, Puntarulo S.
Malanga G, Calmanovici G, Puntarulo S.
McKersie BD, Leshem Y.
Minibayeva F, Beckett RP.
Mittler R.
Morita S, Kaminaka H, Masumura T, Tanaka K.
Munné-Bosch S, Alegre L.
Nakano Y, Asada K.
Neil SJ, Desikan R, Hancock J.
Neil SJ, Desikan R, Clarke A, Hurst RD, Hancock JT.
Noctor G, Foyer CH.
Okuda T, Matsuda Y, Yamanaka A, Sagisaka S.
Provasoli L.
Rijstenbil JW.
Russell JM, Luo MZ, Cicerone RJ, Deaver LE.
Sagisaka S.
Seckmeyer G, McKenzie RL.
Smirnoff N, Wheeler GL.
Smith RC, Prezelin BB, Baker KS, et al.
Tschiersch H, Ohmann E.
van de Poll WH, Bischof K, Buma AGJ, Breeman AM.
Vranova E, Inzé D, Breusegem FV.
Willekens H, Chamnogopol S, Davey M, Schraudner M, Langebartels C.
Wojtaszek P.




Comments