-
PDF
- Split View
-
Views
-
Cite
Cite
Donald E. Aylor, Baltazar M. Baltazar, John B. Schoper, Some physical properties of teosinte (Zea mays subsp. parviglumis) pollen, Journal of Experimental Botany, Volume 56, Issue 419, September 2005, Pages 2401–2407, https://doi.org/10.1093/jxb/eri232
Close - Share Icon Share
Abstract
In parts of the world where teosinte and maize are grown in close proximity, there is concern about gene flow between them. Pollen is the primary vehicle for gene flow. Quantifying the biophysical properties of pollen, such as its settling speed and dehydration rate, is important for evaluating outcrossing potential. These properties were measured for teosinte (Zea mays subsp. parviglumis) pollen. Pollen was found to have an average settling speed of 0.165 m s−1, which agrees well with theoretical values based on the size of the pollen grains. The conductance of the pollen wall for water was derived from the time rate of change of pollen grain size and gave an average conductance of 3.42×10−4 m s−1. Water potential, ψ, of teosinte pollen was determined at various values of relative water content (dry-weight basis), 𝛉, by using a thermocouple psychrometer and by allowing samples of pollen to come to vapour equilibrium with various saturated salt solutions. Non-linear regression analysis of the data yielded ψ (MPa) = −4.13 𝛉−1.23 (r2=0.77). Results for conductance and ψ were incorporated into a model equation for the rate of water loss from pollen grains, which yielded results that agreed well (r2=0.96) with observations of water loss from pollen grains in air. The data reported here are important building blocks in a model of teosinte pollen movement and should be helpful in establishing the main factors influencing the degree and the direction of pollination between teosinte populations and between maize and teosinte.
This paper is available online free of all access charges (see http://jxb.oupjournals.org/open_access.html for further details)
The online version of this article has been published under an Open Access model. Users are entitled to use, reproduce, disseminate, or display the Open Access version of this article for non-commercial purposes provided that: the original authorship is properly and fully attributed; the Journal and the Society for Experimental Biology are attributed as the original place of publication with the correct citation details given; if an article is subsequently reproduced or disseminated not in its entirety but only in part or as a derivative work this must be clearly indicated. For commercial re-use, please contact: journals.permissions@oupjournals.org
Introduction
Teosinte (Zea spp.) is an annual and perennial grass endemic to Mexico and Central America. Current theories place teosinte at the centre in the development of and in the variation found among the principal landraces in Mexico (Wellhausen et al., 1952; Mangelsdorf, 1974; Matsuoka et al., 2002). As such, the potential use of teosinte in maize breeding has been evaluated since the 1950s. Researchers concluded that teosinte is a valuable germplasm for maize improvement, providing resistance to diseases and other abiotic stresses (Reeves, 1950), as well as quantitative traits (Cohen and Galinat, 1984; Casas et al., 2001). In addition, teosinte is itself culturally important because of the beverages and special culinary dishes that are prepared, particularly in regions of Mexico where it grows in the wild.
Teosinte is represented by annual diploid species (Zea mays ssp. mexicana and Zea mays ssp. parviglumis) and by perennial diploid (Zea diploperennis) and tetraploid (Zea perennis) species. It is found within tropical and subtropical areas of Mexico in isolated populations of varying sizes, sometimes occupying several square kilometres but most often in patches of less than a square kilometre (Sánchez et al., 1998). Natural gene exchange between maize and teosinte (Wilkes, 1977), as well as hybridization and human selection, have led to the genetic diversity that is found in Mexican farmers' fields today. As a result, there are at least 60 maize landraces in Mexico (Sánchez et al., 2000).
Maize-to-maize gene flow is well documented (Baltazar and Schoper, 2002). Mexican farmers have promoted gene flow by planting side-by-side landraces, improved open-pollinated species, and hybrids, thereby increasing the genetic diversity in Mexico (Bellon and Brush, 1994; Bellon and Risopoulos, 2001). Low levels of gene flow between maize and teosinte have been documented (Baltazar et al., 2005). Results of this study support the hypothesis that gene flow and subsequent introgression of maize alleles into teosinte populations most likely result from crosses where teosinte first pollinates maize. This underscores the importance of understanding teosinte pollen movement. The resultant hybrids then backcross with teosinte to introgress the maize alleles into the teosinte genome. The initial pollination from teosinte onto maize silks most likely represents the rate-limiting step in the eventual introgression of maize alleles into the teosinte genome and may help explain why teosinte continues to coexist as a separate entity, even though it normally grows in the vicinity of much larger populations of maize.
Studies of isolation distance between maize plants in Tapachula, Nayarit, showed that very limited cross-pollination occurred at 100 m and no pollination occurred at distances beyond 200 m (Luna et al., 2001). Wind and atmospheric conditions at the time pollen was released into the atmosphere were critical factors affecting pollen movement and longevity (Luna et al., 2001; Baltazar and Schoper, 2001; Baltazar et al., 2005). Other studies showed that pollination could be as high as 60% between contiguous rows, but pollination from more remote rows decreased with increasing distance such that no outcrosses were found 32 m away from the pollen source (Castillo and Goodman, 1997; Cervantes, 1998). However, wind speed and direction, turbulence, and pollen settling speed affect pollen movement (Di-Giovanni and Kevan, 1991; Aylor et al., 2003), and variability in these factors can theoretically change the distance of cross-pollination.
At present, little is known about teosinte pollen movement in the atmosphere. To help fill this gap, data on the settling speed, water potential, germination rate, drying rate, and the resistance to water loss of teosinte pollen are presented in this paper. These data on the physical properties of teosinte pollen are important building blocks in a model of teosinte pollen movement and, as such, will be helpful in establishing the main factors influencing the degree and direction of pollination between teosinte populations and between maize and teosinte. Consequently, they will help to increase our understanding of the gene-flow process in Mexico and Central America where teosinte and maize populations grow in geographic proximity and flower synchronously.
Materials and methods
Source of pollen
The pollen used in all tests was freshly collected from teosinte plants (Zea mays subsp. parviglumis, Accession: Ames 21785, Lot: 92ncao01, North Central Plant Introduction Station, Ames, Iowa, USA) grown in the greenhouse in 4.0 l plastic pots in a soil mixture of Promix BX, sand, and Perlite in the ratio (2:1:1 by vol.). New plantings were begun in the greenhouse every four to five days to ensure a steady and continuous supply of fresh pollen over several weeks. Fresh pollen was collected from anthers on plants by gently tapping the stems of plants while holding a collecting tray below the tassels. To help ensure that freshly produced pollen was collected, the tassels were ‘milked’ of pollen, either at the end of the previous day or in the morning of the same day, to remove old pollen prior to a collection of fresh pollen used in experiments. Any anthers inadvertently collected with the pollen were removed from the dishes prior to exposure using forceps.
Settling speed
Measurements of the settling speed of teosinte pollen in still air, vS, were made by timing the rate of fall of pollen grains inside a glass settling tube (Aylor, 2002). In particular, the time for pollen to fall a distance of 1.0 m was determined using a stopwatch. In addition, a microscope slide was momentarily placed just below the opening at the bottom of the tube to capture (by sedimentation) the particular group of pollen grains being timed. These grains were immediately examined microscopically to determine their characteristic dimensions (principal diameters) and shapes. In this way, individual collections of pollen were used to measure the settling speed and size of pollen grains over a range of pollen sizes.
Teosinte pollen grains are shaped like prolate spheroids, where L1 and L2 are major and minor principal diameters of the pollen grain. A volume-equivalent diameter, De, given by:
Water potential versus water content
The relative water content of teosinte pollen is expressed as a fraction of dry weight, namely, 𝛉=mw/md, where mw is the mass of water in a sample of teosinte pollen and md is the mass of the same sample of oven-dry pollen. The value of mw at a given time was determined by subtracting md from the pollen mass at that time. For the teosinte pollen used in these studies, 𝛉 ranged from ∼1.35 for hydrated teosinte pollen, fresh from the anther, to zero for oven-dry pollen.
A relationship between water potential, ψ (MPa), and relative water content, 𝛉 (dimensionless), for teosinte pollen was obtained using two methods (Aylor, 2003). In the first method, pollen was first allowed to reach moisture equilibrium above various saturated salt solutions having relative humidity ranging from ∼11% to 94% and then the equilibrium water content of the pollen was determined gravimetrically. In the second method, pollen water potential was determined using a thermocouple psychrometer (comprised of a Model HR-33T Dew point microvolt meter and a Model C-52 sample chamber, Wescor, Inc., Logan, Utah, USA) and water content was again determined gravimetrically.
Germination versus water content
Teosinte pollen was placed in individual sample weighing dishes and allowed to dehydrate slowly. Periodically, subsamples of pollen were taken and placed in liquid germination medium, and allowed to germinate. The liquid medium used for germination was essentially that described by Walden (1994), except that the water potential of the medium was adjusted by adding a larger amount (222 g l−1) of sucrose (Aylor, 2003). The pollen remaining in the sample dishes after the subsamples for germination were taken was weighed to obtain a ‘fresh weight’ and then they were placed in a drying oven and weighed again to determine their dry weight (Aylor, 2003). A pollen grain was counted as germinated if the length of the germ tube was ≥ one diameter of the pollen grain (i.e. approximately 75 μm long).
Drying rates
The drying rate of teosinte pollen was determined by two methods (Aylor, 2003): (i) by repeated reweighing of a small amount of pollen (in a thin layer averaging 1–2 grains thick) on the bottom of an aluminium weighing dish that was held over saturated salt solutions between each weighing and (ii) by measuring the principal diameters of individual grains using a microscope (at ×100) at various times during their exposure to air-streams having a range of temperatures and humidity. The second method utilized a small, specially constructed, flow chamber attached to the stage of the microscope (for details see Aylor, 2003). The principal diameters of the grains were measured microscopically at frequent intervals and were recorded. Changes in these diameters were converted to changes in mass of individual pollen grains by converting them to volume changes (using the volume–equivalent diameter, De) and assuming that volume changes were directly a result of water loss. The change in size and shape of five to eight pollen grains was observed for each determination at a particular vapour-pressure deficit (VPD). The experiment was repeated 11 times for VPDs ranging from 0.6 to 1.9 kPa. These data were used to derive pollen ‘wall’ conductance values, described in the next section.
Calculated pollen wall conductance
The driving force for water loss from a pollen grain is the vapour pressure difference between an evaporating surface within the pollen wall and the ambient air outside the grain. Following the arguments in Aylor (2003), we have conceptually combined the conductance of the plasma membrane, the inner and outer wall layers, and the aperture area into a single conductance, which is referred to in this paper as the conductance of the pollen wall.
It can be shown (Aylor, 2003) that the conductance of the pollen wall is much less than the conductance of the surrounding boundary layer, so that gv in Equation 2a can be replaced with gw, where gw is the conductance of the wall. An estimate was obtained for the conductance of the pollen wall following the method described in Aylor (2003). It is reasonable to assume that gv(≃gw), Ap, and hr are all approximately constant (i.e. do not depend on 𝛉) over a limited range of 𝛉 near full hydration. In this case, Equation 2 indicates that 𝛉 decreases approximately linearly with time for a limited range of water content, near full hydration of the pollen grain. Estimated values of gv were obtained from Equation 2 by expressing the change in mass of water in the grain in finite difference form, i.e. as md Δ𝛉/Δt, where the change in mass was obtained from the change in size of individual grains.
Results
Size and settling speed
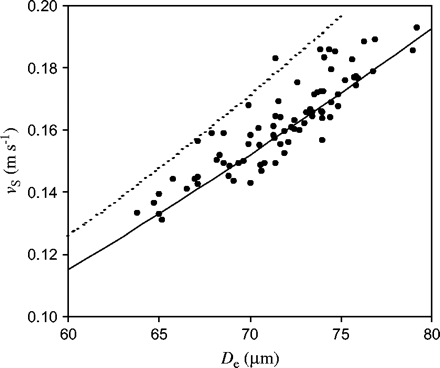
The mean volume-equivalent diameter (De) of the teosinte pollen used in these experiments was 72±3 μm (median=71 μm, range 63–80 μm). The average settling speed, vS, was 0.165 (±0.018) m s−1 (median=0.163, range 0.131–0.216 m s−1) (Fig. 1).
Settling speed in still air, vS (m s−1), versus volume-equivalent diameter, De (μm), for teosinte pollen. Shown are the measured values (solid circles) and the theoretical curve given by Equation 1 (solid line; r2=0.993, n=86). The well-known Stokes law result (dashed line), shown for comparison, gives values that are about 10% too high.
Water potential and germination percentage versus RWC
The water potential, ψ (MPa), of teosinte pollen decreases relatively slowly with changes in relative water content, 𝛉 (dimensionless), for values of 𝛉 >0.5, and it decreases more and more rapidly as 𝛉 approaches zero (Fig. 2A). The relationship between ψ and 𝛉 can be expressed by an equation of the form ψ= −4.13 𝛉−1.225 (r2=0.77, df=63).
(A) Water potential, ψ (MPa), versus relative water content, 𝛉 (dimensionless), for teosinte pollen. Measured values are shown by solid circles and open diamonds. The fitted line is given by ψ= −4.13 𝛉−1.225 (r2=0.77, df=63). (B) Germination, G (%), versus 𝛉 for teosinte pollen (solid circles). The fitted line is a non-linear fit of the data to a Weibull distribution given by
Pollen germination percentage remained high for values of 𝛉 >0.3, but decreased rapidly for lower values of 𝛉 (Fig. 2B). The data over the entire range of 𝛉 were described well by a cumulative Weibull distribution given by
Conductance of pollen wall
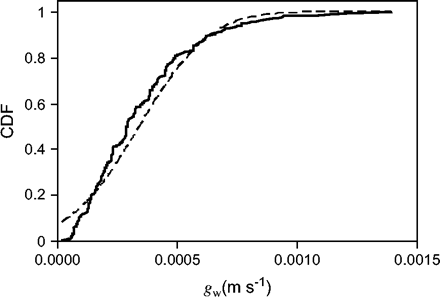
Values for the wall conductance, gw, of teosinte pollen were determined by measuring changes in size of individual grains. The mean value of gw (Fig. 3) was 3.42±0.13 (sem)×10−4 m s−1 (2.91×10−4 median, range 0.2–13.9×10−4 m s−1, n=297), determined by measuring the change in sizes of individual grains exposed to a range of vapour pressure deficits from 0.7 kPa to 1.9 kPa.
Cumulative distribution function (CDF) of pollen wall conductance values, gw (m s−1), for teosinte pollen (solid line). The mean, median, and standard deviation of gw are 3.42, 2.91, and 2.32×10−4 m s−1 (n=297), respectively. The Gaussian CDF (dashed line) for the same mean and standard deviation is shown for comparison.
Drying rate of pollen
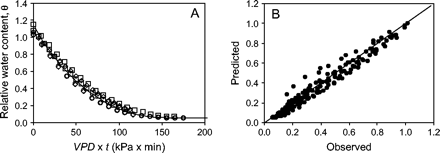
The change in relative water content, 𝛉, of teosinte pollen was strongly dependent on RH. When exposure was expressed as a function of VPD×time (Fig. 4A), then most data fell onto a single curve, confirming the physical dependence on VPD, given by Equation 2. The model results followed closely the shape of the pollen drying curves (Fig. 4A) and explained about 96% of the variation in the data (Fig. 4B). There was no overall trend in the variances over the entire range of predicted 𝛉, however, there was a tendency for the variances to be larger near mid-range values than at either end of the range. Differences between predicted and observed values, tested using a non-parametric Kolmogorov–Smirnov test, were not significantly different (P=0.11).
(A) Decrease in the relative water content, 𝛉, of teosinte pollen plotted as a function of vapour pressure deficit (VPD)×time. Symbols represent successive values of 𝛉 for pollen exposed to air at a temperature of 22.5 °C and RH values of 28% (squares), 33% (circles), and 54% (diamonds). The solid line is calculated using Equation 2 for RH=54%. (B) Predicted versus observed values of 𝛉 during the time-course of teosinte pollen drying in various T and RH conditions were highly correlated (r=0.98, n=180, P <10−7). The solid line is the 1:1 line.
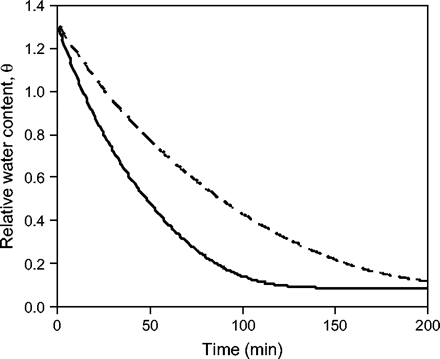
The model was used to compare relative drying rates for maize and teosinte pollen. The model indicates that teosinte pollen dries about 40% faster than hybrid maize pollen (Fig. 5). The difference in the drying rates for teosinte and maize pollen largely reflects the difference in the grouping gwAP/md in the model (Equation 2) for these two kinds of pollen. For these calculations, the wall conductance, gw, was set equal to the median values, i.e. 2.91×10−4 m s−1 for teosinte and 2.19×10−4 m s−1 for maize pollen. The curves level out as they approach the equilibrium values of water content, which are obtained by taking the inverse of Equation 2b and then using the parameter values given in this section. Equilibrium water contents of teosinte pollen (at 23.5 °C) for RH=20, 33, 54, and 75% were about 5.3, 6.9, 10.7, and 18.8%, respectively.
Relative water content, 𝛉, versus time (calculated by Equation 2) for teosinte pollen (solid line) and for corn pollen (dashed line) exposed to a VPD=1.3 kPa (T=23.1 °C, RH=54%). The parameter values used in the calculation for corn pollen were for a commercial hybrid and were taken from Aylor (2003).
Discussion
Pollen movement studies in the environment in the genus Zea have been conducted with maize pollen (Luna et al., 2001). However, very little is known about teosinte pollen mobility in the atmosphere. As far as we are aware, this study is the first to determine physical characteristics of teosinte pollen, which can contribute to defining the main parameters involved in gene flow in the genus Zea as a whole.
It is well known that pollen viability in the genus Zea is sensitive to water loss (Barnabas, 1985; Buitink et al., 1996; Luna et al., 2001); however, few quantitative measures for evaluating this sensitivity are available. Water loss from pollen is a complex process and involves water movement across a membrane and two wall layers. It is difficult to separate the effects on water movement of these various layers in intact pollen. At present, it is not possible to quantify the wall conductance in terms of its component parts, although recent experiments suggest that the exine may not be the main factor in limiting water loss, at least from pine pollen (Bohne et al., 2003). In this paper, the effects of these various layers were combined into an overall wall conductance and then it was used to quantify the rate of water loss from teosinte pollen. The distribution of wall conductance found was skewed toward higher values (Fig. 3), resulting in the mean being considerably higher than the median. It was assumed that the median pollen wall conductance (∼2.91×10−4 m s−1) is most representative of the population and it was then used to evaluate the rate of water loss from teosinte pollen grains. However, it is important to remember that using a single number for the wall conductance represents only part of the story and that models for gene flow in the environment will also need to account for the variation in conductance among members of the population of pollen grains (Fig. 3).
In this study, germination was used as a measure of pollen viability. This measure may tend to overestimate viability because not all of the pollen grains that germinate will necessarily be able to effect fertilization. Nevertheless, the ability of the pollen to recover and germinate in our in vitro test after drying to a particular water content should give a useful indication of how drying might ultimately effect potential fertilization.
Previous studies have focused on the biology of teosinte pollen (Baltazar and Schoper, 2001). Studies in Mexico have shown that teosinte pollen is about 30% smaller than maize pollen (Baltazar and Schoper, 2001; Baltazar et al., 2005). The smaller size of teosinte pollen suggests that it could travel longer distances than maize pollen from the pollen source, increasing the possibility of outcrossing at longer distances. These studies have also shown that teosinte pollen is 30–50% more susceptible to desiccation after being exposed to environmental conditions, which would tend to lessen the likelihood of outcrossing.
In this study, it was found that teosinte pollen settles at a rate that is about 40% slower than found for hybrid maize pollen (Di-Giovanni et al., 1995; Aylor, 2002) and dries about 40% faster than maize pollen (Table 1). This faster drying rate should result in about a 40% shorter survival time for teosinte pollen compared with hybrid maize pollen (Fig. 2B; Aylor, 2004). In view of these differences in physical properties of teosinte and maize pollen several factors related to their dispersal can be considered. Because of the smaller settling speed of teosinte pollen it is expected that it would move farther than maize pollen from the pollen source before being deposited. On a per plant basis, teosinte produces more pollen than landraces and commercial hybrid maize (Baltazar et al., 2005), which also contributes to its ability to pollinate at a distance. The effectiveness of the more prolific and farther-moving pollen might be partially or completely offset by the shorter pollen longevity of teosinte in the atmosphere. The net effect of these dynamics will depend on several environmental factors such as wind speed and turbulence and the vapour pressure deficit of the air. The model equations for settling speed and dehydration rate presented in Equations 1 and 2, respectively, offer a means for evaluating dispersal potential for a range of environmental conditions.
Comparison of mean values of volume equivalent diameter (De), settling speed (vS), and pollen wall conductance to water vapour (gw) for teosinte (Zea mays subsp. parviglumis) and maize (Zea mays L.) pollen
Property (units) . | Teosinte . | Maizea . |
|---|---|---|
| De (μm) | 72 | 91b |
| vS (m s−1) | 0.17 | 0.26b |
| gw (m s−1) | ||
| Mean | 3.42×10−4 | 2.65×10−4c |
| Median | 2.91×10−4 | 2.19×10−4c |
| ψ (MPa) | ||
| Parameter a1 | 4.13 | 3.22c |
| Parameter b1 | 1.23 | 1.35c |
| G (%) | ||
| Parameter c1 | 62 | 65c |
| Parameter d1 | 0.23 | 0.29c |
| Parameter e1 | 1.70 | 1.92c |
Property (units) . | Teosinte . | Maizea . |
|---|---|---|
| De (μm) | 72 | 91b |
| vS (m s−1) | 0.17 | 0.26b |
| gw (m s−1) | ||
| Mean | 3.42×10−4 | 2.65×10−4c |
| Median | 2.91×10−4 | 2.19×10−4c |
| ψ (MPa) | ||
| Parameter a1 | 4.13 | 3.22c |
| Parameter b1 | 1.23 | 1.35c |
| G (%) | ||
| Parameter c1 | 62 | 65c |
| Parameter d1 | 0.23 | 0.29c |
| Parameter e1 | 1.70 | 1.92c |
The coefficients in the expressions for water potential (ψ) versus relative water content (𝛉),
Hybrid maize (variety 37M81, Pioneer Hi-Bred International, Johnston, IA).
Data from Aylor (2002).
Data from Aylor (2003).
Comparison of mean values of volume equivalent diameter (De), settling speed (vS), and pollen wall conductance to water vapour (gw) for teosinte (Zea mays subsp. parviglumis) and maize (Zea mays L.) pollen
Property (units) . | Teosinte . | Maizea . |
|---|---|---|
| De (μm) | 72 | 91b |
| vS (m s−1) | 0.17 | 0.26b |
| gw (m s−1) | ||
| Mean | 3.42×10−4 | 2.65×10−4c |
| Median | 2.91×10−4 | 2.19×10−4c |
| ψ (MPa) | ||
| Parameter a1 | 4.13 | 3.22c |
| Parameter b1 | 1.23 | 1.35c |
| G (%) | ||
| Parameter c1 | 62 | 65c |
| Parameter d1 | 0.23 | 0.29c |
| Parameter e1 | 1.70 | 1.92c |
Property (units) . | Teosinte . | Maizea . |
|---|---|---|
| De (μm) | 72 | 91b |
| vS (m s−1) | 0.17 | 0.26b |
| gw (m s−1) | ||
| Mean | 3.42×10−4 | 2.65×10−4c |
| Median | 2.91×10−4 | 2.19×10−4c |
| ψ (MPa) | ||
| Parameter a1 | 4.13 | 3.22c |
| Parameter b1 | 1.23 | 1.35c |
| G (%) | ||
| Parameter c1 | 62 | 65c |
| Parameter d1 | 0.23 | 0.29c |
| Parameter e1 | 1.70 | 1.92c |
The coefficients in the expressions for water potential (ψ) versus relative water content (𝛉),
Hybrid maize (variety 37M81, Pioneer Hi-Bred International, Johnston, IA).
Data from Aylor (2002).
Data from Aylor (2003).
The genetic exchange between maize and teosinte is dependent on many factors, of which pollen movement is but one. Other factors include (i) the spatial isolation of the two species, (ii) the differences in flowering time of the two species, and (iii) the fitness of the hybrids combined with the types of selection operating in the teosinte populations. Wilkes (1977) and Sánchez et al. (1998) made a thorough review of teosinte distribution and characterization in Mexico. They described dates of planting across the Mexican Republic, and determined the probability of outcrossing with maize due to synchronization of flowering. Typically, the growing season for teosinte in Mexico is June through November. Seeds germinate with the beginning of the summer rains and growth parallels but plant development in teosinte tends to be somewhat later than for the local cultivated maize. Nonetheless, teosinte sheds pollen for about a month, and the earliest shedding teosinte plants tend to overlap flowering in maize at the end of their silking period, which allows for the possibility of maize forming fertile hybrids between maize and teosinte.
The physical properties of teosinte pollen determined in this study are important components of a model for pollen transport in the atmosphere and will aid in evaluating one of the main factors influencing gene flow in the genus Zea in Mexico and Central America. Likewise, it will contribute to our basic knowledge, required as part of any estimation of the potential impact of introducing transgenic maize into maize's centre of origin.
We thank P Thiel for excellent technical assistance, RJ Salvador for supplying the teosinte seed, and N Schultes and M Boehm for reviewing the manuscript. This material is based upon work supported in part by Hatch Funds and by the Cooperative State Research, Education, and Extension Service, U. S. Department of Agriculture, under Agreement No. 2004–33522–15044.
References
Aylor DE.
Aylor DE.
Aylor DE, Schultes NP, Shields EJ.
Baltazar BM, Sánchez GJJ, de la Cruz-Larios L, Schoper JB.
Baltazar BM, Schoper JB.
Baltazar BM, Schoper JB.
Barnabas B.
Bellon MR, Risopoulos J.
Bohne G, Richter E, Woehlecke H, Ehwald R.
Buitink J, Walters-Vertucci C, Hoekstra FA, Leprince O.
Campbell GS, Norman JM.
Casas SJF, Sánchez GJJ, Ramírez DJL, Ron PyS, Montes HJ.
Castillo GF, Goodman MM.
Cervantes MJE.
Cohen JI, Galinat WC.
Di-Giovanni F, Kevan PG.
Di-Giovanni F, Kevan PG, Nasr ME.
Luna VS, Figueroa JM, Baltazar BM, Gomez RL, Townsend R, Schoper JB.
Mangelsdorf PC.
Matsuoka Y, Vigouroux Y, Goodman MM, Sánchez GJJ, Buckler E, Doebley J.
Sánchez GJJ, Kato YTA, Aguilar MSM, Hernandez CJM, Lopez CAR, Ruiz JAC.
Sánchez GJJ, Stuber CW, Goodman MM.
Walden DB.
Wellhausen EJ, Roberts LM, Hernández EX (in collaboration with Mangelsdorf PC).



![(A) Water potential, ψ (MPa), versus relative water content, 𝛉 (dimensionless), for teosinte pollen. Measured values are shown by solid circles and open diamonds. The fitted line is given by ψ= −4.13 𝛉−1.225 (r2=0.77, df=63). (B) Germination, G (%), versus 𝛉 for teosinte pollen (solid circles). The fitted line is a non-linear fit of the data to a Weibull distribution given by \batchmode \documentclass[fleqn,10pt,legalpaper]{article} \usepackage{amssymb} \usepackage{amsfonts} \usepackage{amsmath} \pagestyle{empty} \begin{document} \(G(\%){=}62[1{-}\mathrm{exp}({-}((\mathrm{{\theta}}{-}0.05)/0.23)^{1.7})]\) \end{document}(r2=0.92, df=53).](https://oup.silverchair-cdn.com/oup/backfile/Content_public/Journal/jxb/56/419/10.1093/jxb/eri232/2/m_jexboteri232f02_lw.gif?Expires=1716465000&Signature=gKXPayPoap7pLDvFiIwOxAKbH--WRUTYTE8txKSYxOvwVL--zX6u3~aoeBYdEQWLI9rxhhO560VWIxXOm0dn~mwJudEX~RwECyRPqHJ6OjzU7N6kxFX-mNKix5TgH~r4XK0hnwTdv1aFrcYzEQ84KXV1O0Fyo9oduJddGELGGH~K-xbsS1VmznrVsE0SNtG6HmKRrtfY~61P2Epf19v-y2MdgRpdhkc1~FpUY1yWJeKk-nfTaKzigqAPboKCQKmZ2aHAmUMCi49H5LimPjajd~RvTdjg8rGW9q9js6iE5bQqcb64GKdkLJbfA1RBUsPC~5-u2ELvdhdlqyTHg8Mwbg__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)

Comments