-
PDF
- Split View
-
Views
-
Cite
Cite
Elisa N. Shoff, M. Elizabeth Zaney, Joseph H. Kahl, George W. Hime, Diane M. Boland, Qualitative Identification of Fentanyl Analogs and Other Opioids in Postmortem Cases by UHPLC-Ion Trap-MSn, Journal of Analytical Toxicology, Volume 41, Issue 6, July-August 2017, Pages 484–492, https://doi.org/10.1093/jat/bkx041
Close - Share Icon Share
Abstract
Since 2013, the Miami-Dade County Medical Examiner Department has experienced an increase in the number of opioid-related deaths. The majority of cases coincided with the introduction of fentanyl into the local heroin supply. From 2014 to 2015, Miami-Dade County experienced a near 600% increase in fentanyl-related deaths, followed by an additional 200% increase in 2016. In 2015, two novel fentanyl analogs were identified in medical examiner cases: beta-hydroxythiofentanyl and acetyl fentanyl. In 2016, four additional fentanyl analogs emerged: para-fluoroisobutyryl fentanyl, butyryl fentanyl, furanyl fentanyl and carfentanil, as well as the synthetic opioid U-47700. In order to address this epidemic, a method was developed and validated to identify 44 opioid-related and analgesic compounds in postmortem samples using ultra high performance liquid chromatography ion trap mass spectrometry with MSn capabilities. The limit of detection for all compounds ranged from 0.1 to 5 ng/mL, with a majority having MS3 spectral fragmentation. Blood, urine, liver or brain specimens from ~500 postmortem cases were submitted for analysis based on case history and/or initial screening results. Of those cases, 375 were positive for illicit fentanyl and/or one or more fentanyl analogs. Due to the potency of these compounds, they were almost always included in the cause of death. Worth emphasizing and extremely alarming is the detection of carfentanil in 134 cases, 104 of which were initially missed by gas chromatography mass spectrometry. By incorporating this sensitive, highly specific, and evolving screening procedure into the workflow, the toxicology laboratory continues to effectively assist the medical examiners in determining the cause and manner of death of decedents in Miami-Dade County.
Introduction
Since 2010, the Miami-Dade Medical Examiner Department (MDME) has observed a significant increase in the number of deaths involving opioids. Although the prevalence of opioids has remained constant in death investigations in South Florida, a slight increase in the number of overdose cases involving heroin and prescription fentanyl was noted between 2010 and 2013. During this same time period, the introduction of new laws regulating pain management clinics in the State of Florida resulted in a drastic reduction in the use and abuse of oxycodone, methadone, and other related analgesics (1). Although Florida's prosecution of illegal painkiller distribution drastically reduced the supply of oxycodone and other prescription analgesics, it created a void filled by heroin, a cheaper and more accessible alternative. As a result, and beginning in 2014, Miami-Dade County experienced a drastic increase in the number of deaths associated with illicit opioids, specifically heroin and fentanyl.
Fentanyl is a powerful synthetic opioid analgesic similar to morphine but 50–100 times more potent (2). It is classified under the United States (US) Controlled Substance Act as a Schedule II substance based on its accepted use in medical practice and high abuse potential. When prescribed appropriately, fentanyl relieves pain during anesthesia and treats moderate-to-severe chronic pain in certain patients (3). Although fentanyl still remains a prescribed analgesic, reports suggest that shipments from China have helped fuel illicit use of the drug in the United States (4). In Miami-Dade County, drug paraphernalia, recovered from terminal event scenes in drug overdose cases and analyzed by the MDME Toxicology Laboratory, indicated that illicit fentanyl was present in the community. Based on analytical testing, fentanyl either laced the heroin, or was mixed with heroin prior to use. The combination of heroin and fentanyl significantly increased the lethality of the mixture and deaths involving the combination became common.
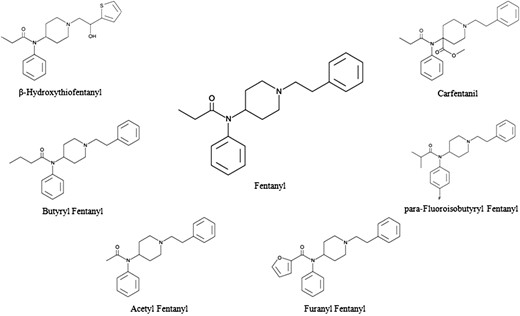
Analysis of a number of MDME cases in 2015 revealed two new fentanyl analogs: beta-hydroxythiofentanyl and acetyl fentanyl. In 2016, five new synthetic opioids emerged that drastically increased the number of accidental overdose deaths: U-47700, furanyl fentanyl, butyryl fentanyl, para-fluoroisobutyryl fentanyl and carfentanil. Figure 1 shows the structures of fentanyl and associated analogs. The presence of fentanyl analogs and other synthetic opioids in postmortem casework is alarming, as many have increased potency compared to fentanyl and exhibit similar pharmacological effects. Fentanyl causes dose-dependent central nervous system and respiratory depression through agonist activity at the μ-opioid receptor (2). Consequently, many fentanyl analogs exhibit enhanced effects at much lower dosages, thereby dramatically increasing the risk of fatal overdose.
Beta-hydroxythiofentanyl, N-[1-(2-hydroxy-2-thiophen-2-ylethyl)piperidin-4-yl]-N-phenylpropanamide, and butyryl fentanyl, N-phenyl-N-[1-(2-phenylethyl)piperidin-4-yl]butanamide, were placed into Schedule I of the US Controlled Substance Act in May 2016 following China's ban in October 2015. The scheduling of both analogs in the US occurred as a result of their prevalence in drug exhibits from laboratories across the country as well as in confirmed fatalities (5). Interestingly, the change in legal status of beta-hydroxythiofentanyl in China coincided with its disappearance in MDME cases; however, deaths attributed to butyryl fentanyl continued through the end of 2016.
Acetyl fentanyl, N-(1-phenethylpiperidin-4-yl)-N-phenylacetamide, is a US Schedule I controlled substance synthesized alongside fentanyl in the 1960s (6). According to the World Health Organization, acetyl fentanyl has been identified as an impurity in the production of fentanyl; however, it has also been identified in confiscated evidence trafficked illegally into the US (7). Studies indicate that acetyl fentanyl is 15 times more potent than morphine but one-third less potent than fentanyl (8). Despite acetyl fentanyl's decreased potency, its use as an abused substance is unequivocal and has been implicated in multiple deaths across the US and Europe (9–12).
U-47700, 3,4-dichloro-N-[2-(dimethylamino)cyclohexyl]-N-methylbenzamide, is a US Schedule I synthetic opioid analgesic originally developed in the 1970s as a more potent but less addictive alternative to morphine (13, 14). In animal models, U-47700 was demonstrated to have approximately eight times the potency of morphine (15). Although not considered an analog of fentanyl, its purpose was to treat severe pain associated with injury, cancer and surgery. Drug testing did not progress to human trials; therefore, U-47700 never became available as a pharmaceutical agent (14). Due to easy access of a patent detailing the synthesis of U-47700, the substance found its way into the illicit drug market. U-47700 alone, and in combination with fentanyl, has been attributed to several deaths in the United States and Europe (16–18).
Furanyl fentanyl, N-phenyl-N-[1-(2-phenylethyl)piperidin-4-yl]furan-2-carboxamide, similar to U-47700, was designated as a US Schedule I substance in November 2016 (19). It was originally synthesized and patented in 1986; (20) however, it was never studied in humans. Animal studies suggest that furanyl fentanyl is equipotent to fentanyl (20). A series of furanyl fentanyl-related deaths were reported in Sweden over a 4-month period, mostly in combination with other drugs (21, 22). Measured concentrations of furanyl fentanyl in these deaths varied over a wide concentration range; therefore, specific lethal concentrations are not well-defined (22).
Para-fluoroisobutyryl fentanyl, N-(4-fluorophenyl)-2-methyl-N-[1-(2-phenylethyl)-4-piperidinyl]-propanamide, is a synthetic fentanyl analog with very limited information regarding its availability and potency. Users on various internet drug forums, such as Bluelight and Reddit, report similar effects and potency to furanyl fentanyl. Although there have been no peer-reviewed articles published regarding para-fluoroisobutyryl fentanyl toxicity, Miami-Dade County has reported several overdoses attributed solely to the analog, indicating its comparable or increased potency to fentanyl.
Carfentanil, a US Schedule II substance, was originally synthesized in the 1970s under the name Wildnil® and marketed as a general anesthetic for large animals (23, 24). Approximately 10,000 times more potent than morphine and 100 times more potent than fentanyl, carfentanil was never intended for human use (25). In 2002, over 120 unintended hostage deaths associated with carfentanil ingestion occurred in Moscow when a Special Forces Unit belonging to the Russian Federal Security Service pumped an aerosolized mixture of it into a sold-out theater during a terrorist attack, unaware of its extreme potency and deadly outcome (26). Like many of the new synthetic opioids, carfentanil has found its way into the illicit drug supply via synthesis in clandestine laboratories and trafficking into the United States (27). Carfentanil has contributed immensely to the nationwide opioid epidemic with reports of accidental overdoses surfacing from coast to coast (28). Miami-Dade County has been no stranger to this epidemic with an alarming number of accidental carfentanil overdoses occurring within a 6-month period.
To address the increase in the number of deaths involving fentanyl, and particularly the fentanyl analogs, the MDME Toxicology Laboratory developed and validated a comprehensive and sensitive screening method using ultra high performance liquid chromatography ion trap mass spectrometry with MSn capabilities (UPHLC-Ion Trap-MSn) to identify illicit substances as well as other commonly detected prescription analgesics in postmortem biological fluids and tissues. Traditional or routine laboratory approaches to drug screening may fail to detect synthetic opioids due to little or no cross-reactivity on immunoassay tests and/or inability to detect low drug concentrations using gas chromatography mass spectrometry (GC–MS). The use of ion trap technology as a robust screening method is unique to postmortem casework. Initially, the laboratory incorporated a previously published method using UPHLC-Ion Trap-MSn as a comprehensive screening tool for ~900 compounds in postmortem blood and tissue (29). Due to the lack of specificity and selectivity of the original method, several targeted panels were developed and optimized by reducing the number of analytes and adding novel substances to the existing library. This allowed for greater sensitivity and the ability to detect low level substances in complex postmortem matrices, while at the same time providing full scan and MSn spectral data for identification.
Experimental
Chemicals and reagents
Carfentanil, butyryl fentanyl, para-fluoroisobutyryl fentanyl, beta-hydroxythiofentanyl, furanyl fentanyl and para-fluorobutyryl fentanyl were purchased from Cayman Chemical (Ann Arbor, Michigan). Acetyl fentanyl was purchased from Lipomed (Cambridge, Massachusetts). The remaining analytes were purchased from Cerilliant (Round Rock, Texas). LCMS grade water and acetonitrile (ACN), methanol, ethyl acetate, ammonium hydroxide, ammonium formate, sodium phosphate dibasic heptahydrate, sodium phosphate monobasic monohydrate, and formic acid were purchased from Fisher Scientific (Hanover Park, Illinois). Dichloromethane and isopropanol were purchased from Honeywell (Morris Plains, New Jersey), hexane was purchased from OmniSolv (McLean, Virginia), and glacial acetic acid was purchased from Macron Fine Chemicals (Center Valley, Pennsylvania). All solvents and reagents were ACS grade or higher. Defibrinated sheep blood was purchased from Hemostat Laboratories (Dixon, California).
Standard and control preparation
A control stock solution was prepared containing fentanyl and oxycodone at a concentration of 100 ng/mL in ACN. Extracted controls at 5 ng/mL were achieved by the addition of 50 μL of the control stock solution into 1 mL aliquots of defibrinated sheep blood. Fentanyl and oxycodone were chosen as markers for extraction efficiency and instrument performance of the qualitative analytical procedure. An internal standard working stock solution of fentanyl-d5 was prepared at a concentration of 100 ng/mL in ACN, 50 μL of which was added to all samples for a final concentration of 5 ng/mL.
Sample preparation and extraction
Whole blood and urine samples (1 mL) and/or tissue homogenates (1 g) were buffered with 0.1 M, pH 6 sodium phosphate (4 mL). Brain and liver tissue homogenates were prepared by diluting with deionized (DI) water (1:1 and 1:4, respectively) and blended. All samples were vortexed, centrifuged for 10 min at 2,300 × g, and extracted by solid phase extraction (SPE) utilizing United Chemical Technologies (UCT) Clean Screen® mixed mode extraction columns (part no. ZSDAU020) coupled with a UCT Positive Pressure Manifold (Bristol, Pennsylvania). Prior to sample introduction, columns were conditioned with methanol (3 mL), DI water (3 mL), and 0.1 M, pH 6 sodium phosphate buffer (1 mL). Samples were added to the columns and subsequently rinsed with DI water (3 mL) and 1 M acetic acid (1 mL) and dried for 5 min at 120 psi. Columns were further rinsed with hexane (2 mL), hexane:ethyl acetate (50:50, 3 mL), and methanol (3 mL) and dried for an additional 3 min at 120 psi. Analytes were eluted with dichloromethane:isopropanol:ammonium hydroxide (78:20:2, 3 mL). The eluents were evaporated to dryness at 40°C with N2 and the residue was reconstituted with 50 μL ACN.
A negative and positive control sample were extracted from whole blood and analyzed prior to every batch of case samples to ensure proper instrument performance (e.g., retention time shifts, column efficiency and signal response). Extracted positive controls were analyzed every 10 case samples for quality control and met the following identification criteria: visual signal-to-noise assessment, full spectral fragmentation, consistency in target precursor ion, and a library match score ≥80% based upon spectral purity, reverse-match and forward-match.
Instrumentation
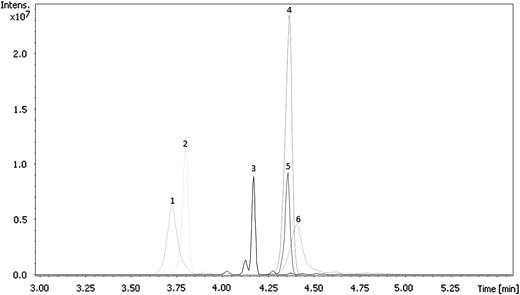
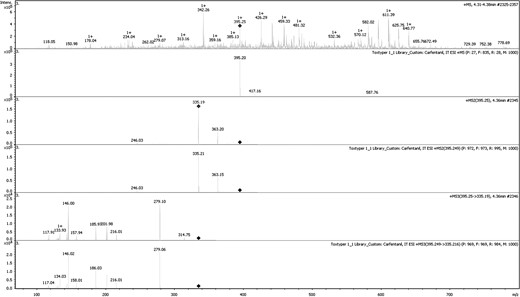
Analysis was conducted using a Thermo Scientific Dionex Ultimate 3000 RSLC ultra high-performance liquid chromatograph (Idstein, Germany) coupled to a Bruker Daltonics AmaZon Speed™ ion trap mass spectrometer (Bremen, Germany). The instrument was equipped with Toxtyper™ software, which allows for data-dependent targeted MSn analysis. Chromatographic separation was achieved using a Thermo Scientific Acclaim® RSLC 120 C18 column (2.1 × 100 mm2, 2.2 μm, 120 A) with an aqueous mobile phase of 2 mM ammonium formate, 0.1% formic acid, and 1% acetonitrile in water (A), and an organic mobile phase of 2 mM ammonium formate, 0.1% formic acid, and 1% water in acetonitrile (B). A 5 μL sample volume was injected with a flow rate of 500 μL/min using a gradient elution. Initial conditions of 99:1 (A:B) were adjusted linearly during the first 8 min. At 8 min, a composition of 1:99 (A:B) is reached. From 8 to 11 min, the system returns to equilibrium. During the 11-min run time, all compounds of interest eluted within the first 8 min, while the remaining 3 min of the run was diverted to waste. Figure 2 illustrates chromatographic separation of the 6 fentanyl analogs detected in MDME case samples. The ion trap was operated using electrospray ionization (ESI) with zero delay alternating polarity. Data were collected using a scan range of 70–800 m/z in full-scan, autoMSn mode, at a speed of 32,500 amu/s. Acquisition was achieved by using a data-dependent Scheduled Precursor List (SPL) which contains 44 targeted compounds with the following information: known retention time, retention time tolerance (+/−0.40 min), polarity (positive or negative), and unique precursor ions used for MS2, and if applicable, MS3 fragmentation. Out of the 44 targeted compounds in the method, 27 have MS3 spectral profiles. Figure 3 demonstrates a full MS3 spectral fragmentation of carfentanil at an extracted concentration of 0.1 ng/mL from whole blood. Fragmentation of carfentanil was achieved by first targeting the parent ion of 395.20 to produce a MS2 spectra, including a main daughter ion of 335.2, which was then targeted to produce the final MS3 breakdown. For proper identification of the compounds, an in-house spectral library and a pre-installed Toxtyper™ library were utilized. The pre-installed Toxtyper™ library contained only three fentanyl analogs, none of which were identified in casework. In order to address the needs of the laboratory, the in-house spectral library was created with the addition of 14 fentanyl analogs and synthetic opioids using certified reference standards. This in-house spectral library continues to evolve as new compounds emerge and certified reference standards become available.
Extracted MS2 ion chromatograms of (1) acetyl fentanyl, (2) beta-hydroxythiofentanyl, (3) furanyl fentanyl, (4) carfentanil, (5) butyryl fentanyl and (6) para-fluoroisobutyryl fentanyl.
Full-scan MS, MS2 and MS3 spectral fragmentation of carfentanil at an extracted concentration of 0.1 ng/mL with associated in-house library match.
Method validation
A method was developed to target 44 different opioid related compounds. Of the 44 targeted compounds, 26 were validated for limit of detection (LOD), stability, selectivity, specificity, matrix effects, ionization suppression/enhancement (ISE), and carryover based on the availability of certified reference materials and necessity. Stock solutions of these analytes were prepared at concentrations of 1000 ng/mL, 100 ng/mL, 10 ng/mL and 1 ng/mL in ACN, and were refrigerated in amber vials.
Limit of detection
To evaluate LOD, replicates of each compound at varying concentrations (0.01, 0.05, 0.1, 0.2, 0.5, 1.0 and 5.0 ng/mL) were extracted from whole blood in triplicate on three different days. LODs were established using the following criteria: visual signal-to-noise assessment, full spectral fragmentation, consistency in target precursor ion, and a library match score ≥80% based upon spectral purity, reverse-match, and forward-match. For all 26 compounds, LODs ranged from 0.1 ng/mL to 5 ng/mL. All compounds, including those without established LODs, are outlined in Table I.
Limits of detection and targeted precursor ions for all analytes
| Name . | LOD (ng/mL) . | Precursor (MS2)a . | Precursor (MS3)b . |
|---|---|---|---|
| 3-Methylfentanyl | – | 351.19 | – |
| 6-O-acetylmorphine | 0.5 | 328.08 | – |
| Acetaminophen | – | 151.91 | 110 |
| Acetyl fentanyl | 0.2 | 323 | 188 |
| Acetylsalicyclic acid | – | 178.92 | 137 |
| Alfentanil | – | 417.15 | – |
| Beta-hydroxythiofentanyl | 0.1 | 359.22 | 341 |
| Buprenorphine | 0.5 | 468.27 | – |
| Butyryl fentanyl | 0.2 | 351 | 188 |
| Carfentanil | 0.1 | 395.15 | 335.2 |
| Codeine | 1 | 300.04 | – |
| Desproprionyl fentanyl | – | 281 | 188 |
| Dihydrocodone | – | 302.08 | – |
| EDDP | 0.5 | 278.03 | – |
| Fentanyl | 0.5 | 337.16 | – |
| Furanyl fentanyl | 0.5 | 375 | 188 |
| Heroin | 0.5 | 370.14 | – |
| Hydrocodone | 0.5 | 300.08 | – |
| Hydromorphone | 0.5 | 285.99 | – |
| Ibuprofen | – | 206.95 | 207 |
| Meperidine | 0.5 | 248.02 | – |
| Methadone | 0.5 | 310.1 | 265 |
| Mitragynine | – | 399.19 | 238 |
| Morphine | 5 | 286 | – |
| Naloxone | 0.5 | 328.1 | 310 |
| Naltrexone | 0.5 | 342.04 | 324 |
| Naproxen | – | 230.89 | 184.8 |
| N-desmethyltramadol | – | 250 | 232 |
| Norbuprenorphine | 1 | 414.4 | – |
| Norcodeine | – | 286.03 | – |
| Norfentanyl | 0.5 | 233.07 | – |
| Normorphine | – | 271.96 | – |
| Noroxycodone | – | 302.12 | 284 |
| Noscapine | – | 414 | 220 |
| O-desmethyltramadol | – | 250 | 232 |
| Oxycodone | 0.5 | 316.1 | 298 |
| Oxymorphone | 0.5 | 301.97 | 284 |
| Para-fluorobutyryl fentanyl | 0.5 | 369 | 188 |
| Para-fluoroisobutyryl fentanyl | 0.5 | 369 | 188 |
| Salicylic acid | – | 136.96 | 93.2 |
| Sufentanil | – | 387.1 | – |
| Tramadol | 5 | 264 | 246 |
| U-47700 | 0.5 | 329 | 284 |
| W-18 | – | 422 | 273 |
| Name . | LOD (ng/mL) . | Precursor (MS2)a . | Precursor (MS3)b . |
|---|---|---|---|
| 3-Methylfentanyl | – | 351.19 | – |
| 6-O-acetylmorphine | 0.5 | 328.08 | – |
| Acetaminophen | – | 151.91 | 110 |
| Acetyl fentanyl | 0.2 | 323 | 188 |
| Acetylsalicyclic acid | – | 178.92 | 137 |
| Alfentanil | – | 417.15 | – |
| Beta-hydroxythiofentanyl | 0.1 | 359.22 | 341 |
| Buprenorphine | 0.5 | 468.27 | – |
| Butyryl fentanyl | 0.2 | 351 | 188 |
| Carfentanil | 0.1 | 395.15 | 335.2 |
| Codeine | 1 | 300.04 | – |
| Desproprionyl fentanyl | – | 281 | 188 |
| Dihydrocodone | – | 302.08 | – |
| EDDP | 0.5 | 278.03 | – |
| Fentanyl | 0.5 | 337.16 | – |
| Furanyl fentanyl | 0.5 | 375 | 188 |
| Heroin | 0.5 | 370.14 | – |
| Hydrocodone | 0.5 | 300.08 | – |
| Hydromorphone | 0.5 | 285.99 | – |
| Ibuprofen | – | 206.95 | 207 |
| Meperidine | 0.5 | 248.02 | – |
| Methadone | 0.5 | 310.1 | 265 |
| Mitragynine | – | 399.19 | 238 |
| Morphine | 5 | 286 | – |
| Naloxone | 0.5 | 328.1 | 310 |
| Naltrexone | 0.5 | 342.04 | 324 |
| Naproxen | – | 230.89 | 184.8 |
| N-desmethyltramadol | – | 250 | 232 |
| Norbuprenorphine | 1 | 414.4 | – |
| Norcodeine | – | 286.03 | – |
| Norfentanyl | 0.5 | 233.07 | – |
| Normorphine | – | 271.96 | – |
| Noroxycodone | – | 302.12 | 284 |
| Noscapine | – | 414 | 220 |
| O-desmethyltramadol | – | 250 | 232 |
| Oxycodone | 0.5 | 316.1 | 298 |
| Oxymorphone | 0.5 | 301.97 | 284 |
| Para-fluorobutyryl fentanyl | 0.5 | 369 | 188 |
| Para-fluoroisobutyryl fentanyl | 0.5 | 369 | 188 |
| Salicylic acid | – | 136.96 | 93.2 |
| Sufentanil | – | 387.1 | – |
| Tramadol | 5 | 264 | 246 |
| U-47700 | 0.5 | 329 | 284 |
| W-18 | – | 422 | 273 |
aParent Ion targeted for MS2.
bDaughter Ion targeted for MS3.
Limits of detection and targeted precursor ions for all analytes
| Name . | LOD (ng/mL) . | Precursor (MS2)a . | Precursor (MS3)b . |
|---|---|---|---|
| 3-Methylfentanyl | – | 351.19 | – |
| 6-O-acetylmorphine | 0.5 | 328.08 | – |
| Acetaminophen | – | 151.91 | 110 |
| Acetyl fentanyl | 0.2 | 323 | 188 |
| Acetylsalicyclic acid | – | 178.92 | 137 |
| Alfentanil | – | 417.15 | – |
| Beta-hydroxythiofentanyl | 0.1 | 359.22 | 341 |
| Buprenorphine | 0.5 | 468.27 | – |
| Butyryl fentanyl | 0.2 | 351 | 188 |
| Carfentanil | 0.1 | 395.15 | 335.2 |
| Codeine | 1 | 300.04 | – |
| Desproprionyl fentanyl | – | 281 | 188 |
| Dihydrocodone | – | 302.08 | – |
| EDDP | 0.5 | 278.03 | – |
| Fentanyl | 0.5 | 337.16 | – |
| Furanyl fentanyl | 0.5 | 375 | 188 |
| Heroin | 0.5 | 370.14 | – |
| Hydrocodone | 0.5 | 300.08 | – |
| Hydromorphone | 0.5 | 285.99 | – |
| Ibuprofen | – | 206.95 | 207 |
| Meperidine | 0.5 | 248.02 | – |
| Methadone | 0.5 | 310.1 | 265 |
| Mitragynine | – | 399.19 | 238 |
| Morphine | 5 | 286 | – |
| Naloxone | 0.5 | 328.1 | 310 |
| Naltrexone | 0.5 | 342.04 | 324 |
| Naproxen | – | 230.89 | 184.8 |
| N-desmethyltramadol | – | 250 | 232 |
| Norbuprenorphine | 1 | 414.4 | – |
| Norcodeine | – | 286.03 | – |
| Norfentanyl | 0.5 | 233.07 | – |
| Normorphine | – | 271.96 | – |
| Noroxycodone | – | 302.12 | 284 |
| Noscapine | – | 414 | 220 |
| O-desmethyltramadol | – | 250 | 232 |
| Oxycodone | 0.5 | 316.1 | 298 |
| Oxymorphone | 0.5 | 301.97 | 284 |
| Para-fluorobutyryl fentanyl | 0.5 | 369 | 188 |
| Para-fluoroisobutyryl fentanyl | 0.5 | 369 | 188 |
| Salicylic acid | – | 136.96 | 93.2 |
| Sufentanil | – | 387.1 | – |
| Tramadol | 5 | 264 | 246 |
| U-47700 | 0.5 | 329 | 284 |
| W-18 | – | 422 | 273 |
| Name . | LOD (ng/mL) . | Precursor (MS2)a . | Precursor (MS3)b . |
|---|---|---|---|
| 3-Methylfentanyl | – | 351.19 | – |
| 6-O-acetylmorphine | 0.5 | 328.08 | – |
| Acetaminophen | – | 151.91 | 110 |
| Acetyl fentanyl | 0.2 | 323 | 188 |
| Acetylsalicyclic acid | – | 178.92 | 137 |
| Alfentanil | – | 417.15 | – |
| Beta-hydroxythiofentanyl | 0.1 | 359.22 | 341 |
| Buprenorphine | 0.5 | 468.27 | – |
| Butyryl fentanyl | 0.2 | 351 | 188 |
| Carfentanil | 0.1 | 395.15 | 335.2 |
| Codeine | 1 | 300.04 | – |
| Desproprionyl fentanyl | – | 281 | 188 |
| Dihydrocodone | – | 302.08 | – |
| EDDP | 0.5 | 278.03 | – |
| Fentanyl | 0.5 | 337.16 | – |
| Furanyl fentanyl | 0.5 | 375 | 188 |
| Heroin | 0.5 | 370.14 | – |
| Hydrocodone | 0.5 | 300.08 | – |
| Hydromorphone | 0.5 | 285.99 | – |
| Ibuprofen | – | 206.95 | 207 |
| Meperidine | 0.5 | 248.02 | – |
| Methadone | 0.5 | 310.1 | 265 |
| Mitragynine | – | 399.19 | 238 |
| Morphine | 5 | 286 | – |
| Naloxone | 0.5 | 328.1 | 310 |
| Naltrexone | 0.5 | 342.04 | 324 |
| Naproxen | – | 230.89 | 184.8 |
| N-desmethyltramadol | – | 250 | 232 |
| Norbuprenorphine | 1 | 414.4 | – |
| Norcodeine | – | 286.03 | – |
| Norfentanyl | 0.5 | 233.07 | – |
| Normorphine | – | 271.96 | – |
| Noroxycodone | – | 302.12 | 284 |
| Noscapine | – | 414 | 220 |
| O-desmethyltramadol | – | 250 | 232 |
| Oxycodone | 0.5 | 316.1 | 298 |
| Oxymorphone | 0.5 | 301.97 | 284 |
| Para-fluorobutyryl fentanyl | 0.5 | 369 | 188 |
| Para-fluoroisobutyryl fentanyl | 0.5 | 369 | 188 |
| Salicylic acid | – | 136.96 | 93.2 |
| Sufentanil | – | 387.1 | – |
| Tramadol | 5 | 264 | 246 |
| U-47700 | 0.5 | 329 | 284 |
| W-18 | – | 422 | 273 |
aParent Ion targeted for MS2.
bDaughter Ion targeted for MS3.
Sample stability
Sample stability was evaluated in extracted case samples that were positive for targeted compounds. All samples upon initial analysis were re-injected both 24 and 48 h post-extraction. Samples remained stored in the refrigerated autosampler (8°C) for the duration of the experiment. Stability was assessed by observing a decrease in signal and/or detection of compounds. Over 48 h, all compounds were stable with minimal decrease in signal and were positively detected. Freeze–thaw and long-term storage studies were not performed as part of this validation due to the sample storage procedures (refrigeration only) of the laboratory and the recent emergence of many of these novel substances, respectfully.
Selectivity/specificity
All compounds with established LODs were evaluated for exogenous interferences using solutions containing a total of 80 non-targeted, commonly detected analytes. To assess selectivity and to mimic the presence of therapeutic and fatal concentrations, the 80 non-targeted analytes were added at a concentration of 500 ng/mL into whole blood containing 5 ng/mL of each targeted analyte. To assess specificity, the 80 non-targeted analytes were extracted from whole blood without the addition of the targeted analytes. No false positives were detected with any of the untargeted analytes; however, the presence of quetiapine at a concentration of 500 ng/mL suppressed the fentanyl signal at a concentration of 5 ng/mL but did not prevent its detection.
To evaluate endogenous interferences, three sets of decomposed samples (n = 15, 5 of each specimen type) that previously screened negative for targeted analytes (blood, brain homogenate and liver homogenate) were extracted to assess the potential for false positive results due to matrix interferences. No interferences were detected from any of the analyzed matrices resulting in a false positive identification.
Ionization suppression/enhancement
ISE was evaluated by a post-extraction addition approach for each analyte. Based upon the change in signal response, little to no significant ionization enhancement or suppression was noted in the decomposed blood samples; however, ionization suppression was noted with decomposed liver and brain homogenates (%ISE ≥50%). Despite this, the detection of the analytes was not affected.
Carryover
Neat mixtures of all compounds were prepared at a concentration of 1000 ng/mL for all opiate-related drugs, and 100 ng/mL for fentanyl and associated analogs. Each mixture was bracketed by solvent vials containing ACN. The solvents were evaluated for the presence of any compounds injected in the prior mixture; all solvents were negative.
Results and Discussion
Case results
Approximately 500 postmortem MDME cases were analyzed using the validated method previously described. These cases were tested primarily based on case history. Criteria included a history of intravenous drug abuse, presence of drug paraphernalia at the terminal event scene, eyewitness accounts and other suspicious deaths of otherwise healthy, young individuals. Cases were also assigned based on preliminary screening results by immunoassay and GC–MS (i.e., the presence of illicit substances such as cocaine, heroin and fentanyl). Of these cases, 375 were positive for illicit fentanyl and/or one or more fentanyl analogs. The fentanyl analogs were detected in 176 of these cases from 2015 to the end of 2016. Approximately 60% of all fentanyl and fentanyl analog cases were also positive for morphine/codeine/6-monoacetylmorphine (heroin) or cocaine, and 20% of all cases had evidence of both heroin and cocaine use. There were six different fentanyl analogs detected in MDME cases as well as the synthetic opioid U-47700. In 2015, beta-hydroxythiofentanyl was detected in nine cases and acetyl fentanyl was detected in 13 cases. Beginning in July 2016, carfentanil, furanyl fentanyl and para-fluoroisobutyryl fentanyl first emerged (134, 37 and 26 cases, respectively), followed by butyryl fentanyl and U-47700 (3 and 4 cases, respectively) towards the end of 2016. An interesting observation was that in 90% of the cases positive for furanyl fentanyl, the compound despropionyl fentanyl was also detected. To a much lesser extent, it was detected in blood specimens alongside other fentanyl analogs, as well as in drug paraphernalia recovered from the terminal event scene.
Out of the 375 cases positive for illicit fentanyl and/or one or more fentanyl analogs, 70% were white males, 16% were white females, 12% were black males, and black females comprised the remaining 2% of cases. Decedents ranged in age from 17 to 73 years with a mean of 38 years and a median of 36 years. Of note, the average age of white decedents was 10 years younger than black decedents.
Table II highlights 12 of the positive cases at MDME in which fentanyl analogs and U-47700 were detected using the UPHLC-Ion Trap-MSn method. All but one case lists a fentanyl analog in the official cause of death.
Representative sampling of cases analyzed including demographic information, case history, toxicology findings and cause and manner of death
| . | Demographic . | History . | Fentanyl analogs detected . | Additional reported compounds . | Cause and manner of death . |
|---|---|---|---|---|---|
| 1 | 28 W/M | Found unresponsive on the floor with a syringe next to him | Beta-hydroxythiofentanyl | Norfentanyl, ethanol 0.331% (blood), 0.352% (bile), metoclopramide, cannabinoids |
|
| 2 | 28 W/M | Found by his mother in his sister's bedroom with his face prone on the floor and a syringe in his underwear | Acetyl fentanyl | Fentanyl 14 ng/mL, cocaine <0.05 mg/L, benzoylecgonine 0.096 mg/L, methylecgonine, levamisole |
|
| 3 | 28 B/F | She was drinking alcoholic frozen drinks with her boyfriend when they both became nauseous, dizzy and vomited. They eventually went to sleep and the boyfriend later woke up and found her unresponsive. She was a known drug user | Para-fluoroisobutyryl fentanyl | Alprazolam 0.033 mg/L, cannabinoids, ibuprofen |
|
| 4 | 39 W/M | He lived in a homeless shelter and was found dead in bed. Known to use multiple drugs including heroin and cocaine | Furanyl fentanyl | Desproprionyl fentanyl, ethanol 0.301% (chest fluid), 0.268% (brain), morphine <0.01 mg/kg, codeine <0.01 mg/kg, hydrocodone <0.01 mg/kg, hydromorphone <0.01 mg/kg, trazodone, chlorphenyl piperazine |
|
| 5 | 34 W/F |
| Carfentanil and furanyl fentanyl | Despropionyl fentanyl, cocaine 0.011 mg/L, benzoylecgonine 0.124 mg/L, methylecgonidine, methylecgonine, levamisole, cannabinoids |
|
| 6 | 37 W/M | Found behind bushes decomposed | Carfentanil | Ethanol 0.089% (bile), 0.051% (brain), dextromethorphan, diphenhydramine, hydroxyzine, ibuprofen, meprobamate, quetiapine, sertraline, norsertraline, cannabinoids |
|
| 7 | 27 W/F | Prostitute found unresponsive in a field with drug paraphernalia littering the area | Carfentanil | Fentanyl 12 ng/mL, morphine 0.013 mg/L, codeine <0.01 mg/L, 6-monoacetylmorphine <0.001 mg/L, cocaine <0.01 mg/L, benzoylecgonine 0.125 mg/L, methylecgonine, chlorcyclizine, hydroxyzine |
|
| 8 | 23 W/M | Found unrespsonive in bathroom by his family with drug paraphernalia | Carfentanil and para-fluoroisobutyryl fentanyl | Ethanol 0.084% (blood), 0.107% (ocular), dextromethorphan | Acute combined drug toxicity (carfentanil, para-fluorobutyryl/para-fluoroisobutyryl fentanyl and ethanol) accident |
| 9 | 19 W/M |
| Carfentanil and furanyl fentanyl | Despropionyl fentanyl, morphine <0.01 mg/L, codeine <0.01 mg/L, cocaine <0.01 mg/L, benzoylecgonine 0.209 mg/L, methylecgonidine, methylecgonine, levamisole, 7-aminoclonazepam, ibuprofen, lidocaine, desethyllidocaine, naloxone, cannabinoids |
|
| 10 | 41 W/M | Found unresponsive in a field | Carfentanil and U-47700 | Fentanyl <1 ng/mL, morphine 0.032 mg/L, codeine <0.01 mg/L, cocaine <0.01 mg/L, methylecgonine, atropine, diphenhydramine, gabapentin, ibuprofen, naloxone, quetiapine, norquetiapine, quinine |
|
| 11 | 41 B/M |
| Para-fluoroisobutyryl fentanyl and butyryl fentanyl | Oxycodone 0.021 mg/L, noroxycodone, alprazolam 0.073 mg/L, dextromethorphan, promethazine, cannabinoids |
|
| 12 | 30 W/M | Found unresponsive on the street with a foamy discharge around his mouth | Carfentanil, para-fluoroisobutyryl fentanyl and U-47700 | Sertraline, norsertraline, diphenhydramine |
|
| . | Demographic . | History . | Fentanyl analogs detected . | Additional reported compounds . | Cause and manner of death . |
|---|---|---|---|---|---|
| 1 | 28 W/M | Found unresponsive on the floor with a syringe next to him | Beta-hydroxythiofentanyl | Norfentanyl, ethanol 0.331% (blood), 0.352% (bile), metoclopramide, cannabinoids |
|
| 2 | 28 W/M | Found by his mother in his sister's bedroom with his face prone on the floor and a syringe in his underwear | Acetyl fentanyl | Fentanyl 14 ng/mL, cocaine <0.05 mg/L, benzoylecgonine 0.096 mg/L, methylecgonine, levamisole |
|
| 3 | 28 B/F | She was drinking alcoholic frozen drinks with her boyfriend when they both became nauseous, dizzy and vomited. They eventually went to sleep and the boyfriend later woke up and found her unresponsive. She was a known drug user | Para-fluoroisobutyryl fentanyl | Alprazolam 0.033 mg/L, cannabinoids, ibuprofen |
|
| 4 | 39 W/M | He lived in a homeless shelter and was found dead in bed. Known to use multiple drugs including heroin and cocaine | Furanyl fentanyl | Desproprionyl fentanyl, ethanol 0.301% (chest fluid), 0.268% (brain), morphine <0.01 mg/kg, codeine <0.01 mg/kg, hydrocodone <0.01 mg/kg, hydromorphone <0.01 mg/kg, trazodone, chlorphenyl piperazine |
|
| 5 | 34 W/F |
| Carfentanil and furanyl fentanyl | Despropionyl fentanyl, cocaine 0.011 mg/L, benzoylecgonine 0.124 mg/L, methylecgonidine, methylecgonine, levamisole, cannabinoids |
|
| 6 | 37 W/M | Found behind bushes decomposed | Carfentanil | Ethanol 0.089% (bile), 0.051% (brain), dextromethorphan, diphenhydramine, hydroxyzine, ibuprofen, meprobamate, quetiapine, sertraline, norsertraline, cannabinoids |
|
| 7 | 27 W/F | Prostitute found unresponsive in a field with drug paraphernalia littering the area | Carfentanil | Fentanyl 12 ng/mL, morphine 0.013 mg/L, codeine <0.01 mg/L, 6-monoacetylmorphine <0.001 mg/L, cocaine <0.01 mg/L, benzoylecgonine 0.125 mg/L, methylecgonine, chlorcyclizine, hydroxyzine |
|
| 8 | 23 W/M | Found unrespsonive in bathroom by his family with drug paraphernalia | Carfentanil and para-fluoroisobutyryl fentanyl | Ethanol 0.084% (blood), 0.107% (ocular), dextromethorphan | Acute combined drug toxicity (carfentanil, para-fluorobutyryl/para-fluoroisobutyryl fentanyl and ethanol) accident |
| 9 | 19 W/M |
| Carfentanil and furanyl fentanyl | Despropionyl fentanyl, morphine <0.01 mg/L, codeine <0.01 mg/L, cocaine <0.01 mg/L, benzoylecgonine 0.209 mg/L, methylecgonidine, methylecgonine, levamisole, 7-aminoclonazepam, ibuprofen, lidocaine, desethyllidocaine, naloxone, cannabinoids |
|
| 10 | 41 W/M | Found unresponsive in a field | Carfentanil and U-47700 | Fentanyl <1 ng/mL, morphine 0.032 mg/L, codeine <0.01 mg/L, cocaine <0.01 mg/L, methylecgonine, atropine, diphenhydramine, gabapentin, ibuprofen, naloxone, quetiapine, norquetiapine, quinine |
|
| 11 | 41 B/M |
| Para-fluoroisobutyryl fentanyl and butyryl fentanyl | Oxycodone 0.021 mg/L, noroxycodone, alprazolam 0.073 mg/L, dextromethorphan, promethazine, cannabinoids |
|
| 12 | 30 W/M | Found unresponsive on the street with a foamy discharge around his mouth | Carfentanil, para-fluoroisobutyryl fentanyl and U-47700 | Sertraline, norsertraline, diphenhydramine |
|
Representative sampling of cases analyzed including demographic information, case history, toxicology findings and cause and manner of death
| . | Demographic . | History . | Fentanyl analogs detected . | Additional reported compounds . | Cause and manner of death . |
|---|---|---|---|---|---|
| 1 | 28 W/M | Found unresponsive on the floor with a syringe next to him | Beta-hydroxythiofentanyl | Norfentanyl, ethanol 0.331% (blood), 0.352% (bile), metoclopramide, cannabinoids |
|
| 2 | 28 W/M | Found by his mother in his sister's bedroom with his face prone on the floor and a syringe in his underwear | Acetyl fentanyl | Fentanyl 14 ng/mL, cocaine <0.05 mg/L, benzoylecgonine 0.096 mg/L, methylecgonine, levamisole |
|
| 3 | 28 B/F | She was drinking alcoholic frozen drinks with her boyfriend when they both became nauseous, dizzy and vomited. They eventually went to sleep and the boyfriend later woke up and found her unresponsive. She was a known drug user | Para-fluoroisobutyryl fentanyl | Alprazolam 0.033 mg/L, cannabinoids, ibuprofen |
|
| 4 | 39 W/M | He lived in a homeless shelter and was found dead in bed. Known to use multiple drugs including heroin and cocaine | Furanyl fentanyl | Desproprionyl fentanyl, ethanol 0.301% (chest fluid), 0.268% (brain), morphine <0.01 mg/kg, codeine <0.01 mg/kg, hydrocodone <0.01 mg/kg, hydromorphone <0.01 mg/kg, trazodone, chlorphenyl piperazine |
|
| 5 | 34 W/F |
| Carfentanil and furanyl fentanyl | Despropionyl fentanyl, cocaine 0.011 mg/L, benzoylecgonine 0.124 mg/L, methylecgonidine, methylecgonine, levamisole, cannabinoids |
|
| 6 | 37 W/M | Found behind bushes decomposed | Carfentanil | Ethanol 0.089% (bile), 0.051% (brain), dextromethorphan, diphenhydramine, hydroxyzine, ibuprofen, meprobamate, quetiapine, sertraline, norsertraline, cannabinoids |
|
| 7 | 27 W/F | Prostitute found unresponsive in a field with drug paraphernalia littering the area | Carfentanil | Fentanyl 12 ng/mL, morphine 0.013 mg/L, codeine <0.01 mg/L, 6-monoacetylmorphine <0.001 mg/L, cocaine <0.01 mg/L, benzoylecgonine 0.125 mg/L, methylecgonine, chlorcyclizine, hydroxyzine |
|
| 8 | 23 W/M | Found unrespsonive in bathroom by his family with drug paraphernalia | Carfentanil and para-fluoroisobutyryl fentanyl | Ethanol 0.084% (blood), 0.107% (ocular), dextromethorphan | Acute combined drug toxicity (carfentanil, para-fluorobutyryl/para-fluoroisobutyryl fentanyl and ethanol) accident |
| 9 | 19 W/M |
| Carfentanil and furanyl fentanyl | Despropionyl fentanyl, morphine <0.01 mg/L, codeine <0.01 mg/L, cocaine <0.01 mg/L, benzoylecgonine 0.209 mg/L, methylecgonidine, methylecgonine, levamisole, 7-aminoclonazepam, ibuprofen, lidocaine, desethyllidocaine, naloxone, cannabinoids |
|
| 10 | 41 W/M | Found unresponsive in a field | Carfentanil and U-47700 | Fentanyl <1 ng/mL, morphine 0.032 mg/L, codeine <0.01 mg/L, cocaine <0.01 mg/L, methylecgonine, atropine, diphenhydramine, gabapentin, ibuprofen, naloxone, quetiapine, norquetiapine, quinine |
|
| 11 | 41 B/M |
| Para-fluoroisobutyryl fentanyl and butyryl fentanyl | Oxycodone 0.021 mg/L, noroxycodone, alprazolam 0.073 mg/L, dextromethorphan, promethazine, cannabinoids |
|
| 12 | 30 W/M | Found unresponsive on the street with a foamy discharge around his mouth | Carfentanil, para-fluoroisobutyryl fentanyl and U-47700 | Sertraline, norsertraline, diphenhydramine |
|
| . | Demographic . | History . | Fentanyl analogs detected . | Additional reported compounds . | Cause and manner of death . |
|---|---|---|---|---|---|
| 1 | 28 W/M | Found unresponsive on the floor with a syringe next to him | Beta-hydroxythiofentanyl | Norfentanyl, ethanol 0.331% (blood), 0.352% (bile), metoclopramide, cannabinoids |
|
| 2 | 28 W/M | Found by his mother in his sister's bedroom with his face prone on the floor and a syringe in his underwear | Acetyl fentanyl | Fentanyl 14 ng/mL, cocaine <0.05 mg/L, benzoylecgonine 0.096 mg/L, methylecgonine, levamisole |
|
| 3 | 28 B/F | She was drinking alcoholic frozen drinks with her boyfriend when they both became nauseous, dizzy and vomited. They eventually went to sleep and the boyfriend later woke up and found her unresponsive. She was a known drug user | Para-fluoroisobutyryl fentanyl | Alprazolam 0.033 mg/L, cannabinoids, ibuprofen |
|
| 4 | 39 W/M | He lived in a homeless shelter and was found dead in bed. Known to use multiple drugs including heroin and cocaine | Furanyl fentanyl | Desproprionyl fentanyl, ethanol 0.301% (chest fluid), 0.268% (brain), morphine <0.01 mg/kg, codeine <0.01 mg/kg, hydrocodone <0.01 mg/kg, hydromorphone <0.01 mg/kg, trazodone, chlorphenyl piperazine |
|
| 5 | 34 W/F |
| Carfentanil and furanyl fentanyl | Despropionyl fentanyl, cocaine 0.011 mg/L, benzoylecgonine 0.124 mg/L, methylecgonidine, methylecgonine, levamisole, cannabinoids |
|
| 6 | 37 W/M | Found behind bushes decomposed | Carfentanil | Ethanol 0.089% (bile), 0.051% (brain), dextromethorphan, diphenhydramine, hydroxyzine, ibuprofen, meprobamate, quetiapine, sertraline, norsertraline, cannabinoids |
|
| 7 | 27 W/F | Prostitute found unresponsive in a field with drug paraphernalia littering the area | Carfentanil | Fentanyl 12 ng/mL, morphine 0.013 mg/L, codeine <0.01 mg/L, 6-monoacetylmorphine <0.001 mg/L, cocaine <0.01 mg/L, benzoylecgonine 0.125 mg/L, methylecgonine, chlorcyclizine, hydroxyzine |
|
| 8 | 23 W/M | Found unrespsonive in bathroom by his family with drug paraphernalia | Carfentanil and para-fluoroisobutyryl fentanyl | Ethanol 0.084% (blood), 0.107% (ocular), dextromethorphan | Acute combined drug toxicity (carfentanil, para-fluorobutyryl/para-fluoroisobutyryl fentanyl and ethanol) accident |
| 9 | 19 W/M |
| Carfentanil and furanyl fentanyl | Despropionyl fentanyl, morphine <0.01 mg/L, codeine <0.01 mg/L, cocaine <0.01 mg/L, benzoylecgonine 0.209 mg/L, methylecgonidine, methylecgonine, levamisole, 7-aminoclonazepam, ibuprofen, lidocaine, desethyllidocaine, naloxone, cannabinoids |
|
| 10 | 41 W/M | Found unresponsive in a field | Carfentanil and U-47700 | Fentanyl <1 ng/mL, morphine 0.032 mg/L, codeine <0.01 mg/L, cocaine <0.01 mg/L, methylecgonine, atropine, diphenhydramine, gabapentin, ibuprofen, naloxone, quetiapine, norquetiapine, quinine |
|
| 11 | 41 B/M |
| Para-fluoroisobutyryl fentanyl and butyryl fentanyl | Oxycodone 0.021 mg/L, noroxycodone, alprazolam 0.073 mg/L, dextromethorphan, promethazine, cannabinoids |
|
| 12 | 30 W/M | Found unresponsive on the street with a foamy discharge around his mouth | Carfentanil, para-fluoroisobutyryl fentanyl and U-47700 | Sertraline, norsertraline, diphenhydramine |
|
Table III details the number of cases in which fentanyl analogs were detected by UHPLC-Ion Trap-MSn compared to GC–MS, as well as corresponding LODs for both instruments. When comparing the number of cases screened using both the UHPLC-Ion Trap-MSn and the routine GC–MS method, only 24% of fentanyl analogs were detected by GC–MS. It is apparent that without this method, many of these cases would remain undetermined due to the difference in detection limits between the two instruments. A good example is carfentanil where 78% of positive cases were undetected in the initial GC–MS screening, due to the 10-fold increase in detection limit.
Comparison of number of positive cases for synthetic opioids on LC-Ion Trap MSn and GC–MS with respective limits of detection
| Analog . | No. of cases detected by LC-Ion Trap MSn . | No. of cases detected by GC–MS . | Limits of detection on LC-Ion Trap MSn (ng/mL) . | Limits of detection on GC–MS (ng/mL) . |
|---|---|---|---|---|
| Acetyl fentanyl | 13 | 4 | 0.2 | 5 |
| Beta-hydroxythiofentanyl | 9 | 0 | 0.1 | a |
| Butyryl fentanyl | 3 | 1 | 0.2 | 0.5 |
| Carfentanil | 134 | 30 | 0.1 | 1 |
| Furanyl fentanyl | 37 | 9 | 0.5 | 2.5 |
| 4-Fluoroisobutyryl fentanyl | 22 | 16 | 0.5 | 1 |
| U-47700 | 4 | 1 | 0.5 | – |
| Analog . | No. of cases detected by LC-Ion Trap MSn . | No. of cases detected by GC–MS . | Limits of detection on LC-Ion Trap MSn (ng/mL) . | Limits of detection on GC–MS (ng/mL) . |
|---|---|---|---|---|
| Acetyl fentanyl | 13 | 4 | 0.2 | 5 |
| Beta-hydroxythiofentanyl | 9 | 0 | 0.1 | a |
| Butyryl fentanyl | 3 | 1 | 0.2 | 0.5 |
| Carfentanil | 134 | 30 | 0.1 | 1 |
| Furanyl fentanyl | 37 | 9 | 0.5 | 2.5 |
| 4-Fluoroisobutyryl fentanyl | 22 | 16 | 0.5 | 1 |
| U-47700 | 4 | 1 | 0.5 | – |
aBeta-hydroxythiofentanyl cannot be detected via GC–MS.
Comparison of number of positive cases for synthetic opioids on LC-Ion Trap MSn and GC–MS with respective limits of detection
| Analog . | No. of cases detected by LC-Ion Trap MSn . | No. of cases detected by GC–MS . | Limits of detection on LC-Ion Trap MSn (ng/mL) . | Limits of detection on GC–MS (ng/mL) . |
|---|---|---|---|---|
| Acetyl fentanyl | 13 | 4 | 0.2 | 5 |
| Beta-hydroxythiofentanyl | 9 | 0 | 0.1 | a |
| Butyryl fentanyl | 3 | 1 | 0.2 | 0.5 |
| Carfentanil | 134 | 30 | 0.1 | 1 |
| Furanyl fentanyl | 37 | 9 | 0.5 | 2.5 |
| 4-Fluoroisobutyryl fentanyl | 22 | 16 | 0.5 | 1 |
| U-47700 | 4 | 1 | 0.5 | – |
| Analog . | No. of cases detected by LC-Ion Trap MSn . | No. of cases detected by GC–MS . | Limits of detection on LC-Ion Trap MSn (ng/mL) . | Limits of detection on GC–MS (ng/mL) . |
|---|---|---|---|---|
| Acetyl fentanyl | 13 | 4 | 0.2 | 5 |
| Beta-hydroxythiofentanyl | 9 | 0 | 0.1 | a |
| Butyryl fentanyl | 3 | 1 | 0.2 | 0.5 |
| Carfentanil | 134 | 30 | 0.1 | 1 |
| Furanyl fentanyl | 37 | 9 | 0.5 | 2.5 |
| 4-Fluoroisobutyryl fentanyl | 22 | 16 | 0.5 | 1 |
| U-47700 | 4 | 1 | 0.5 | – |
aBeta-hydroxythiofentanyl cannot be detected via GC–MS.
Reporting
The majority of the compounds identified by this screening panel were confirmed and quantified by other analytical procedures in the laboratory. Confirmation of the fentanyl analogs, otherwise not detected by GC–MS, was based upon the combination of retention time and full-scan, MS2, and MS3 spectral integrity, and were reported accordingly. The laboratory is currently in the process of validating an LC-TQ-MS quantitative method for the most commonly encountered fentanyl analogs; when complete, all cases that previously screened positive for fentanyl analogs will be quantified.
Conclusion
As a result of the sudden and unexpected rise in illegal and abused synthetic drugs entering the community of Miami-Dade County over the last 6 years, the MDME Toxicology Laboratory developed and validated an effective screening procedure utilizing UHPLC-Ion Trap-MSn. To the authors’ knowledge, this is the first screening method developed in the United States to identify these synthetic drugs utilizing this instrumentation. Although several new methods using this technology were created in the MDME Toxicology Laboratory to screen for a range of new substances during this time period, the method described targeted the most lethal of the group: the synthetic opioids. The goal of the new method was to provide a simple, robust, and qualitatively accurate assay to identify these opioids at low concentrations in postmortem fluids and tissues. The ability to detect these substances at concentrations relevant to their potency was critical. As a result, this method was able to achieve minimal limits of detection between 0.1 and 0.5 ng/mL for most compounds. Additionally, the qualitative spectral information must be strong enough to identify these substances with a high degree of certainty. This was achieved by the method's ability to provide MS2 and/or MS3 full spectral data for all 44 compounds. The method described was applied to over 500 postmortem cases from 2014 to the end of 2016, with 375 being positive for one or more fentanyl or synthetic opioid analogs with the resulting cause of death attributed to the drug or drugs detected. The presented cases represent a cross-section of those investigated using this new method. Their similarities in manner of death and pathology findings tell the tale of this epidemic that has afflicted not only Miami-Dade County, but many other regions of the United States (30). The analytical abilities of forensic toxicology laboratories investigating postmortem samples containing these deadly new synthetic drugs has become essential in the last 6 years. The application of Ion Trap-MSn technology coupled to UHPLC at the MDME has been an invaluable tool to identify these new lethal substances in our community and will be essential in the future investigation of the unknown compounds to come.
Acknowledgments
The authors thank and acknowledge the hard work of Alex Giachetti, Susan Humphrey and Erin Walsh from the Miami-Dade County Medical Examiner Department in the preparation of samples and data presentation. They also acknowledge the contributions from scientists and engineers at Bruker Daltonics.
References
(2016) Rules − 2016 – Final Order: Temporary Placement of Butyryl Fentanyl and Beta-Hydroxythiofentanyl Into Schedule I. Drug Enforcement Administration. https://www.deadiversion.usdoj.gov/fed_regs/rules/2016/fr0512_2.htm (accessed Feb 09, 2017).
(
(2015) Acetylfentanyl: Critical Review Report. World Health Organization. http://www.who.int/medicines/access/controlled-substances/5.2_Acetylfentanyl_CRev.pdf (accessed Feb 09, 2017).
(2016) EMCDDA–Europol 2015 Annual Report on the implementation of Council Decision 2005/387/JHA. European Monitoring Centre for Drugs and Drug Addiction. http://www.emcdda.europa.eu/system/files/publications/2880/TDAS16001ENN.pdf (accessed Feb 09, 2017).
(2016) Rules − 2016 – Final Order: Temporary Placement of U-47700 Into Schedule I. Drug Enforcement Administration. https://www.deadiversion.usdoj.gov/fed_regs/rules/2016/fr1114.htm (accessed Feb 09, 2017).
(2016) Rules − 2016 – Final Order: Temporary Placement of Furanyl Fentanyl Into Schedule I. Drug Enforcement Administration. https://www.deadiversion.usdoj.gov/fed_regs/rules/2016/fr1129.htm (accessed Feb 09, 2017).
Fentanyl drug profile (chemistry, effects, other names, synthesis, mode of use, pharmacology, medical use, control status). European Monitoring Centre for Drugs and Drug Addiction. http://www.emcdda.europa.eu/publications/drug-profiles/fentanyl (accessed Feb 09, 2017).