Summary
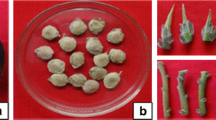
A protocol was developed for rapid clonal propagation of the important medicinal climber, Tinospora cordifolia, through in vitro culture of mature nodal explants. Shoots were initiated on both Murashige and Skoog (MS) medium and woody plant medium (WPM) supplemented with 2.32 μM kinetin (KIN). Of the two basal media tested, WPM was found to be superior to MS medium for the induction of multiple shoots. Among the cytokinins tested, N6-benzyladenine (BA) was more effective than KIN for axillary shoot proliferation. KIN was superior to BA in terms of shoot elongation. An average multiplication rate of 6.3 shoots per explant was obtained with WPM supplemented with 8.87 μM BA. Shoot clumps harvested from this medium were transferred to WPM supplemented with 2.22 μM BA and 4.65 μM KIN for shoot elongation. Elongated shoots were rooted in half-strength MS medium supplemented with 2.85 μM indole-3-acetic acid (IAA). Rooted plantlets were successfully transferred to sand and established with 80% survival.
Similar content being viewed by others

References
Anonymous. The wealth of India, raw materials, vol. 10. New Delhi, India: Central Scientific and Industrial Research (CSIR); 1976:251.
Bajaj, Y. P. S.; Furmanowa, M.; Olszowska, O. Biotechnology of the micropropagation of medicinal and aromatic plants. In: Bajaj, Y. P. S., ed. Biotechnology in agriculture and forestry, vol. 4, Medicinal and aromatic plants-I. Berlin: Springer; 1988:60–103.
Baruah, A.; Sarma, D.; Saud, J.; Sing, R. S.. In vitro regeneration of Hypericum patulum Thumb.—a medicinal plant. Ind. J. Exp. Biol. 39:947–949; 2001.
Joshi, M.; Dhar, U. In vitro propagation of Saussurea obvallata (DC.) Edgew.—an endangered ethnoreligious medicinal herb of Himalaya. Plant Cell Rep. 21:933–939; 2003.
Komalavalli, N.; Rao, M. V.. In vitro micropropagation of Gymnema sylvestre—a multipurpose medicinal plant. Plant Cell Tiss. Organ Cult. 61:97–105; 2000.
Kumar, R.; Sharma, K.; Agarwal, V.. In vitro clonal propagation of Holarrhena antidysenterica (L.) Wall. through nodal explants from mature trees. In Vitro Cell. Dev. Biol.—Plant 41:137–144; 2005.
Kumar, S.; Narula, A.; Sharma, M. P.; Srivastava, P. S. Effect of copper and zinc on growth, secondary metabolite content and micropropagation of Tinospora cordifolia: a medicinal plant. Phytomorphology 53(1):79–91; 2003.
Lloyd, G.; McCown, B. Commercially feasible micropropagation of mountain laurel, Kalmia latifolia, by the use of shoot tip culture. Proc. Int. Plant Prop. Soc. 30:421–427; 1980.
Martin, G.; Geetha, S. P.; Raghu, A. V.; Balachandran, I.; Ravindran, P. N. Micropropagation of Holarrhena pubescens. J. Trop. Med. Plants 6(1):111–117; 2005.
Mathur, J.; Mukunthakumar Micropropagation of Bauhinia variegata and Parkinsoma aceuleata from nodal explants of mature trees. Plant Cell Tiss. Organ Cult. 28:119–121; 1992.
Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 15:473–497; 1962.
Nadkarni, A. K. Indian materia medica, vol. 1, 3rd ed. Bombay, India: Popular Book Depot; 1954:1220–1221.
Nayampalli, S. S.; Desai, N. K.; Ainapure, S. S. Anti-allergic properties of T. cordifolia in animal models. Ind. J. Pharmacol. 18:250–252; 1986.
Nirmal Babu, K.; Sajina, A.; Minoo, D.; John, P. M.; Mini, P. M.; Tushar, K. V.; Rema, J.; Ravindran, P. N. Micropropagation of camphor tree (Cinnamomum camphora). Plant Cell Tiss. Organ Cult. 9:81–88; 2003.
Pathak, A. K.; Jain, D. C.; Sharma, R. P. Chemistry and biological activities of the genera Tinospora—a review. Int. J. Pharma. 33:277–287; 1995.
Pruski, K.; Astatkie, T.; Nowak, J. Tissue culture propagation of Mongolian cherry (Prunus fruticosa) and Nanking cherry (Prunus tomentosa). Plant Cell Tiss. Organ Cult. 82:207–211; 2005.
Purohit, K. V.; Lok, M. S.; Palani; Shyamal, K.; Nandi; Rikhari, H. C. In vitro regeneration of Quercus floribunda Lindl. through cotyledonary nodes: an important tree of central Himalaya. Curr. Sci. 83(3):312–315; 2002.
Raha, S.; Roy, S. C. In vitro plant regeneration in Holarrhena antidysenterica Wall. through high frequency axillary shoot proliferation. In Vitro Cell. Dev. Biol.—Plant. 37:232–236; 2001.
Rege, N.; Dahanukar, S.; Karandikar, S. M. Hepatoprotective effect of T. cordifolia against carbon tetrachloride induced liver damage. Indian Drugs 21:544–555; 1984.
Sivarajan, V. V.; Balachandran, I. Ayurvedic drugs and their plant sources. New Delhi: Oxford and IBH Publishing Co.; 1994:38–39.
Tornero, O. P.; Lopez, J. M.; Egea, J.; Burgos, L. Effect of basal media and growth regulators on the in vitro propagation of apricot (Prunus armenica L.) cv. Canino. J. Hort. Sci. Biotech. 75:283–286; 2000.
Warrier, P. K.; Nambiar, V. P. K.; Ramankutty, C. Indian medicinal plants, vol. 5. Chennai: Orient Longman; 1996:283–290.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Raghu, A.V., Geetha, S.P., Martin, G. et al. In vitro clonal propagation through mature nodes of Tinospora cordifolia (Willd.) Hook. F. & Thoms.: An important ayurvedic medicinal plant. In Vitro Cell.Dev.Biol.-Plant 42, 584–588 (2006). https://doi.org/10.1079/IVP2006824
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1079/IVP2006824