Abstract
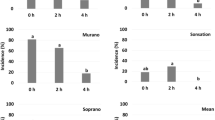
A series of experiments was performed to determine the effectiveness of stubble burning and dry heat on reducing the viability of sclerotia of Sclerotinia sclerotiorum, the causal agent of Sclerotinia stem rot of canola. An intense fire in a uniformly dense triticale stubble led to the death of all sclerotia. However, in another burn with wheat stubble of varying densities, sclerotia survived well when the amount of stubble cover on the ground was 70% or less and sclerotial viability was only reduced when 90% or more of the surface was covered in stubble. Further experiments in dense rice straw showed that temperatures in a stubble fire are variable within a field and also within the rice stubble profile with lower temperatures under fallen stubble and among standing straw than on top of fallen straw. Most sclerotia (98.1 and 98.4%) survived temperatures under 93°C, ∼43% survived temperatures between 93 and 107°C, and very few (27.5 and 1.8%) survived temperatures above 121°C. These results were then verified in a 90s oven experiment where all sclerotia survived under 93°C, 70% survived between 93 and 107°C and none survived temperatures above 121°C. The ability of sclerotia to germinate myceliogenically and carpogenically was significantly reduced when exposed to oven temperatures between 93 and 107°C. These sclerotia also took longer to germinate myceliogenically but were earlier to germinate carpogenically than sclerotia in the control or sclerotia exposed to temperatures below 93°C. Size did not affect the ability of the sclerotia to survive a stubble fire. Burning of stubble does not appear to be an effective method of control of Sclerotinia stem rot in the field.
Similar content being viewed by others
References
Archibold OW, Nelson LJ, Ripley EA, Delanoy L (1998) Fire temperatures in plant communities of the northern mixed prairie. Canadian Field Naturalist 112, 234–240.
Bailey KL (1996) Diseases under conservation tillage systems. Canadian Journal of Plant Science 76, 635–639.
Bedi KS (1961) Factors affecting the viability of sclerotia of Sclerotinia sclerotiorum (Lib.) de Bary. Indian Journal of Agricultural Science 31, 236–245.
Beresford RM, Mulholland RI (1987) Mint rust on cultivated peppermint in Canterbury: disease cycle and control by flaming. New Zealand Journal of Experimental Agriculture 15, 229–233.
Daubenmire RF (1968) Ecology of fire in grasslands. Advances in Ecological Research 5, 209–266.
Dueck J, Morrall RAA, Klassen AJ, Vose J (1981) Heat inactivation of sclerotia of Sclerotinia sclerotiorum. Canadian Journal of Plant Pathology 3, 73–75.
Dueck J, Morrall RAA, McKenzie DL (1983) Control of Sclerotinia sclerotiorum in rapeseed with fungicides. Canadian Journa I of Plant Pathology 5, 289–293.
Gilbert RG (1991) Burning to reduce sclerotia of Sclerotinia sclerotiorum in alfalfa seed fields of southeastern Washington. Plant Disease 75, 141–142.
Hardison JR (1976) Fire and flame for plant disease control. Annual Review of Phytopathology 14, 355–379. doi: 10.1146/annurev.py.l4.090176.002035
Harris NC, Hemmerling EM, Mallmann AJ (1990) ‘Physics principles and applications.’ (McGraw-Hill Publishing Company: New York)
Hind TL, Ash GJ, Murray GM (2003) Prevalence of Sclerotinia stem rot of canola in New South Wales. Australian Journal of Experimental Agriculture 43, 163–168.
Hogan J, Barrett D, Berry P, Lawrance L, Connell P, Mollard W (2004) tralian Crop Report, No. 129, ABARE, Australian Crop Report, Canberra, Australia.
Joshi SD (1924) The wilt disease of safflower (Carthemus tinctorium). Memoirs — Department of Agriculture in India. Botanical Series 13, 39–46.
Kharbanda PD, Tewari JP (1996) Integrated management of canola diseases using cultural methods. Canadian Journal of Plant Pathology 18, 168–175.
Kosasih BD, Willetts HJ (1975) Ontogenetic and histochemical studies of the apothecium of Sclerotinia sclerotiorum. Annals of Botany 39, 185–191.
Lamey HA (1995) Survey of blackleg and Sclerotinia stem rot of canola in North Dakota in 1991 and 1993. Plant Disease 79, 322–324.
Mailer RJ, Colton RT (1997) ‘Quality of Australian canola 1996. Vol.4.’ (Canola Association of Australia: Wagga Wagga, NSW)
Marcroft SJ, Sprague SJ, Salisbury PA, Howlett BJ (2001) Blackleg epidemiology in Australia. In ‘Conference proceedings 12th Australian research assembly on Brassicas’. (Ed. S Marcroft) pp. 78–81. (Deakin Management Centre: Geelong, Vic.)
Mundkur BB (1934) A Sclerotinia rot of Hibiscus sabdariffa L. Indian Journal of Agricultural Science 4, 758–778.
Nimir MB, Payne GF (1978) Effects of spring burning on a mountain range. Journal of Range Management 31, 259–263.
Singh UP, Singh RB, Gupta S (1985) Effect of temperature on sclerotial germination, growth and sclerotia formation in Sclerotinia sclerotiorum. Zeitschriftfur Pflanzen krankheiten und Pflanzenschutz 92, 281–287.
Steadman JR (1979) Control of plant diseases caused by Sclerotinia species. Phytopathology 69, 904–907.
Van Loenen MCA, Turbett Y, Mullins CE, Feilden NEH, Wilson MJ, Leifert C, Seel WE (2003) Low temperature-short duration steaming of soil kills soil-borne pathogens, nematode pests and weeds. European Journal of Plant Pathology 109, 993–1002. doi: 10.1023/B:EJPP0000003830.49949.34
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hind-Lanoiselet, T.L., Lanoiseler, V.M., Lewington, F.K. et al. Survival of Sclerotinia sclerotia under fire. Australasian Plant Pathology 34, 311–317 (2005). https://doi.org/10.1071/AP05048
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1071/AP05048