Abstract
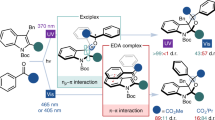
A new series of all-heterahomocalixarene-type structures was prepared. The new hetera[2n]metacyclophanes obtained contain a benzophenone moiety as photoactive component. Although both forms possess identical building blocks and differ only in ring size, they are markedly different in photochemical reactivity. The cyclophane size is the driving force for the photochemical behaviour of the benzophenone.
Similar content being viewed by others
Notes and references
Encyclopedia of Supramolecular Chemistry, ed. J. L. Atwood and J. W. Steed, Marcel Dekker, New York, 2004.
Modern Cyclophane Chemistry, ed. R. Gleiter and H. Hopf, Wiley-VCH, Weinheim, 2004.
B. Koning, Carbon rich cyclophanes with unusual properties–an update, Top. Curr. Chem. 1998, 196, 91–136.
C. Seel and F. Vögtle, Molecules with large cavities in supramolecular chemistry, Angew. Chem., Int. Ed. Engl. 1992, 31, 528–549.
Calixarenes Revisited, ed. C. D. Gutsche, Royal Society of Chemistry, Cambridge, 1998.
P. A. Gale, P. Anzenbacher and J. L. Sessler, Calixpyrroles II, Coord. Chem. Rev. 2001, 222, 57.
S. Kumar, D. Paul and H. Singh, Syntheses, structures and interactions of heterocalixarenes, Adv. Heterocycl. Chem. 2005, 89, 65–124.
B. Koning and M. H. Fonseca, Heteroatom-bridged calixarenes, Eur. J. Inorg. Chem. 2000, 2000, 2303–2310.
W. Maes and W. Dehaen, Oxacalix[ n](het)arenes, Chem. Soc. Rev. 2008, 37, 2393–2402.
M.-X. Wang, Heterocalixaromatics, new generation macrocyclic host molecules in supramolecular chemistry, Chem. Commun. 2008 4541–4551.
M.-X. Wang, Nitrogen and oxygen bridged calixaromatics: synthesis, structure, functionalization, and molecular recognition, Acc. Chem. Res. 2012, 45, 182–195.
S. Ibach, V. Prautzsch, F. Vögtle, C. Chartroux and K. Gloe, Homocalixarenes, Acc. Chem. Res. 1999, 32, 729–740.
Y. Chen, D.-X. Wang, Z.-T. Huang and M.-X. Wang, Synthesis, structure and functionalization of homoheterocalix[2]arene[2] triazines: Versatile conformation and cavity structures regulated by the bridging elements, J. Org. Chem. 2010, 75, 3786–3796.
Molecular and Supramolecular Photochemistry, ed. V. Ramamurthy and K. S. Schanze, Optical Sensors and Switches, Marcel Decker Inc., New York, 2001, vol.7,.
L. F. Vieira Ferreira, I. Ferreira Machado, J. P. Da Silva and T. J. F. Branco, Surface photochemistry: benzophenone as a probe for the study of silica and reversed-phase silica surfaces, Photochem. Photobiol. Sci. 2006, 5, 665–673.
L. F. Vieira Ferreira, M. R. Vieira Ferreira, J. P. Da Silva, I. Ferreira Machado, A. S. Oliveira and J. V. Prata, Novel laser-induced luminescence from benzophenone/ o-propylated p-tert-butylcalix[4] arene complexes. A diffuse reflectance study, Photochem. Photobiol. Sci. 2003, 2, 1002–1010.
N. J. Turro, I. R. Gould, J. Liu, W. S. Jenks, H. Staab and R. Alt, Investigations of the influence of molecular geometry on the spectroscopic and photochemical properties of a-Oxo[1. n] paracyclophanes (cyclophanobenzophenones), J. Am. Chem. Soc. 1989, 111, 6378–6383.
CRC Handbook of Organic Photochemistry and Photobiology, ed. W. M. Horspool and P.-S. Song, CRC Press, Boca Raton, FL, 1995.
IUPAC, Commission on Nomenclature of Organic Chemistry, Phane nomenclature. Part I: Phane parent names, Pure Appl. Chem., 1998, 70, 1513–1545.
IUPAC, Commission on Nomenclature of Organic Chemistry, Phane nomenclature. Part II: Modification of the degree of hydrogenation and substitution derivatives of phane parent hydrides, Pure Appl. Chem. 2002, 74, 809–834.
K. K. Park, H. Seo, J.-G. Kim and I.-H. Suh, Synthesis and structure of new cyclophanes containing benzofuran and benzene rings, Tetrahedron Lett. 2000, 41, 1393–1396.
K. K. Park, I. K. Han and J. W. Park, Photochemical synthesis of cyclophanes containing tethered benzofuran rings, J. Org. Chem. 2001, 66, 6800–6802.
More information in ESI
P. J. Wagner, 1,5-Birradicals and five-membered rings generated by d-hydrogen abstraction in photoexcited ketones, Acc. Chem. Res. 1989, 22, 83.
M. Abdul-Aziz, J. V. Auping and M. A. Meador,: Synthesis of substituted 2,3,5,6-tetraarylbenzo[1,2- b:5,4- b’]difurans, J. Org. Chem. 1995, 60, 1303.
K. K. Park, H. Lim, S.-H. Kim and D.-H. Bae, Synthesis of novel cyclophanes containing benzo[1,2- b:5,4- b’]difuran and naphthalene rings, J. Chem. Soc., Perkin Trans. 1 2002 1393–1396.
P. Wagner, B.-S. Park, in Organic Photochemistry, ed. A. Pawda, Dekker, New York, 1991, vol.11, p.227.
G. Cainelli, G. Cardillo, M. Orena and S. Sandri, Polymer supported reagents. Chromic acid on anion exchange resisns. A simple and practical oxidation of alcohols to aldehydes and ketones, J. Am. Chem. Soc. 1976, 98, 6737–6738.
P. J. Wagner, M. A. Meador and J. C. Scaiano, Photocyclizations of o-(benzyloxy)acetophenone and benzophenone: effects of variable rotational freedom on biradical behaviour, J. Am. Chem. Soc. 1984, 106, 7988–7989.
P. J. Wagner, B. Zhou, T. Hasegawa and D. Ward, Diverse photochemistry of sterically congested a-arylacetophenones: ground-state conformational control of reactivity, J. Am. Chem. Soc. 1991, 113, 9640–9654.
P. J. Wagner, M. A. Meador and B.-S. Park, The photocyclization of o-alkoxy phenyl ketones, J. Am. Chem. Soc. 1990, 112, 5199–5211.
T. E. Lehmann, G. Müller and A. Berkessel, Photochemistry of 4’-benzophenone-substituted nucleoside derivatives as models for ribonucleotide reductases: competing generation of 3’-radicals and photoenols, J. Org. Chem. 2000, 65, 2508–2516.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published as part of a themed issue in honour of Jean-Pierre Desvergne on the occasion of his 65th birthday.
Electronic supplementary information (ESI) available: Materials and methods; NMR spectra; characterization of compounds. See DOI: 10.1039/c2pp25025j
Rights and permissions
About this article
Cite this article
Vida, Y., Perez-Inestrosa, E. Cyclophane size drives the photochemical behaviour of benzophenone. Photochem Photobiol Sci 11, 1645–1651 (2012). https://doi.org/10.1039/c2pp25025j
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c2pp25025j