Abstract
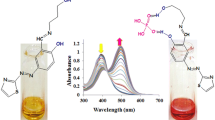
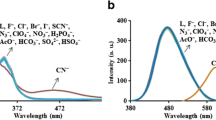
The conjugate N,N-dimethyl curcumin analogue fluorophore dye 1 has been synthesized and its performance as a sensor was demonstrated. As a fluoride and cyanide sensor it enabled visual detection, and showed changes in UV-vis and fluorescence spectra in the presence of fluoride and cyanide ions in aqueous medium. The Job’s plot indicated that the formation of a complex between dye-1 fluoride ions has a 1 : 1 stoichiometric ratio.
Similar content being viewed by others
Notes and references
C. R. Bondy and S. J. Loeb, Coord. Chem. Rev., 2003, 240, 77
F. P. Schmidtchen and M. Berger, Chem. Rev., 1997, 97, 1609
X.-B. Cheng, H. Li, F. Zheng, Q. Lin, H. Yao, Y.-M. Zhang, T.-B. Wei RSC Adv., 2016, 6, 27130
Y.-R. Zhu, H. Li, G.-T. Yan, B. Shi, Y.-M. Zhang, Q. Lin, H. Yao, T.-B. Wei RSC Adv., 2015, 51, 49953
P. A. Gale, Chem. Commun., 2008, 94, 4525
C. Schmuck and M. Schwegmann, J. Am. Chem. Soc., 2005, 127, 337
B. P. Hay, T. K. Firman and B. A. Moyer, J. Am. Chem. Soc., 2005, 127, 1810
J. L. Sessler and D. Seidel, Angew. Chem., Int. Ed., 2003, 42, 5134
A. P. Davis and J. B. Joos, Coord. Chem. Rev., 2003, 240, 143
Q. Li, J.-H. Zhang, Y. Cai, W.-J. Qu, G.-Y. Gao, Q. Lin, H. Yao, Y.-M. Zhang, T.-B. Wei Tetrahedron, 2015, 71, 857
A. Dhillon, M. Nair and D. Kumar, Anal. Methods, 2016, 8, 5338
B. Qiu, Y. Zeng, L. Cao, R. Hu, X. Zhang, T. Yu, J. Chen, G. Yang and Y. Li, RSC Adv., 2016, 6, 49158
H. Miyaji, W. Sato and J. L. Sessler, Angew. Chem., Int. Ed., 2000, 39, 1777
G. Sivaraman, T. Anand and D. Chellappa, ChemPlusChem, 2014, 79, 1761
S. Ghosh, A. Ganguly, M. R. Uddin, S. Mandal, M. A. Alam and N. Guchhait, Dalton Trans., 2016, 45, 11042
S. N. Sahu, S. K. Padhan and P. K. Sahu, RSC Adv., 2016, 6, 90322
J. O. Yu, C. S. Browning and D. H. Farrar, Chem. Commun., 2008, 4, 1020
C. Caltagirone, J. R. Hiscock, M. B. Hurst house, M. E. Light and P. A. Gale, Chem. - Eur. J., 2008, 14, 10236
F. M. Pfeffer, K. F. Lim and K. J. Sedgwick, Org. Biomol. Chem., 2007, 5, 1795
K. J. Chang, B. N. Kang, M. H. Lee and K. S. Jeong, J. Am. Chem. Soc., 2005, 127, 12214
U. I. Kim, J. M. Suk, V. R. Naidu and K. S. Jeong, Chem. - Eur. J., 2008, 14, 11406
K. C. Song, K. M. Lee, N. V. Nghia, W. Y. Sung, Y. Do and M. H. Lee, Organometallics, 2013, 32, 817
V. R. Naidu, M. C. Kim, J. M. Suk, H. J. Kim, M. Lee, E. Sim and K. S. Jeong, Org. Lett., 2008, 10, 5373
G. Jiang, X. Liu, Y. Wu, J. Wang, X. Dong, G. Zhang, Y. Li and X. Fan, RSC Adv., 2016, 6, 59400
Y.-K. La, J.-A. Hong, Y.-J. Jeong and J. Lee, RSC Adv., 2016, 6, 84098
G. Sivaraman and D. Chellappa, J. Mater. Chem. B, 2013, 1, 5768
T. S. Reddy, R. Maragani and R. Misra, Dalton Trans., 2016, 45, 2549
Z. Xu and L. Xu, Chem. Commun., 2016, 52, 1094
R. Ali, S. S. Razi, R. C. Gupta, S. K. Dwivedi and A. Misra, New J. Chem., 2016, 40, 162
J. M. Suk and K. S. Jeong, J. Am. Chem. Soc., 2008, 130, 11868
J. R. Hiscock, C. Caltagirone, M. E. Light, M. B. Hursthouse and P. A. Gale, Org. Biomol. Chem., 2009, 7, 1781
M. J. Chmielewski, L. Y. Zhao, A. Brown, D. Curiel, M. R. Sambrook, A. L. Thompson, S. M. Santos, V. Felix, J. J. Davis and P. D. Beer, Chem. Commun., 2008, 45, 3154
P. V. Piatek, M. Lynch and J. L. Sessler, J. Am. Chem. Soc., 2004, 126, 16073
M. J. Chmielewski, M. Charon and J. Jurczak, Org. Lett., 2004, 6, 3501
E. J. Cho, B. J. Ryu, Y. J. Lee and K. C. Nam, Org. Lett., 2005, 7, 2607
M. Boiocchi, L. D. Boca, D. Esteban-Gomez, L. Fabbrizzi, M. Licchelli and E. Monzani, J. Am. Chem. Soc., 2004, 126, 16507
J. Y. Kwon, Y. J. Jang, S. K. Kim, K.-H. Lee, J. S. Kim and J. Yoon, J. Org. Chem., 2004, 69, 5155
C. B. Black, B. Andrioletti, A. C. Try, C. Ruiperez and J. L. Sessler, J. Am. Chem. Soc., 1999, 121, 10438
A. D. Jose, D. K. Kumar, B. Ganguly and A. Das, Org. Lett., 2004, 6, 3445
H. Miyaji and J. L. Sessler, Angew. Chem., Int. Ed., 2001, 40, 154
D. Jimenez, R. Martinez-Manez, F. Sancenon and J. Soto, Tetrahedron Lett., 2002, 43, 2823
H. Miyaji, W. Sato and J. L. Sessler, Angew. Chem., Int. Ed., 2000, 39, 1777
D. H. Lee, J. H. Im, S. U. Son, Y. K. Chung and J. I. Hong, J. Am. Chem. Soc., 2003, 125, 7752
F. Sancenon, R. Martinez-Manez and J. Soto, Angew. Chem., Int. Ed., 2002, 41, 1416
D. H. Lee, K. H. Lee and J. I. Hong, Org. Lett., 2001, 3, 5
R. Nishiyabu and P. Anzenbacher, J. Am. Chem. Soc., 2005, 127, 8270
D. Esteban-Gomez, L. Fabbrizzi and M. Licchelli, J. Org. Chem., 2005, 70, 5717
C.-I. Lin, S. Selvi, J.-M. Fang, P.-T. Chou, C.-H. Lai, Y.-M. Cheng J. Org. Chem., 2007, 72, 3537
X.-L. Lu and M. Xia, RSC Adv., 2016, 6, 85787
E. C. Figueria, L. C. S. Neres, M. R. S. Ruy, G. T. Troiano and M. D. P. T. Sotomayor, Anal. Methods, 2016, 8, 6353
Y. K. Wu, X. J. Peng, J. L. Fan, S. Gao, M. Z. Tian, J. Z. Zhao and S. G. Sun, J. Org. Chem., 2007, 72, 62
M. Binechi, M. Baglan and S. Atilgan, Sens. Actuators, B, 2016, 222, 315
E. B. Veale and T. Gunnlaugsson, J. Org. Chem., 2008, 73, 8073
P. Jayasudha, R. Manivannan and K. Elango, Sens. Actuators, B, 2016, 237, 230
A. Dahan, T. Ashkkenazi, V. Kuznetsov, S. Makievski, E. Drug, L. Fadeev, M. Bramson, S. Schokoroy, E. Rozenshine-Kemelmakher and M. Gozin, J. Org. Chem., 2007, 72, 2289
S. Kumar, V. Luxami and A. Kumar, Org. Lett., 2008, 10, 5542
J. Li, X. Qi, W. Wei, G. Zuo and W. Dong, Sens. Actuators, B, 2016, 232, 666
D. Saravanakumar, N. Sengottuvelan, M. Kandaswamy, P. G. Aravindan and D. Velmurugan, Tetrahedron Lett., 2005, 46, 7255
G. Sivaraman and D. Chellappa, RSC Adv., 2014, 4, 30828
C. F. Chen and Q. Y. Chen, Tetrahedron Lett., 2004, 45, 3957
S. Wang, X. Fei, J. Guo, Q. Yang, Y. Li and Y. Song, Sens. Actuators, B, 2016, 148, 229
K. Ghosh and S. Adhikari, Tetrahedron Lett., 2006, 47, 8165
S.-T. Wang, Y.-W. Sie, C.-F. Wan, A.-T. Wu J. Lumin., 2016, 173, 25
A. Aydogan, A. Koca, M. K. Sener and J. L. Sessler, Org. Lett., 2014, 16, 3764
Y. M. Hijji, B. Barare, A. P. Kennedy and R. Butcher, Sens. Actuators, B, 2009, 136, 297
F. Huo, J. Kang, C. Yin, J. Chao and Y. Zhang, Sens. Actuators, B, 2016, 215, 93
Y. G. Zhao, B. G. Zhang, C. Y. Duan, Z. H. Lin and Q. J. Meng, New J. Chem., 2006, 30, 1207
T.-B. Wei, W.-T. Li, Q. Li, J.-X. Su, W.-J. Qu, Q. Lin, H. Yao, Y.-M. Zhang Tetrahedron Lett., 2016, 57, 2767
G. X. Xu and M. A. Tarr, Chem. Commun., 2004, 9, 1050
K. Prakash, P. R. Sahoo and S. Kumar, Sens. Actuators, B, 2016, 237, 856
S. K. Kim, J. H. Bok, R. A. Bartsch, J. Y. Lee and J. S. Kim, Org. Lett., 2005, 7, 4839
A. Ghosh, D. A. Jose and R. Kaushik, Sens. Actuators, B, 2016, 229, 545
S. Camiolo, P. A. Gale, M. B. Hursthouse, M. E. Light and C. N. Warriner, Tetrahedron Lett., 2003, 44, 1367
L. Wan, Q. Shu, J. Zhu, S. Sin, N. Li, X. Chen and S. Chen, Talanta, 2016, 152, 39
R. M. F. Batista, E. Oliveira, S. P. G. Costa, C. Lodeiro, M. Manuela and M. Raposo, Org. Lett., 2007, 9, 3201
C. B. Rosen, D. S. Hansen and K. V. Gothelf, Org. Biomol. Chem., 2013, 11, 7916
T. Gunnlaugsson, A. P. Davis and M. Glynn, Chem. Commun., 2001, 24, 2556
D. Lin, X. Zhao, Y. Li, X. Yan and L. Chen, RSC Adv., 2016, 8, 6452
S. Y. Kim and J. Hong, Org. Lett., 2007, 9, 3109
Y. Zhou, X. Dong, Y. Zhang, P. Tong and J. Qu, Dalton Trans., 2016, 45, 6839
J. S. Wu, J. H. Zhou, P. F. Wang, X. H. Zhang and S. K. Wu, Org. Lett., 2005, 7, 2133
S. M. Kim, M. King, I. Choi, J. J. Lee and C. Kim, New J. Chem., 2016, 40, 7768
Z. Q. Liu, M. Shi, F. Y. Li, Q. Fang, Z. H. Chen, T. Yi and C. H. Huang, Org. Lett., 2005, 7, 5481
X. Yang, X. Chen, X. Lu, C. Yan, Y. Yu, X. Hang, J. Qu and R. Liu, J. Mater. Chem., 2016, 4, 383
Q. Y. Chen and C. F. Chen, Tetrahedron Lett., 2004, 45, 6493
D. H. Lee, K. H. Lee and J. I. Hong, Org. Lett., 2001, 3, 5
S. K. Kim, J. H. Bok, R. A. Bartsch, J. Y. Lee and J. S. Kim, Org. Lett., 2005, 7, 4839
S. Xu, K. Chen and H. Tian, J. Mater. Chem., 2005, 15, 2676
S. Sun, Q. Shu, P. Lin, Y. Li, S. Sin, X. Chen and D. Wang, RSC Adv., 2016, 6, 93826
F. Cheng, E. M. Bonder and F. Jakle, J. Am. Chem. Soc., 2013, 136, 17288
P. G. Sutariya, A. Pandya, A. Lodha and S. K. Menon, Analyst, 2013, 138, 2531
Y. Jo, N. Chidalla, D.-G. Cho J. Org. Chem., 2014, 79, 9418
S. Madhu and M. Ravikanth, Inorg. Chem., 2014, 53, 1646
D. Jeyanthi, M. Iniya, K. Krishnaveni and D. Chellappa, Spectrochim. Acta, Part A, 2015, 136, 1269
C. Saravanan, S. Easwaramoorthi, C.-Y. Hsiow, K. Wang, M. Hayashi and L. Wang, Org. Lett., 2014, 16, 354
J. L. Fillaut, J. Andrei’s, L. Toupet and J. P. Desvergne, Chem. Commun., 2005, 2924
D. H. Lee, H. Y. Lee, K. H. Lee and J. I. Hong, Chem. Commun., 2001, 1188
M. Boiocchi, L. D. Boca, D. E. Gomez, L. Fabbrizzi, M. Licchelli and M. Monzani, J. Am. Chem. Soc., 2004, 126, 16507
S. Yang, Y. Liu and G. Feng, RSC Adv., 2013, 3, 20171
L.-B. Xing, B. Yang, X.-J. Wang, J.-J. Wang, B. Chen, Q. Wu, H.-X. Peng, L.-P. Zhang, C.-H. Tung, L.-Z. Wu Langmuir, 2013, 29, 2843
K. Kamada, T. Namikawa, S. Senatore, C. Matthews, P.-F. Lenne, O. Maury, C. Andraud, M. Ponce-Vargas, B. L. Guennic, D. Jacquemin, P. Agbo, D. D. An, S. S. Gauny, X. Liu, R. S. Abergel, F. Fages and A. D. Aleo, Chem. - Eur. J., 2016, 22, 1
Y. Yue, C. Yin, F. Huo, J. Chao and Y. Zhang, Sens. Actuators, B, 2014, 202, 551
W.-C. Lin, S.-K. Fang, J.-W. Hu, H.-Y. Tsai, K.-Y. Chen Anal. Chem., 2014, 86, 4648
H. Lenormand, J.-P. Goddard and L. Fensterbank, Org. Lett., 2013, 15, 748
X. Zhou, R. Lai, H. Li and C. I. Stains, Anal. Chem., 2015, 87, 408
G.-L. Fu, H. Pan, Y.-H. Zhao, C.-H. Zhao Org. Biomol. Chem., 2011, 37, 3152
K. Ponnuvel and V. Padmini, J. Lumin., 2016, 169, 289
Y.-W. Liu, M. X. Kao and A. T. Wu, Sens. Actuators, B, 2015, 208, 429
Y. Yang, C. Yin, F. Huo, J. Chao, Y. Zhang and F. Cheng, Sens. Actuators, B, 2014, 193, 220
S. Pramanik, V. Bhalla and M. Kumar, ACS Appl. Mater. Interfaces, 2014, 6, 5930
X. Cheng, R. Tang, H. Jia, J. Feng, J. Qin and Z. Li, ACS Appl. Mater. Interfaces, 2012, 4, 4387
L. Zhang, D. Wei, S. Wang and S. Jiang, Tetrahedron, 2012, 68, 636
V. Kumar, M. P. Kaushik, A. K. Srivastava, A. Pratap, V. Thiruvenkatam, T. N. Guru Row Anal. Chim. Acta, 2010, 663, 77
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery Jr., J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, O. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski and D. J. Fox, Gaussian 09, Revision A.02, Gaussian, Inc., Wallingford CT, 2009
G. Banuppriya, R. Sribalan, V. Padmini and V. Shanmugaiah, Bioorg. Med. Chem. Lett., 2016, 26, 1655
K. Ponnuvel, G. Banuppriya and V. Padmini, Sens. Actuators, B, 2016, 234, 34
Author information
Authors and Affiliations
Corresponding author
Additional information
Electronic supplementary information (ESI) available: Synthesis of compound dye 1, characterization data, and UV-vis and fluorescence studies. See DOI: 10.1039/C6PP00254D
Rights and permissions
About this article
Cite this article
Ponnuvel, K., Santhiya, K. & Padmini, V. Curcumin based chemosensor for selective detection of fluoride and cyanide anions in aqueous media. Photochem Photobiol Sci 15, 1536–1543 (2016). https://doi.org/10.1039/c6pp00254d
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c6pp00254d