Abstract
Decision-making is a complex process that is important for everyday life. This study examined the effect of the degree of success, and outcome uncertainty, on decision-making and associated neural substrate activation in schizophrenia subjects (SZS) and normal comparison subjects (NCS). A total of 15 subjects with a diagnosis of schizophrenia and 15 age- and education-matched NCS participated in this study. These subjects completed the two-choice prediction task during functional magnetic resonance imaging. Decision-making characteristics and activation of neural substrates were obtained at 20, 50, or 80% error rate. Success and uncertainty influenced the behavioral characteristics on the two-choice prediction task, and the task-related activation in SZS and NCS. Neither success nor uncertainty differentially affected the behavioral characteristics of SZS relative to NCS during the two-choice prediction task. Nonetheless, there was a significant interaction between group and error rate in bilateral parietal cortex. The activation in NCS was the highest when the outcome was most uncertain. In contrast, task-related activation in SZS was not modulated by the degree of uncertainty. Thus, SZS failed to utilize the parietal cortex to process decision-making situations with highly uncertain outcomes.
Similar content being viewed by others
INTRODUCTION
Decision-making, that is, selecting an action from a number of alternatives when the outcome is uncertain, is a complex process that is important for everyday life (Tversky and Kahneman, 1981). Both cognitive and affective functions are involved in the decision-making process. Among these functions, two key factors influence the response selection during decision-making. First, the degree of success associated with an action strongly influences response selection when the actions are not explicitly related to a reward (Schultz et al, 2000). Second, uncertainty of an outcome, that is the degree to which the subject can predict whether the decision will be associated with success or failure, critically contributes to the ability to form an internal prediction model (Egelman et al, 1998). In the absence of explicit reward, and when the outcome of a decision is uncertain, subjects use the history of successes and failures associated with different response alternatives to form a model and guide their action in order to obtain the highest likelihood of success (Calfee and Atkinson, 1966; Goulet and Barclay, 1967; Ludvigson, 1966).
Decision-making can be dysfunctional in patients with different neuropsychiatric disorders, including schizophrenia (Mogg et al, 1991; Lyon et al, 1986; Garety et al, 1991; Brebion et al, 1997; American Psychiatric Association, 1994; Rahman et al, 1999). Although no single task can probe all aspects of decision-making, we have used the two-choice prediction task to examine the sequential organization of responses during decision-making, and the influence of history of uncertainty or success influence the selection of responses (Paulus et al, 1996,1999,2001b). During the two-choice prediction task, subjects are asked to predict repeatedly the location of a stimulus on a computer screen. Within an experimental session, individual schizophrenia patients generate response sequences that are both highly predictable and rigid or highly unpredictable (Paulus et al, 1996) and are not well predicted by the previous stimulus or the previous outcome (Paulus et al, 1999). In contrast, normal comparison subjects (NCS) generate sequences that are moderately unpredictable, and are often related to the previous outcome or the previous stimulus. In schizophrenia patients, as opposed to NCS, the current response can be predicted not only by the immediately preceding response but also by responses that were made many trials before, which yields extensive temporal correlations (Paulus et al, 1999). These decision-making characteristics in schizophrenia patients are independent of psychopathology and are stable over time (Paulus et al, 2001b). In combination, these results support the hypothesis that decision-making dysfunctions in schizophrenia patients are because of abnormal processing of response sequences and altered processing of external stimuli. However, it is unclear whether these dysfunctions are because of changes in the association between response and outcome, or because of the inability to form accurately a prediction model.
Based on behavioral studies, decision-making has been separated into: assessment, that is, relating a stimulus to outcome probabilities; evaluation, that is, optimizing among competing responses; and executive processes, selecting or updating ongoing response strategies (Kahneman and Tversky, 1984). Not surprisingly, functional neuroimaging studies have shown that decision-making is critically dependent on a widely distributed neural network that includes the inferior prefrontal cortex (Paulus et al, 2001a; Ernst et al, 2002), ventromedial and ventro-lateral frontal cortex (Elliott et al, 2000,1999; Rogers et al, 1999), anterior cingulate (Elliott et al, 2000), insula (Critchley et al, 2001), and parietal cortex (Paulus et al, 2001a). Specifically, the inferior prefrontal areas have been linked to the detection and selection of advantageous over disadvantageous responses (Elliott et al, 2000; Rogers et al, 1999; O'Doherty et al, 2001). The dorsolateral prefrontal cortex has been linked to the processes of response selection during decision-making (Paulus et al, 2001a) and the anterior cingulate to the evaluative components, that is, the distinction between hypotheses testing and guessing (Elliott and Dolan, 1998) or reward-based response selection (Bush et al, 2002; Gehring and Willoughby, 2002). The parietal areas have been implicated in the assessment of functions, for example, anticipatory processing during decision-making (Critchley et al, 2001), relative to the utility of behavioral alternatives (Platt and Glimcher, 1999; Shadlen and Newsome, 2001).
Our previous studies of error-rate-related decision-making in normal controls showed that inferior prefrontal areas show more activation at low error rates, that is, when response selection was associated with a high success rate, and posterior parietal activation was linked to outcome-related responses (Paulus et al, 2002a). Therefore, if alterations in processing during decision-making in schizophrenia subjects (SZS) were because of changes in success-related response selection, one would predict that there would be differences in the inferior prefrontal cortical activation. Alternatively, if an alteration in uncertainty processing underlies decision-making dysfunctions in schizophrenia patients, one would predict to see differences predominantly in the posterior parietal cortex.
Functional neuroimaging studies with SZS have shown dysfunctions of the neural substrates critical for decision-making, that is, prefrontal cortex, anterior cingulate, and parietal cortex. Specifically, working-memory studies during functional magnetic resonance imaging (fMRI) have shown that both chronic and first-episode never-medicated schizophrenia patients showed deficits in working memory performance associated with an increased or decreased activation (Callicott et al, 1998; Barch et al, 2001), or an increased spatial heterogeneity of dorsolateral prefrontal cortex activation (Manoach et al, 2000). The dorsolateral prefrontal cortex dysfunction in SZS during a working memory task has been associated with disorganization symptoms (Perlstein et al, 2001). Moreover, anterior cingulate dysfunction, related to the evaluation or detection of response conflict during the Stroop task, has been observed in both PET (Nordahl et al, 2001) and fMRI studies (Carter et al, 2001). Finally, both reduced (Artiges et al, 2000; Fletcher et al, 1998) and increased posterior parietal activation (Callicott et al, 1998; Paulus et al, 2002b) have been observed in response to different tasks in SZS. The heterogeneity of these findings has led some investigators to propose that schizophrenia patients show a disruption of processing across different areas in the brain, rather than a circumscribed abnormality in one area (Friston, 1998). This hypothesis has been supported by some studies (Volz et al, 1999; Meyer-Lindenberg et al, 2001; Lawrie et al, 2002) but not others (Spence et al, 2000; Curtis et al, 1999; Fletcher et al, 1998).
This study examined the effect of the degree of success and the uncertainty on decision-making and associated neural substrate activation in SZS, relative to NCS. Based on the response of the subject during the two-choice prediction task, a computer program determined a priori whether the prediction would be ‘correct’ or ‘incorrect’. Specifically, decision-making characteristics and activation of neural substrates were obtained at 20% error rate (2/10 correct predictions), 50% error rate, or 80% error rate. This design enables one to examine the effect of success, that is, the number of correct vs incorrect predictions (high success −20% error rate, low success 80% error rate), and of uncertainty, that is, the degree to which a response predicts an outcome irrespective of success or failure (high unpredictability −50% error rate, low unpredictability −20 and 80% error rate), on decision-making. It was hypothesized that if decision-making dysfunctions in SZS are because of altered processing of failure vs success, neural substrate differences should be most pronounced at high or low error rate, respectively, and should be related to differences in inferior prefrontal cortex processing. Alternatively, if the decision-making dysfunctions are because of altered processing of uncertainty, neural substrate differences should be most pronounced when the outcome is most unpredictable (ie at 50% error rate) and should be related to differences in posterior parietal cortex.
METHODS
This study was approved by the UCSD Human Research Protections Program (000730) and all subjects signed informed consent. Initially, 17 SZS and 16 NCS were enrolled in the study. Two SZS and one NCS were excluded from subsequent analyses because of movement artifact in the echoplanar images. Included were 15 right-handed subjects with the diagnosis of schizophrenia (four females, 11 males), continuous, according to the DSM-IV (1994) with an average age of 41.7±1.6 years (range 30–53), an average education level of 14.4±0.7 years (range 12–23), an average age of onset of 25.9±1.8 years (range 18–39), and an average illness duration of 15.7±2.1 years (range 7–34). The behavioral and functional neuroimaging data from these subjects were compared to a group of 15 right-handed NCS (four females, 11 males) that were matched on age (mean 41.0±2.1, range 21–54) and education (mean 15.3±0.56, range 12–21). All subjects were trained to perform the two-choice prediction and two-choice response task prior to testing during fMRI scanning.
Diagnoses for all subjects were obtained by using a structured clinical interview for DSM-IV diagnosis (SCID-P) (Spitzer et al, 1992). Subjects with a major depressive disorder, bipolar, post-traumatic stress, panic, or obsessive–compulsive disorder were excluded from the study. Subjects with nonremovable materials that respond to high magnetic fields, for example, metal fragments, were also excluded. At the time of testing, the SZS were clinically stable, nine subjects were treated with atypical antipsychotic medication, three subjects were treated with typical antipsychotic medications, and three subjects were not treated with any antipsychotic medications at the time of testing.
Task
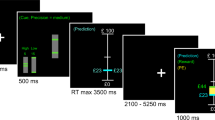
The two-choice prediction task has been described in detail elsewhere (Paulus, 1997). Briefly, a house flanked by a person to the left and right is shown on a computer screen. The goal for the subject is to match a person on the computer screen with a car that is presented on the far left or right side of the screen. For the two-choice prediction task, the subject's goal is to predict which side the car will be presented and to select the left or right button, respectively. After the subject has made a response, the car is presented for 300 ms on the far left or right side. If the selected response matches the side where the car is presented, the person meets up with the car. For the two-choice response task, the car is presented on the left or right side before the subject is asked to respond, and there is no prediction.
Unbeknown to the subject, on the two-choice prediction task the car is presented according to a predetermined schedule. Specifically, the computer program takes the response of the subject into account, and determines whether a response will be ‘correct’ or ‘incorrect’. The key difference between these two tasks is that during the two-choice prediction task, the subject does not know the correct response in advance, and has to decide in the presence of uncertainty, using the previous responses, stimuli, and outcomes to determine their response. In comparison, during the two-choice response task the subject knows the correct answer before selecting a response, decides in the presence of certainty, and does not need to use the sequences of previous responses, stimuli, or outcomes. The task was presented to the subjects in the MRI using an LCD projector, back-projected onto a screen at the subjects' feet, which could be seen via a mirror attached to the head coil. Subjects requiring corrective lenses were provided with a pair of plastic-framed lenses that approximated their degree of correction. Motor responses were made by the right hand using a button box.
A block design was used for this study. The two-choice prediction task was divided into three trial-blocks, each lasting 90 s. During the first trial-block the computer program assured that 50% of all responses were ‘correct’, during the second trial-block 20% of all responses ‘correctly predicted’ the location of the car, and during the third trial-block 80% of all responses were ‘correct’ predictions. Therefore, the first trial-block corresponds to ‘evenly right or wrong’, the second trial-block to being ‘mostly wrong’, and the third trial-block to being ‘mostly right’. The two-choice prediction task was contrasted with the two-choice response task, which was presented between the 50 and 20%, as well as between the 20 and 80% trial blocks for 30 s, to examine task-related activation. The duration of each trial depended on the latency to make a decision, that is, the time between presentation of the initial situation and the selection of the response.
Behavioral Measures
For both the two-choice prediction task and the two-choice response task, the following variables were recorded: (1) the choice selected by the subject (left or right), (2) the response selected by the computer (left or right), and (3) the subjects' latency of response selection (time from the presentation of the current situation to the selection of the response). Based on these variables, the strategies of decision-making in the presence of uncertainty were assessed using the following three sets of measures: (1) General response biases: the number of left or right responses or stay responses (a left response followed by left response), vs switch (left followed by right response) responses. (2) The degree to which the current response was determined by the previous response, the previous stimulus, or a combination of both, quantified by mutual information measures (Herzel and Grosse, 1995). The mutual information between two events, A and B, expressed in units of bits, is the logarithmic likelihood ratio of the observed occurrence rate over the expected chance occurrence rate of A and B. The win-stay/lose-shift mutual information is computed by coding event A as a win (correct) or lose (incorrect) outcome at trial i-1 and B as the same (stay) or different (shift) response at trial i. (3) The predictability of the response sequence measured by the average entropy and the range of subsequence fluctuations between highly predictable (minimum entropy sequence) and highly unpredictable response sequences (maximum entropy sequence), which operationally defines the degree of response dysregulation during the two-choice prediction task. The dynamical entropy is computed via the determination of unique subsequences of responses during the two-choice prediction task as detailed in Grassberger (1989) and Paulus (1997).
fMRI Protocol and Image Analysis Pathway
Magnetic resonance images were obtained using a 1.5 T whole-body system (Siemens, Erlangen). Anatomical T1-weighted images of the whole brain (MPRAGE, TR=11.4 ms, TE=4.4 ms, flip angle=10°, FOV=256 × 256, 1 mm3 voxels) were obtained sagitally to identify the anterior/posterior commissure, to coregister the functional image, and to transform the images into Talairach space (Talairach and Tournoux, 1988). A total of 32 slices of T2*-weighted images were obtained in the axial plane using gradient-recalled echo planar imaging (TE=40 ms, flip angle=90°, 64 × 64 pixel FOV=220 × 220 mm, 3 mm contiguous slice thickness) every 3000 ms for 112 repetitions, yielding a voxel size of 3.43 mm × 3.43 mm × 3 mm in order to minimize signal dropout related to magnetic susceptibility variations in the orbitofrontal cortex.
All structural and functional image processing was done using the analysis of functional neuroimages (AFNI) software package (Cox, 1996). Echoplanar images were coregistered using a 3D coregistration algorithm to the echoplanar image that resulted in the smallest amount of image translation and rotation, relative to all other images. The main dependent measure to assess task-related brain activation was the percent signal change during the two-choice prediction task, relative to the two-choice response task across three delay times. Multiple regression analysis was used to quantify the fMRI time series data (Courtney et al, 1997). Three different reference functions were used to measure the degree of echoplanar signal change during the 50, 20, and 80% reinforcement conditions, respectively. The AFNI program 3dDeconvolve was used to calculate the estimated impulse response function between the reference function and the echoplanar time series using a time shift of 1–3 TR (ie 3–9 s) at each voxel. The relative signal change was computed by dividing the regressor coefficients for each time shift by the zeroth-order regressor coefficient, which measures the average signal intensity during the two-choice response task trial-blocks. A Gaussian filter with FWHM 6 mm was applied to voxelwise percent signal change data to account for individual variations of the anatomical landmarks and to yield the highest detection power (Skudlarski et al, 1999). The data of each subject were normalized to Talairach coordinates, and the measure of relative change of signal intensity during a task of interest (two-choice prediction task during different reinforcement conditions), relative to the baseline task (two-choice response task), was entered into a mixed model nested ANOVA. Specifically, task condition (20, 50, 80%) was used as a fixed factor, subjects as a random factor, which together were nested under the group as a fixed factor (SZS, NCS). A threshold adjustment method, based on Monte-Carlo simulations, was used to guard against identifying false positive areas of activation (Forman et al, 1995). Based on these simulations, it was determined that a voxelwise a priori probability of 0.01 would result in a corrected clusterwise activation probability of 0.05 if a minimum volume of 512 μl (or eight contiguous 4 mm3 voxels) and a connectivity radius of 4 mm was considered. All graphical results are presented as volume-threshold % signal difference or F-maps. Labels for brain activation foci were obtained in Talairach coordinates using the Talairach Demon software (Lancaster et al, 2000).
Statistical Analysis
All analyses for the behavioral data were carried out with SPSS 10.0 (Norusis, 1990). A mixed model ANOVA (fixed factor: task conditions; random factor: subjects) was used to analyze the behavioral measures. The planned comparisons were evaluated using the least significant difference (LSD) post hoc analysis. In order to adjust the degrees of freedom for the correlations in within-subject designs (violations of sphericity), Greenhouse–Geisser (GG) corrections were applied. All analyses were conducted using a response number as a covariate; however, the results did not differ significantly, and the results presented here are based on the mixed model described above.
BEHAVIORAL RESULTS
Error rate significantly affected basic response characteristics during the two-choice prediction task in both groups (Table 2). The error rate effects on response biases, mutual information functions, and the dynamical entropy, however, were similar for both SZS and NCS (Table 2). Specifically, the latency to select a response and the degree to which the current response predicted the next response (mutual information), in both groups, was lowest when the outcome was most unpredictable (Table 1 and Table 2). Thus, the degree of response predictability matches the uncertainty of the outcome. In comparison, the probability of switching from the current response to the alternative response was proportional to the error rate for both SZS and NCS (Table 1). Both SZS- and NCS-generated response sequences were most predictable as measured by the average entropy when the error rate was low (Figure 1). The dysregulation of response sequences, as measured by the range of response sequence predictabilities, differed significantly across groups (Table 1), but not across error-rate conditions. As shown in Figure 1, SZS showed a slightly but significantly higher degree of dysregulation, when averaged across the different error-rate conditions. Although the latency to select a response during the two-choice response task did not differ between SZS and NCS (Table 1, F(1,28)=1.7, NS), SZS made significantly more errors than NCS during the two-choice response task (Table 1, F(1,28)=11.3, p<0.01).
FMRI RESULTS
The main effect of the task, that is, the difference between two-choice prediction and two-choice response task, as evaluated separately for NCS and SZS, revealed a distributed activation across inferior prefrontal, anterior cingulate, and posterior parietal areas in NCS (Figure 2, top) and across inferior prefrontal and anterior cingulate but not posterior parietal areas in SZS (Figure 2, bottom). When the groups were combined, different error-rate conditions significantly affected distributed areas of the brain including the inferior prefrontal, anterior cingulate, and posterior parietal cortex (Table 3). First, prefrontal areas included bilateral inferior frontal gyrus (BA 44/45) extending to the middle frontal gyrus (BA 9/46) and medially to the medial prefrontal cortex (BA 10/11). Second, both dorsal and rostral anterior cingulate (BA 32) showed error-rate- related activation differences across tasks. Third, several posterior parietal areas, including bilateral precuneus (BA 7/18) and left inferior parietal lobule (BA 40), showed error-rate-related activation changes. When the task × error-rate analyses was conducted separately for NCS and SZS, significant error-rate-related changes were found in both the inferior prefrontal cortex and the rostral anterior cingulate, but not in the posterior parietal cortex. The average activation across these functional regions of interest showed a U-shaped relation to the error rate (Figure 4). That is, for both NCS and SZS these regions were most active when the subjects were most successful in predicting the outcome, and were least activated when the outcome was most unpredictable.
Task-related activation averaged across all functional regions of interest that showed a significant effect of error rate (Table 1). Significant increase in activation at low error rates was common in both SZS and NCS.
Several closely related areas revealed a significant task × group × error-rate interaction (Figure 3, Table 2). Bilateral precuneus (BA 7) and superior parietal lobule (BA 7) showed different error-rate-related activation in comparison subjects relative to SZS. As shown in Figure 3, whereas most NCS showed a larger task-related activation at 50% error rate, most SZS did not show larger activation at 50% error rates in the precuneus and superior parietal regions.
Task × group × error rate interaction (upper left) and contrast effect (upper right) in NCS. The contrast was inversely correlated with illness durationin SZS (lower right). Activation patterns in individual subjects show an uncertainty-related pattern in NCS (bottom left) but not SZS (bottom right).
In order to examine whether the interaction between task and error rate across groups was because of uncertainty-related processing in NCS but not in SZS, a contrast vector (activation at 50% error rate−½(activation at 20%+activation at 80% error rate)) was created and tested separately for NCS and SZS. As shown in Figure 3, NCS exhibited a significant contrast effect in the posterior parietal cortex (precuneus, BA 7); however, no such area was observed for SZS (figure not shown). Moreover, the average activation from these functional regions of interest showed a significant contrast × group effect (F(1,28)=11.45, p<0.01). A similar contrast analysis with contrasting high success rate with high error rate (20% error rate−80% error rate) did not yield significant differences between NCS and SZS (F(1,28)=3.27, NS).
To determine whether the difference in the precuneus area was because of the fact that SZS made more errors during the two-choice response task, an ANCOVA, with response errors as covariate, was conducted between NCS and SZS. The corrected model was highly significant (F(3,27)=7.9, p<0.01) and there was a main effect of group (F(1,27)=5.8, p<0.05), but not response error (F(1,27)=3.3, NS), on the contrast vector in the precuneus (BA 7). Whereas the posterior parietal cortex of NCS was relatively more active when the outcome of the decision was most uncertain, no such modulation was found in SZS. Moreover, the lack of modulation was not related to differences in errors made during the two-choice prediction task.
As shown in Figure 3, most SZS showed no change or reduced activation during the 50% error-rate condition. The degree to which this pattern was expressed correlated with duration of illness (r=0.67, p<0.01), but not age (r=0.36, NS) or education (−0.46, NS), in SZS (Figure 3). Thus, the difference in processing uncertainty in the posterior parietal cortex between NCS and SZS increased with increasing duration of illness. There were no significant correlations between the contrast vector in this area and behavioral contrasts (ie difference in response biases, mutual information functions, and dynamical entropy at 50% error rate and 20 or 80% error rate) for both NCS and SZS for the task × group × error-rate interaction areas.
DISCUSSION
SZS relative to NCS show altered processing of outcome uncertainty, but do not show altered processing of response success or failure. Specifically, there was a significant interaction between group and error rate in several areas of the posterior parietal cortex comprising the precuneus (BA7), bilateral superior parietal lobule (BA 40), and right inferior parietal lobule (BA 40). In each of these areas, NCS, but not SZS, showed the largest activation at 50% error rate and less activation at 20 or 80% error rate. Finally, the degree to which SZS lacked an increase in uncertainty-related activation in the precuneus was correlated with illness duration. These results extend previous reports of reduced posterior parietal activation during the two-choice prediction task in SZS (Paulus et al, 2002b) and support the hypothesis of an altered processing of outcome uncertainty during decision-making in SZS that involves the posterior parietal cortex.
In comparison, the behavioral results of this study do not support the hypothesis that the degree of success or the outcome uncertainty differentially affects response selection during decision-making in SZS relative to NCS. As for response switching, the degree to which previous responses or stimuli predicted the current response; the average response entropy and the degree of dysregulation did not reveal an interaction between error rate and group. Thus, both NCS and SZS responded similarly to different error rates.
Success-related changes of activation in neural substrates were similar for both SZS and NCS. At low and high error rates, predominant response strategies develop, for example, lose-shift resulting in repetitive response switching or win-stay in long response repetition. In contrast, when the outcome is highly uncertain, subjects are required to adjust strategies. The lack of the inverted U-shaped relation between error rate and task-related activation in posterior parietal cortex in SZS may reflect an altered processing of outcome uncertainty as previously observed (Paulus et al, 2002b). The key aspect of high uncertainty, relative to low or high success rate, is the inability to establish repetitive response strategies, for example, repeated win-stay or lose-shift responses, as evidenced by the decrease in response predictability. The repeated adjustment of response selection, because of highly unpredictable success or failure, appears to engage posterior parietal in NCS but not in SZS. Thus, the current results are consistent with the general hypothesis that there is a disturbance in the processing of decision-making in SZS when there are no dominant response strategies established.
Both structural (Weinberger et al, 1992; Seidman et al, 1994; Gur et al, 2000) and functional (Weinberger et al, 1996; Callicott et al, 1998; Manoach et al, 1999; Rubia et al, 2001a; Volz et al, 2001) imaging studies, using a number of different neurocognitive tasks, in SZS have implicated primarily, and some selectively (Barch et al, 2001), dorsolateral prefrontal cortex dysfunction in SZS. Others still have correlated evaluative dysfunctions in schizophrenia with impaired performance monitoring in the anterior cingulate (Carter et al, 2001,2000,1997; Nordahl et al, 2001). However, there is increasing evidence of both structural (as reviewed in Shenton et al, 2001) and functional parietal cortex dysfunction in SZS. Moreover, blood flow changes in the parietal cortex have been reported to increase with duration of illness in SZS (Schultz et al, 2002).
The posterior parietal cortex has been implicated in a number of processes important for decision-making. Specifically, attentional processes that involve sustained, and possibly selective, attention (Coull et al, 1996), switching from task-relevant local to global targets (Fink et al, 1996; Lamb et al, 1989), voluntary attentional control (Hopfinger et al, 2000), and the distinction between task-irrelevant and task-relevant events (Downar et al, 2001; Kiehl et al, 2001; McCarthy et al, 1997) support the view that this area is critical for the extraction and selection of task-relevant information. Moreover, this area has been implicated in inhibitory control in a number of different paradigms (Garavan et al, 1999; Menon et al, 2001; Rubia et al, 2001b; Steel et al, 2001; Doricchi et al, 1997), that is, the allocation of resources to a response that has to compete with a highly overlearned and potentially habitual behavior. Several studies using decision-making paradigms have implicated the right posterior parietal cortex in autonomic arousal processes (Tranel and Damasio, 1994; Critchley et al, 2000), risk-taking decision-making (Ernst et al, 2002), and guessing (Elliott et al, 1999).
Dissociation between frontoparietal processing has been reported in other neuroimaging studies with SZS. For example, whereas SZS showed performance-related activation deficits during a working-memory task in the dorsolateral prefrontal cortex, these subjects also showed a working-memory load-independent reduction in the parietal cortex (Fletcher et al, 1998). Moreover, working-memory-related activation in the posterior parietal cortex has been found to be correlated with somatic hallucinations (Shergill et al, 2001; Lennox et al, 2000). Others have suggested that parietal cortex dysfunction in SZS may contribute to the difference in semantic fluency vis-à-vis phonologic fluency (Feinstein et al, 1998). SZS showed significantly less activation in posterior parietal areas during a randomization task, which are though to contribute to the perseverative tendencies in these subjects (Artiges et al, 2000). Electrophysiological studies have shown reduced parietal P300 amplitude, which has been interpreted to signify a dysfunction in the continuous memory updating of current events (Nieman et al, 2002).
The lack of uncertainty-related activation in posterior parietal areas in SZS would suggest that SZS engage less processing resources than NCS. How is this compatible with the previously reported increase in outcome-related strategy (Paulus et al, 2002b)? The win-stay/lose-shift strategy consists of two steps. First, the subject needs to remember where the stimulus was presented during the previous trial. Second, the subject needs to determine whether the prediction was correct or incorrect. If the current choice was simply based on the presentation of the previous stimulus, irrespective of the previous correct or incorrect prediction, one would expect increased win-stay/lose-shift consistent responses across all error-rate conditions. The current behavioral results are consistent with this view, showing that SZS tend to select the response based on the location of the previous stimulus and irrespective of whether the prediction was ‘correct’ or ‘incorrect’. In NCS, increased activation in the posterior parietal cortex was associated with a decreased frequency of the dominant response strategy (win-stay/lose-shift) (Paulus et al, 2001a). Processing of prospective gains during decision-making is most difficult when the outcome of the decision is most uncertain and the outcomes of previous responses are most variable at the 50% error-rate condition. Thus, outcome uncertainty and variable history of ‘correct’ and ‘incorrect’ responses may force NCS to increase assessment processing during decision-making using the posterior parietal cortex, a process that is missing in SZS.
SZS, relative to NCS, adjusted their response characteristics similarly for different error rates. Specifically, both groups changed the switching rate, proportional to the error rate, and exhibited the lowest response predictability when the outcome associated with a response was most uncertain (ie at 50% error rate). Similarly, both groups showed a network of neural substrates that showed error-rate-related changes in activation. Bilateral inferior frontal gyrus (BA 44/45) and middle frontal gyrus (BA 9/46), ventromedial prefrontal cortex (BA 10/11), and anterior cingulate (BA 32) showed error-rate-related activation changes. These areas have been implicated in spatial attentional processes (Mesulam, 1999), conflict and error monitoring (Carter et al, 2000; Botvinick et al, 1999), inhibitory processes (Menon et al, 2001; Liddle et al, 2001; Rubia et al, 2001b), and control of eye movements (Luna et al, 1998). Thus, both groups allocate similar error-rate-related processing resources during this decision-making task. This finding is consistent with the observation that SZS respond similar to NCS concerning reinforcement contingencies (for a review, see Stieper et al, 1972).
There are several caveats to the conclusions and limitations of the current study. First, the functional imaging differences between NCS and SZS were not corroborated by behavioral differences that correlated with activation differences in the posterior parietal cortex. The divergence between behavioral measures and functional imaging results may signify that the current measures do not adequately quantify the degree of assessment prior to selecting a response. Event-related fMRI designs or electrophysiological methods that provide high temporal resolution may need to be employed to clarify the relation between behavioral differences and brain processes during the assessment phase of decision-making. Second, the degree of dysregulation in SZS, although significant across error rates, was small and did not reach significance for 50% error rates. It has been pointed out by others (Carter et al, 2001) that fMRI studies may select higher-functioning SZS and, thus, may create a selection bias that reduces performance differences between these groups (Resnick, 1992). Third, the presentation of the error-rate conditions during the fMRI experiment does not control for order effects. Although both behavioral and neuroimaging effects showed order-unrelated changes, future designs may need to employ longer functional sessions with multiple error-rate conditions. Fourth, symptom status was not evaluated in close proximity to testing. Thus, it is unclear if the relation between (1) the lack of uncertainty-related activation in the posterior parietal cortex and (2) illness duration is mediated by the severity of illness at the time of testing. Fifth, SZS committed significantly more errors during the two-choice response task. Although the effect of uncertainty was still significant when the number of errors was entered into the analysis as a covariate, there is still a possibility that activation differences between the two-choice prediction task and the two-choice response task may relate to the number of errors during the two-choice response task.
In conclusion, SZS and NCS show similar task-related activation in response outcome success, which encompasses a distributed network comprising inferior prefrontal, posterior parietal, and cingulate cortex. Whereas NCS showed uncertainty-related activation in the posterior parietal cortex, SZS did not show error-rate-related differences in these areas. Thus, inadequate processing of uncertainty in the posterior parietal cortex may be key substrates in the error-rate-related decision-making dysfunctions in schizophrenia.
Abbreviations
- NCS:
-
normal comparison subjects
- SZS:
-
schizophrenia subjects
- BA:
-
Brodmann area
- fMRI:
-
functional magnetic resonance imaging
- DSM:
-
Diagnostic and Statistical Manual of Mental Disorders
- SCID:
-
Structured Clinical Interview for DSM-IV diagnoses
- MPRAGE:
-
magnetization-prepared rapid acquisition of gradient echo
- FOV:
-
field of view
- AFNI:
-
analysis of functional neuroimages
- FWHM:
-
full width half maximum
- ANOVA:
-
analysis of variance
- ANCOVA:
-
analysis of covariance
References
American Psychiatric Association (1994). Diagnostic and Statistical Manual of Mental Disorders (4th Edition): DSM-IV. The American Psychiatric Association: Washington.
Artiges E, Salame P, Recasens C, Poline JB, Attar-Levy D, De La RA et al (2000). Working memory control in patients with schizophrenia: a PET study during a random number generation task. Am J Psychiatry 157: 1517–1519.
Barch DM, Carter CS, Braver TS, Sabb FW, MacDonald A, Noll DC et al (2001). Selective deficits in prefrontal cortex function in medication-naive patients with schizophrenia. Arch Gen Psychiatry 58: 280–288.
Botvinick M, Nystrom LE, Fissell K, Carter CS, Cohen JD (1999). Conflict monitoring vs selection-for-action in anterior cingulate cortex. Nature 402: 179–181.
Brebion G, Smith MJ, Gorman JM, Amador X (1997). Discrimination accuracy and decision biases in different types of reality monitoring in schizophrenia. J Nerv Ment Dis 185: 247–253.
Bush G, Vogt BA, Holmes J, Dale AM, Greve D, Jenike MA et al (2002). Dorsal anterior cingulate cortex: a role in reward-based decision making. Proc Natl Acad Sci USA 99: 523–528.
Calfee RC, Atkinson RC (1966). Two-choice behavior under limiting cases of contingent reinforcement schedules. J Comp Physiol Psychol 62: 193–200.
Callicott JH, Ramsey NF, Tallent K, Bertolino A, Knable MB, Coppola R et al (1998). Functional magnetic resonance imaging brain mapping in psychiatry: methodological issues illustrated in a study of working memory in schizophrenia. Neuropsychopharmacology 18: 186–196.
Carter CS, Macdonald AM, Botvinick M, Ross LL, Stenger VA, Noll D et al (2000). Parsing executive processes: strategic vs evaluative functions of the anterior cingulate cortex. Proc Natl Acad Sci USA 97: 1944–1948.
Carter CS, MacDonald AW, Ross LL, Stenger VA (2001). Anterior cingulate cortex activity and impaired self-monitoring of performance in patients with schizophrenia: an event-related fMRI study. Am J Psychiatry 158: 1423–1428.
Carter CS, Mintun M, Nichols T, Cohen JD (1997). Anterior cingulate gyrus dysfunction and selective attention deficits in schizophrenia: [15O]H2O PET study during single-trial Stroop task performance. Am J Psychiatry 154: 1670–1675.
Coull JT, Frith CD, Frackowiak RS, Grasby PM (1996). A fronto-parietal network for rapid visual information processing: a PET study of sustained attention and working memory. Neuropsychologia 34: 1085–1095.
Courtney SM, Ungerleider LG, Keil K, Haxby JV (1997). Transient and sustained activity in a distributed neural system for human working memory [see comments]. Nature 386: 608–611.
Cox RW (1996). AFNI: software for analysis and visualization of functional magnetic resonance neuroimages. Comput Biomed Res 29: 162–173.
Critchley HD, Elliott R, Mathias CJ, Dolan RJ (2000). Neural activity relating to generation and representation of galvanic skin conductance responses: a functional magnetic resonance imaging study. J Neurosci 20: 3033–3040.
Critchley HD, Mathias CJ, Dolan RJ (2001). Neural activity in the human brain relating to uncertainty and arousal during anticipation. Neuron 29: 537–545.
Curtis VA, Bullmore ET, Morris RG, Brammer MJ, Williams SC, Simmons A et al (1999). Attenuated frontal activation in schizophrenia may be task dependent. Schizophr Res 37: 35–44.
Doricchi F, Perani D, Incoccia C, Grassi F, Cappa SF, Bettinardi V et al (1997). Neural control of fast-regular saccades and antisaccades: an investigation using positron emission tomography. Exp Brain Res 116: 50–62.
Downar J, Crawley AP, Mikulis DJ, Davis KD (2001). The effect of task relevance on the cortical response to changes in visual and auditory stimuli: an event-related fMRI study. Neuroimage 14: 1256–1267.
Egelman DM, Person C, Montague PR (1998). A computational role for dopamine delivery in human decision-making. J Cogn Neurosci 10: 623–630.
Elliott R, Dolan RJ (1998). Activation of different anterior cingulate foci in association with hypothesis testing and response selection. Neuroimage 8: 17–29.
Elliott R, Dolan RJ, Frith CD (2000). Dissociable functions in the medial and lateral orbitofrontal cortex: evidence from human neuroimaging studies. Cereb Cortex 10: 308–317.
Elliott R, Rees G, Dolan RJ (1999). Ventromedial prefrontal cortex mediates guessing. Neuropsychologia 37: 403–411.
Ernst M, Bolla K, Mouratidis M, Contoreggi C, Matochik JA, Kurian V et al (2002). Decision-making in a risk-taking task. A PET study. Neuropsychopharmacology 26: 682–691.
Feinstein A, Goldberg TE, Nowlin B, Weinberger DR (1998). Types and characteristics of remote memory impairment in schizophrenia. Schizophr Res 30: 155–163.
Fink GR, Halligan PW, Marshall JC, Frith CD, Frackowiak RS, Dolan RJ (1996). Where in the brain does visual attention select the forest and the trees? Nature 382: 626–628.
Fletcher PC, McKenna PJ, Frith CD, Grasby PM, Friston KJ, Dolan RJ (1998). Brain activations in schizophrenia during a graded memory task studied with functional neuroimaging. Arch Gen Psychiatry 55: 1001–1008.
Forman SD, Cohen JD, Fitzgerald M, Eddy WF, Mintun MA, Noll DC (1995). Improved assessment of significant activation in functional magnetic resonance imaging (fMRI): use of a cluster-size threshold. Magn Reson Med 33: 636–647.
Friston KJ (1998). The disconnection hypothesis. Schizophr Res 30: 115–125.
Garavan H, Ross TJ, Stein EA (1999). Right hemispheric dominance of inhibitory control: an event-related functional MRI study. Proc Natl Acad Sci USA 96: 8301–8306.
Garety PA, Hemsley DR, Wessely S (1991). Reasoning in deluded schizophrenic and paranoid patients. Biases in performance on a probabilistic inference task. J Nerv Ment Dis 179: 194–201.
Gehring WJ, Willoughby AR (2002). The medial frontal cortex and the rapid processing of monetary gains and losses. Science 295: 2279–2282.
Goulet LR, Barclay A (1967). Guessing behavior of normal and retarded children under two random reinforcement conditions. Child Dev 38: 545–552.
Grassberger P (1989). Estimating the information content of symbol sequences and efficient codes. IEEE Trans Inform Theory 35: 669–675.
Gur RE, Cowell PE, Latshaw A, Turetsky BI, Grossman RI, Arnold SE et al (2000). Reduced dorsal and orbital prefrontal gray matter volumes in schizophrenia. Arch Gen Psychiatry 57: 761–768.
Herzel HP, Grosse I (1995). Measuring correlations in symbol sequences. Physica A 216: 518–542.
Hopfinger JB, Buonocore MH, Mangun GR (2000). The neural mechanisms of top-down attentional control. Nat Neurosci 3: 284–291.
Kahneman D, Tversky A (1984). Choices, values, and frames. Am Psychol 39: 341–350.
Kiehl KA, Laurens KR, Duty TL, Forster BB, Liddle PF (2001). Neural sources involved in auditory target detection and novelty processing: an event-related fMRI study. Psychophysiology 38: 133–142.
Lamb MR, Robertson LC, Knight RT (1989). Attention and interference in the processing of global and local information: effects of unilateral temporal-parietal junction lesions. Neuropsychologia 27: 471–483.
Lancaster JL, Woldorff MG, Parsons LM, Liotti M, Freitas CS, Rainey L et al (2000). Automated Talairach atlas labels for functional brain mapping. Hum Brain Mapp 10: 120–131.
Lawrie SM, Buechel C, Whalley HC, Frith CD, Friston KJ, Johnstone EC (2002). Reduced frontotemporal functional connectivity in schizophrenia associated with auditory hallucinations. Biol Psychiatry 51: 1008–1011.
Lennox BR, Park SB, Medley I, Morris PG, Jones PB (2000). The functional anatomy of auditory hallucinations in schizophrenia. Psychiatry Res 100: 13–20.
Liddle PF, Kiehl KA, Smith AM (2001). Event-related fMRI study of response inhibition. Hum Brain Mapp 12: 100–109.
Ludvigson HW (1966). Response units in the prediction of simple event patterns. J Exp Psychol 72: 335–360.
Luna B, Thulborn KR, Strojwas MH, McCurtain BJ, Berman RA, Genovese CR et al (1998). Dorsal cortical regions subserving visually guided saccades in humans: an fMRI study. Cereb Cortex 8: 40–47.
Lyon N, Mejsholm B, Lyon M (1986). Stereotyped responding by schizophrenic outpatients: cross-cultural confirmation of perseverative switching on a two-choice task. J Psychiatr Res 20: 137–150.
Manoach DS, Gollub RL, Benson ES, Searl MM, Goff DC, Halpern E et al (2000). Schizophrenic subjects show aberrant fMRI activation of dorsolateral prefrontal cortex and basal ganglia during working memory performance. Biol Psychiatry 48: 99–109.
Manoach DS, Press DZ, Thangaraj V, Searl MM, Goff DC, Halpern E et al (1999). Schizophrenic subjects activate dorsolateral prefrontal cortex during a working memory task, as measured by fMRI. Biol Psychiatry 45: 1128–1137.
McCarthy G, Luby M, Gore J, Goldman-Rakic P (1997). Infrequent events transiently activate human prefrontal and parietal cortex as measured by functional MRI. J Neurophysiol 77: 1630–1634.
Menon V, Adleman NE, White CD, Glover GH, Reiss AL (2001). Error-related brain activation during a Go/NoGo response inhibition task. Hum Brain Mapp 12: 131–143.
Mesulam MM (1999). Spatial attention and neglect: parietal, frontal and cingulate contributions to the mental representation and attentional targeting of salient extrapersonal events. Philos Trans R Soc London B Biol Sci 354: 1325–1346.
Meyer-Lindenberg A, Poline JB, Kohn PD, Holt JL, Egan MF, Weinberger DR et al (2001). Evidence for abnormal cortical functional connectivity during working memory in schizophrenia. Am J Psychiatry 158: 1809–1817.
Mogg K, Mathews A, Eysenck M, May J (1991). Biased cognitive operations in anxiety: artefact, processing priorities or attentional search? Behav Res Ther 29: 459–467.
Nieman DH, Koelman JH, Linszen DH, Bour LJ, Dingemans PM, Ongerboer de Visser BW (2002). Clinical and neuropsychological correlates of the P300 in schizophrenia. Schizophr Res 55: 105–113.
Nordahl TE, Carter CS, Salo RE, Kraft L, Baldo J, Salamat S et al (2001). Anterior cingulate metabolism correlates with stroop errors in paranoid schizophrenia patients. Neuropsychopharmacology 25: 139–148.
Norusis MJ (1990). SPSS Base System User's Guide. SPSS Inc.: Chicago.
O'Doherty J, Kringelbach ML, Hornak J, Andrews C, Rolls ET (2001). Abstract reward and punishment representations in the human orbitofrontal cortex. Nat Neurosci 4: 95–102.
Paulus MP (1997). Long-range interactions in sequences of human behavior. Phys Rev E 55: 3249–3256.
Paulus MP, Geyer MA, Braff DL (1996). Use of methods from chaos theory to quantify a fundamental dysfunction in the behavioral organization of schizophrenic patients. Am J Psychiatry 153: 714–717.
Paulus MP, Geyer MA, Braff DL (1999). Long-range correlations in choice sequences of schizophrenic patients. Schizophr Res 35: 69–75.
Paulus MP, Hozack N, Frank L, Brown GG (2002a). Error rate and outcome predictability affect neural activation in prefrontal cortex and anterior cingulate during decision-making. Neuroimage 15: 836–846.
Paulus MP, Hozack N, Zauscher B, McDowell JE, Frank L, Brown GG et al (2001a). Prefrontal, parietal, and temporal cortex networks underlie decision-making in the presence of uncertainty. Neuroimage 13: 91–100.
Paulus MP, Hozack NE, Zauscher BE, Frank L, Brown GG, McDowell J et al (2002b). Parietal dysfunction is associated with increased outcome-related decision-making in schizophrenia patients. Biol Psychiatry 51: 995–1004.
Paulus MP, Rapaport MH, Braff DL (2001b). Trait contributions of complex dysregulated behavioral organization in schizophrenic patients. Biol Psychiatry 49: 71–77.
Perlstein WM, Carter CS, Noll DC, Cohen JD (2001). Relation of prefrontal cortex dysfunction to working memory and symptoms in schizophrenia. Am J Psychiatry 158: 1105–1113.
Platt ML, Glimcher PW (1999). Neural correlates of decision variables in parietal cortex. Nature 400: 233–238.
Rahman S, Sahakian BJ, Hodges JR, Rogers RD, Robbins TW (1999). Specific cognitive deficits in mild frontal variant frontotemporal dementia. Brain 122: 1469–1493.
Resnick SM (1992). Matching for education in studies of schizophrenia. Arch Gen Psychiatry 49: 246.
Rogers RD, Owen AM, Middleton HC, Williams EJ, Pickard JD, Sahakian BJ et al (1999). Choosing between small, likely rewards and large, unlikely rewards activates inferior and orbital prefrontal cortex. J Neurosci 19: 9029–9038.
Rubia K, Russell T, Bullmore ET, Soni W, Brammer MJ, Simmons A et al (2001a). An fMRI study of reduced left prefrontal activation in schizophrenia during normal inhibitory function. Schizophr Res 52: 47–55.
Rubia K, Russell T, Overmeyer S, Brammer MJ, Bullmore ET, Sharma T et al (2001b). Mapping motor inhibition: conjunctive brain activations across different versions of Go/No-Go and stop tasks. Neuroimage 13: 250–261.
Schultz SK, O'Leary DS, Boles Ponto LL, Arndt S, Magnotta V, Watkins GL et al (2002). Age and regional cerebral blood flow in schizophrenia: age effects in anterior cingulate, frontal, and parietal cortex. J Neuropsychiatry Clin Neurosci 14: 19–24.
Schultz W, Tremblay L, Hollerman JR (2000). Reward processing in primate orbitofrontal cortex and basal ganglia. Cereb Cortex 10: 272–284.
Seidman LJ, Yurgelun-Todd D, Kremen WS, Woods BT, Goldstein JM, Faraone SV et al (1994). Relationship of prefrontal and temporal lobe MRI measures to neuropsychological performance in chronic schizophrenia. Biol Psychiatry 35: 235–246.
Shadlen MN, Newsome WT (2001). Neural basis of a perceptual decision in the parietal cortex (area LIP) of the rhesus monkey. J Neurophysiol 86: 1916–1936.
Shenton ME, Dickey CC, Frumin M, McCarley RW (2001). A review of MRI findings in schizophrenia. Schizophr Res 49: 1–52.
Shergill SS, Cameron LA, Brammer MJ, Williams SC, Murray RM, McGuire PK (2001). Modality specific neural correlates of auditory and somatic hallucinations. J Neurol Neurosurg Psychiatry 71: 688–690.
Skudlarski P, Constable RT, Gore JC (1999). ROC analysis of statistical methods used in functional MRI: individual subjects. Neuroimage 9: 311–329.
Spence SA, Liddle PF, Stefan MD, Hellewell JS, Sharma T, Friston KJ et al (2000). Functional anatomy of verbal fluency in people with schizophrenia and those at genetic risk. Focal dysfunction and distributed disconnectivity reappraised. Br J Psychiatry 176: 52–60.
Spitzer RL, Williams JB, Gibbon M, First MB (1992). The structured clinical interview for DSM-III–R (SCID): I. History, rationale, and description. Arch Gen Psychiatry 49: 624–629.
Steel C, Haworth EJ, Peters E, Hemsley DR, Sharma T, Gray JA et al (2001). Neuroimaging correlates of negative priming. Neuroreport 12: 3619–3624.
Stieper DR, Ells EM, Farkas E, Caplan A (1972). The effects of reinforcement procedures upon schizophrenic patients: a survey of the data. Prog Exp Perspect Res 6: 149–242.
Talairach J, Tournoux P (1988). Co-planar stereotaxic atlas of the human brain: a 3-dimensional proportional system, an approach to cerebral imaging. Thieme Medical Publishers: Stuttgart, New York.
Tranel D, Damasio H (1994). Neuroanatomical correlates of electrodermal skin conductance responses. Psychophysiology 31: 427–438.
Tversky A, Kahneman D (1981). The framing of decisions and the psychology of choice. Science 211: 453–458.
Volz H, Gaser C, Hager F, Rzanny R, Ponisch J, Mentzel H et al (1999). Decreased frontal activation in schizophrenics during stimulation with the continuous performance test—a functional magnetic resonance imaging study. Eur Psychiatry 14: 17–24.
Volz HP, Nenadic I, Gaser C, Rammsayer T, Hager F, Sauer H (2001). Time estimation in schizophrenia: an fMRI study at adjusted levels of difficulty. Neuroreport 12: 313–316.
Weinberger DR, Berman KF, Suddath R, Torrey EF (1992). Evidence of dysfunction of a prefrontal-limbic network in schizophrenia: a magnetic resonance imaging and regional cerebral blood flow study of discordant monozygotic twins. Am J Psychiatry 149: 890–897.
Weinberger DR, Mattay V, Callicott J, Kotrla K, Santha A, van Gelderen P et al (1996). fMRI applications in schizophrenia research. Neuroimage 4: S118–S126.
Acknowledgements
We acknowledge the invaluable help of Nikki Hozack in conducting the experiments and Kelly Winternheimer in editing the manuscript. This work was supported by grants from NIMH (MH R37-42228, DLB), NARSAD (MPP), and support from the Veterans Administration via a VISN 22 MIRECC and a Merit Grant (MPP).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Paulus, M., Frank, L., Brown, G. et al. Schizophrenia Subjects Show Intact Success-Related Neural Activation but Impaired Uncertainty Processing during Decision-Making. Neuropsychopharmacol 28, 795–806 (2003). https://doi.org/10.1038/sj.npp.1300108
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.npp.1300108
This article is cited by
-
Abnormal Frontostriatal Activity During Unexpected Reward Receipt in Depression and Schizophrenia: Relationship to Anhedonia
Neuropsychopharmacology (2016)