Abstract
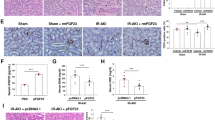
During acute rejection leukocyte–endothelial cell interaction fuelled by costimulatory molecules such as the CD40/CD154 receptor/ligand dyad disrupts microcirculation of the small bowel. Downregulating endothelial CD40 expression by employing a decoy oligonucleotide (dODN) neutralizing the transcription factor signal transducer and activator of transcription-1 (STAT-1) may protect the graft. Therefore allogenic small bowel transplantation was performed in the Brown Norway to Lewis rat model. Graft vessels were pretreated with STAT-1 dODN, mutant control ODN (20 μ M) or vehicle (n=8). CD40 antisense ODN and scrambled control ODN-treated transplants served as target control (n=3 each). Intravital microscopy, histology, immunohistochemistry and Western blot analyses were performed 7 days later. Functional capillary density, red blood cell velocity and perfusion index in STAT-1 dODN and CD40 antisense ODN-treated transplants were improved whereas stasis index was reduced. Leukocyte–endothelial cell interaction showed no difference. Histological parameters of rejection, infiltrating CD3-positive cells and apoptotic bodies were also reduced in STAT-1 dODN and CD40 antisense ODN-treated transplants 7 days post-transplantation. CD40 protein abundance was reduced to less than 10% of control in STAT-1 dODN-treated grafts. STAT-1 dODN blockade of CD40 expression improves mucosal perfusion, reduces graft rejection, T-cell infiltration and apoptosis in rat small bowel allografts during acute rejection.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Grant D . Intestinal transplantation: 1997 Report of the Intestinal Transplant Registry. Transplantation 1999; 67: 1061–1064.
Cho CS, Hamawy MM, Knechtle SJ . CD40-CD154 interactions and allograft survival. Curr Opin Organ Transplant 2000; 5: 10–15.
Rose ML . Role of endothelial cells in allograft rejection. Vasc Med 1997; 2: 105–114.
Rocha PN, Plumb TJ, Crowley SD, Coffman TM . Effector mechanisms in transplant rejection. Immunological Reviews 2003; 196: 51–64.
Stojanovic T, Bedke J, Grone HJ, Proudfoot AE, Becker H, Markus P et al. Met-RANTES inhibition of mucosal perfusion failure in acute intestinal transplant rejection – role of endothelial cell–leukocyte interaction. J Vasc Res 2002; 39: 51–58.
Van Kooten C, Bancheraeu J . CD40-CD40 ligand. J Leukoc Biol 2000; 67: 2–17.
Schoenbeck U, Libby P . The CD40/CD154 receptor/ligand dyad. Cell Mol Life Sci 2001; 58: 4–43.
Hollenbaugh D, Mischel-Petty N, Edwards CP, Simon JC, Denfeld RW, Kiener PA et al. Expression of functional CD40 by vascular endothelial cells. J Exp Med 1995; 182: 33–40.
Yellin MJ, Brett J, Baum D, Matsushima A, Szabolcs M, Stern D et al. Functional interactions of T-cells with endothelial cells: the role of CD40L-CD40-mediated signals. J Exp Med 1995; 182: 1857–1864.
Lienenlüke B, Germann T, Kroczek RA, Hecker M . CD154 stimulation of interleukin-12 synthesis in human endothelial cells. Eur J Immunol 2000; 30: 2864–2870.
Ode-Hakim S, Docke WD, Kern F, Emmrich F, Volk HD, Reinke P . Delayed-type hypersensitivity-like mechanisms dominate late acute rejection episodes in renal allograft recipients. Transplantation 1996; 61: 1233–1240.
D'Elios MM, Josien R, Manghetti M, Amedei A, de Carli M, Cuturi MC et al. Predominant Th1 cell infiltration in acute rejection episodes of human kidney grafts. Kidney Int 1997; 51: 1876–1884.
Kirk AD, Burkly LC, Batty DS, Baumgartner RE, Berning JD, Buchanan K et al. Treatment with humanized monoclonal antibody against CD154 prevents acute renal allograft rejection in nonhuman primates. Nat Med 1999; 5: 686–693.
Hancock WW . Current trends in transplant immunology. Curr Opin Nephrol Hypertens 1999; 8: 317–324.
Gudmundsdottir H, Turka LA . T cell costimulatory blockade: new therapies for transplant rejection. J Am Soc Nephrol 1999; 10: 1356–1365.
Waldmann H . Transplantation tolerance – where do we stand? Nat Med 1999; 5: 1245–1248.
Kawai T, Andrews D, Colvin RB, Sachs DH, Cosimi AB . Thromboembolic complications after treatment with monoclonal antibody against CD40 ligand. Nat Med 2000; 6: 114.
Koyama I, Kawai T, Andrews D, Boskovic S, Nadazdin O, Wee SL et al. Thrombophilia associated with anti-CD154 monoclonal antibody treatment and its prophylaxis in nonhuman primates. Transplantation 2004; 77: 460–462.
Andre P, Prasad KS, Denis CV, He M, Papalia, Hynes RO et al. CD40L stabilizes arterial thrombi by a beta3 intergrin-dependent mechanism. Nat Med 2002; 8: 247–252.
Andre P, Prasad KS, Denis CV, He M, Papalia JM, Hynes RO et al. Differential regulation of CD40-mediated human B cell responses by antibodies directed against different CD40 epitopes. Cell Immunol 2000; 201: 109–123.
Mann MJ, Dzau VJ . Therapeutic applications of transcription factor decoy oligonucleotides. J Clin Invest 2000; 106: 1071–1075.
Krzesz R, Wagner AH, Cattaruzza M, Hecker M . Cytokine-inducible CD40 gene expression in vascular smooth muscle cells is mediated by nuclear factor B and signal transducer and activatior of transcription-1. FEBS Lett 1999; 453: 191–196.
Wagner AH, Gebauer M, Pollok-Kopp B, Hecker M . Cytokine-inducible CD40 expression in human endothelial cells is mediated by interferon regulatory factor-1. Blood 2002; 99: 520–525.
Gao D, Wagner AH, Fankhaenel S, Stojanovic T, Schweyer S, Panzner S et al. CD40 antisense oligonucleotide inhibition of trinitrobenzene sulphonic acid induced rat colitis. Gut 2005; 54: 70–77.
Caux C, Massacrier C, Vanbervliet B, Dubois B, Van Kooten C, Durand I et al. Activation of human dendritic cells through CD40 cross-linking. J Exp Med 1994; 180: 1263–1272.
Karmann K, Hughes CC, Schechner J, Fanslow WC, Pober JS . CD40 on human endothelial cells: inducibility by cytokines and functional regulation of adhesion molecule expression. Proc Natl Acad Sci USA 1995; 92: 4342–4356.
Hölschermann H, Stadlbauer THW, Wagner AH, Fingerhuth H, Muth H, Rong S et al. STAT-1 and AP-1 decoy oligonucleotide therapy delays acute rejection and prolongs cardiac allograft survival. Cardiovasc Res 2006; 71: 527–536.
Neish AS, Read MA, Thanos D, Pine R, Maniatis T, Collins T . Endothelial interferon regulatory factor 1 cooperates with NF-kappa B as a transcriptional activator of vascular cell adhesion molecule 1. Mol Cell Biol 1995; 15: 2558–2569.
De Caterina R, Bourcier T, Laufs U, La Fata V, Lazzerini G, Neish AS et al. Induction of endothelial-leukocyte interaction by interferon-gamma requires coactivation of nuclear factor-kappaB. Arterioscler Thromb Vasc Biol 2001; 21: 227–232.
Menger MD, Pelikan S, Steiner D, Messmer K . Microvascular ischemia-reperfusion injury in striated muscle: significance of ‘reflow paradox’. Am J Physiol 1992; 263: H1901–1906.
Monchik GJ, Russel PS . Transplantation of small bowel in the rat: technical and immunological consideration. Surgery 1971; 70: 693–703.
Post S, Menger MD, Rentsch M, Messmer K . The impact of arterialization on hepatic microcirculation and leukocyte accumulation after liver transplantation in the rat. Transplantation 1992; 54: 789–794.
Gonzalez AP, Sepulveda S, Massberg S, Baumeister R, Menger MD . In vivo fluorescence microscopy for the assessment of microvascular reperfusion injury in small bowel transplants in rats. Transplantation 1994; 58: 403–408.
Zeintl H, Sack FU, Intaglietta M, Messmer K . Computer assisted leukocyte adhesion measurement in intravital microscopy. Int J Microcirc Clin Exp 1989; 8: 293–302.
Wu T, Abu-Elmagd K, Bond G, Nalesnik MA, Randhawa P, Demetris AJ . Schema for histologic grading of small intestine allograft acute rejection. Transplantation 2003; 75: 1241–1248.
Acknowledgements
This work was supported by the Deutsche Forschungsgemeinschaft (SFB 405 project B17). The expert technical assistance of Annette Bennemann is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Stojanovic, T., Scheele, L., Wagner, A. et al. STAT-1 decoy oligonucleotide improves microcirculation and reduces acute rejection in allogeneic rat small bowel transplants. Gene Ther 14, 883–890 (2007). https://doi.org/10.1038/sj.gt.3302931
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302931