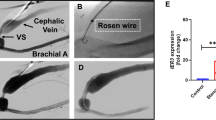
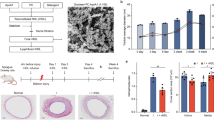
Abstract
Endothelial cell loss is a critical event in the pathological repair of the injured blood vessel. Impaired endothelial function results in reduced production of key vascular mediators such as nitric oxide (NO) within the vessel wall leading to enhanced smooth muscle cell proliferation and migration and ultimately intimal hyperplasia. The aim of the present study was to directly compare the effects of adenoviral-mediated gene delivery of two nitric oxide synthase (NOS) isoforms, eNOS and iNOS on endothelial regeneration and intimal hyperplasia following endothelial injury in the rabbit carotid artery. The right carotid arteries of male New Zealand white rabbits were denuded by passing a 3French Fogarty balloon catheter along the artery three times. In all, 1 × 109 PFU of adenoviral(Ad)eNOS, AdiNOS or Adβ-galactosidase (Adβ-Gal) was then delivered intraluminally and allowed to dwell for 20 min. Transgene expression was sought after 3 days by immunohistochemistry and at 7 days by quantitative reverse transcriptase PCR. The effect on intimal hyperplasia was sought using histological staining after 14 days. Evans blue staining was used to determine the effect on endothelial regeneration. eNOS and iNOS expression was detected in transduced arteries. Neointima/media ratios were significantly reduced in eNOS (0.07±0.044) and iNOS (0.087±0.086) transduced arteries compared with Adβ-Gal (0.332±0.14) transduced arteries (n=7). In addition, AdeNOS treatment (4.21±3.12% de-endothelialized area) enhanced endothelial regeneration compared to Adβ-Gal treatment (10.05±4.98), while treatment with AdiNOS (25.17±11.92) inhibited endothelial regeneration in the injured rabbit carotid artery (n=7–8). These results highlight the potential of NOS gene therapy, in particular, eNOS gene therapy as a potential therapeutic strategy for the prevention of restenosis after vascular injury.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others

References
Rutanen J, Puhakka H, Yla-Herttuala S . Post-intervention vessel remodeling. Gene Therapy 2002; 9: 1487–1491.
Pauletto P, Sartore S, Pessina AC . Smooth-muscle-cell proliferation and differentiation in neointima formation and vascular restenosis. Clin Sci (London) 1994; 87: 467–479.
Lafont A, Durand E, Samuel JL, Besse B, Addad F, Levy BI et al. Endothelial dysfunction and collagen accumulation: two independent factors for restenosis and constrictive remodeling after experimental angioplasty. Circulation 1999; 100: 1109–1115.
Kong D, Melo LG, Gnecchi M, Zhang L, Mostoslavsky G, Liew CC et al. Cytokine-induced mobilization of circulating endothelial progenitor cells enhances repair of injured arteries. Circulation 2004; 110: 2039–2046.
Losordo DW, Isner JM, Diaz-Sandoval LJ . Endothelial recovery: the next target in restenosis prevention. Circulation 2003; 107: 2635–2637.
Mellion BT, Ignarro LJ, Ohlstein EH, Pontecorvo EG, Hyman AL, Kadowitz PJ . Evidence for the inhibitory role of guanosine 3′, 5′-monophosphate in ADP-induced human platelet aggregation in the presence of nitric oxide and related vasodilators. Blood 1981; 57: 946–955.
Kubes P, Suzuki M, Granger DN . Nitric oxide: an endogenous modulator of leukocyte adhesion. Proc Natl Acad Sci USA 1991; 88: 4651–4655.
Garg UC, Hassid A . Nitric oxide-generating vasodilators and 8-bromo-cyclic guanosine monophosphate inhibit mitogenesis and proliferation of cultured rat vascular smooth muscle cells. J Clin Invest 1989; 83: 1774–1777.
Sarkar R, Meinberg EG, Stanley JC, Gordon D, Webb RC . Nitric oxide reversibly inhibits the migration of cultured vascular smooth muscle cells. Circ Res 1996; 78: 225–230.
Chen AF, O'Brien T, Katusic ZS . Transfer and expression of recombinant nitric oxide synthase genes in the cardiovascular system. Trends Pharmacol Sci 1998; 19: 276–286.
Khurana VG, Smith LA, Baker TA, Eguchi D, O'Brien T, Katusic ZS . Protective vasomotor effects of in vivo recombinant endothelial nitric oxide synthase gene expression in a canine model of cerebral vasospasm. Stroke 2002; 33: 782–789.
Sato J, Mohacsi T, Noel A, Jost C, Gloviczki P, Mozes G et al. In vivo gene transfer of endothelial nitric oxide synthase to carotid arteries from hypercholesterolemic rabbits enhances endothelium-dependent relaxations. Stroke 2000; 31: 968–975.
Zanetti M, Sato J, Katusic ZS, O'Brien T . Gene transfer of endothelial nitric oxide synthase alters endothelium-dependent relaxations in aortas from diabetic rabbits. Diabetologia 2000; 43: 340–347.
Mozes G, Kullo IJ, Mohacsi TG, Cable DG, Spector DJ, Crotty TB et al. Ex vivo gene transfer of endothelial nitric oxide synthase to atherosclerotic rabbit aortic rings improves relaxations to acetylcholine. Atherosclerosis 1998; 141: 265–271.
Cable DG, Pompili VJ, O'Brien T, Schaff HV . Recombinant gene transfer of endothelial nitric oxide synthase augments coronary artery relaxations during hypoxia. Circulation 1999; 100: II335–II339.
Onoue H, Tsutsui M, Smith L, Stelter A, O'Brien T, Katusic ZS . Expression and function of recombinant endothelial nitric oxide synthase gene in canine basilar artery after experimental subarachnoid hemorrhage. Stroke 1998; 29: 1959–1965; discussion 1965–1956.
Laukkanen MO, Kivela A, Rissanen T, Rutanen J, Karkkainen MK, Leppanen O et al. Adenovirus-mediated extracellular superoxide dismutase gene therapy reduces neointima formation in balloon-denuded rabbit aorta. Circulation 2002; 106: 1999–2003.
Pels K, Deiner C, Coupland SE, Noutsias M, Sutter AP, Schultheiss HP et al. Effect of adventitial VEGF(165) gene transfer on vascular thickening after coronary artery balloon injury. Cardiovasc Res 2003; 60: 664–672.
Leppanen O, Rutanen J, Hiltunen MO, Rissanen TT, Turunen MP, Sjoblom T et al. Oral imatinib mesylate (STI571/gleevec) improves the efficacy of local intravascular vascular endothelial growth factor-C gene transfer in reducing neointimal growth in hypercholesterolemic rabbits. Circulation 2004; 109: 1140–1146.
Turunen MP, Puhakka HL, Koponen JK, Hiltunen MO, Rutanen J, Leppanen O et al. Peptide-retargeted adenovirus encoding a tissue inhibitor of metalloproteinase-1 decreases restenosis after intravascular gene transfer. Mol Ther 2002; 6: 306–312.
Kullo IJ, Schwartz RS, Pompili VJ, Tsutsui M, Milstien S, Fitzpatrick LA et al. Expression and function of recombinant endothelial NO synthase in coronary artery smooth muscle cells. Arterioscler Thromb Vasc Biol 1997; 17: 2405–2412.
Sato J, Nair K, Hiddinga J, Eberhardt NL, Fitzpatrick LA, Katusic ZS et al. eNOS gene transfer to vascular smooth muscle cells inhibits cell proliferation via upregulation of p27 and p21 and not apoptosis. Cardiovasc Res 2000; 47: 697–706.
Kibbe MR, Li J, Nie S, Watkins SC, Lizonova A, Kovesdi I et al. Inducible nitric oxide synthase (iNOS) expression upregulates p21 and inhibits vascular smooth muscle cell proliferation through p42/44 mitogen-activated protein kinase activation and independent of p53 and cyclic guanosine monophosphate. J Vasc Surg 2000; 31: 1214–1228.
von der Leyen HE, Gibbons GH, Morishita R, Lewis NP, Zhang L, Nakajima M et al. Gene therapy inhibiting neointimal vascular lesion: in vivo transfer of endothelial cell nitric oxide synthase gene. Proc Natl Acad Sci USA 1995; 92: 1137–1141.
Janssens S, Flaherty D, Nong Z, Varenne O, van Pelt N, Haustermans C et al. Human endothelial nitric oxide synthase gene transfer inhibits vascular smooth muscle cell proliferation and neointima formation after balloon injury in rats. Circulation 1998; 97: 1274–1281.
Varenne O, Pislaru S, Gillijns H, Van Pelt N, Gerard RD, Zoldhelyi P et al. Local adenovirus-mediated transfer of human endothelial nitric oxide synthase reduces luminal narrowing after coronary angioplasty in pigs. Circulation 1998; 98: 919–926.
Shears II LL, Kibbe MR, Murdock AD, Billiar TR, Lizonova A, Kovesdi I et al. Efficient inhibition of intimal hyperplasia by adenovirus-mediated inducible nitric oxide synthase gene transfer to rats and pigs in vivo. J Am Coll Surg 1998; 187: 295–306.
Kong D, Melo LG, Mangi AA, Zhang L, Lopez-Ilasaca M, Perrella MA et al. Enhanced inhibition of neointimal hyperplasia by genetically engineered endothelial progenitor cells. Circulation 2004; 109: 1769–1775.
He T, Smith LA, Harrington S, Nath KA, Caplice NM, Katusic ZS . Transplantation of circulating endothelial progenitor cells restores endothelial function of denuded rabbit carotid arteries. Stroke 2004; 35: 2378–2384.
Michel T, Feron O . Nitric oxide synthases: which, where, how, and why? J Clin Invest 1997; 100: 2146–2152.
Zanetti M, d'Uscio LV, Kovesdi I, Katusic ZS, O'Brien T . In vivo gene transfer of inducible nitric oxide synthase to carotid arteries from hypercholesterolemic rabbits. Stroke 2003; 34: 1293–1298.
Six I, Van Belle E, Bordet R, Corseaux D, Callebert J, Dupuis B et al. L-Arginine and L-NAME have no effect on the reendothelialization process after arterial balloon injury. Cardiovasc Res 1999; 43: 731–738.
Kennedy S, Preston AA, McPhaden AR, Miller AM, Wainwright CL, Wadsworth RM . Correlation of changes in nitric oxide synthase, superoxide dismutase and nitrotyrosine with endothelial regeneration and neointimal hyperplasia in the balloon-injured rabbit subclavian artery. Coron Artery Dis 2004; 15: 337–346.
Leite PF, Danilovic A, Moriel P, Dantas K, Marklund S, Dantas APV et al. Sustained decrease in superoxide dismutase activity underlies constrictive remodeling after balloon injury in rabbits. Arterioscler Thromb Vasc Biol 2003; 23: 2197–2202.
Livak KJ, Schmittgen TD . Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) method. Methods 2001; 25: 402–408.
Lund CO, Mortensen A, NIlas L, Breinholt VM, Larsen JJ, Ottesen B . Estrogen and phytoestrogens: effect on eNOS expression and in vitro vasodilation in cerebral arteries in ovariectomized Watanabe hertibale hyperlipidemic rabbits. Eur J Obstet Gynecol Reprod Biol 2006, (E-pub ahead of print).
Acknowledgements
This work was funded by the Health Research Board of Ireland. We thank Tina Harte and Aoife Duffy for technical assistance. TOB is supported by a Centre for Science Engineering and Technology award from Science Foundation Ireland.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cooney, R., Hynes, S., Sharif, F. et al. Effect of gene delivery of NOS isoforms on intimal hyperplasia and endothelial regeneration after balloon injury. Gene Ther 14, 396–404 (2007). https://doi.org/10.1038/sj.gt.3302882
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302882
Keywords
This article is cited by
-
Intraprocedural endothelial cell seeding of arterial stents via biotin/avidin targeting mitigates in-stent restenosis
Scientific Reports (2022)
-
Gene-eluting stents: non-viral, liposome-based gene delivery of eNOS to the blood vessel wall in vivo results in enhanced endothelialization but does not reduce restenosis in a hypercholesterolemic model
Gene Therapy (2012)
-
Non-viral eNOS gene delivery and transfection with stents for the treatment of restenosis
BioMedical Engineering OnLine (2010)
-
Therapeutisches Potenzial der Stickstoffmonoxid-Synthase-Isoformen in Restenose, Transplantat- und Bypassarteriosklerose
Gefässchirurgie (2009)
-
Gene-eluting Stents: Adenovirus-mediated Delivery of eNOS to the Blood Vessel Wall Accelerates Re-endothelialization and Inhibits Restenosis
Molecular Therapy (2008)