Abstract
Trait-based approaches are increasingly relevant to understand ecological and evolutionary patterns. A comprehensive trait database for extant reef corals is already available and widely used to reveal vulnerabilities to environmental disturbances including climate change. However, the lack of similar trait compilations for extinct reef builders prevents the derivation of generalities from the fossil record and to address similar questions. Here we present the Ancient Reef Traits Database (ARTD), which aims to compile trait information of various reef-building organisms in one single repository. ARTD contains specimen-level data from both published and unpublished resources. In this first version, we release 15 traits for 505 genera and 1129 species, comprising a dataset of 17,841 trait values of Triassic to mid-Holocene scleractinian corals, the dominant reef-builders in the modern ocean. Other trait data, including for other reef-building organisms, are currently being collated.
Measurement(s) | fossil coral traits |
Technology Type(s) | digital curation |
Sample Characteristic - Organism | Scleractinian corals |
Sample Characteristic - Location | Global |
Similar content being viewed by others
Background & Summary
Trait-based approaches are one way to correlate different characteristics, or traits, of organisms to environmental changes with the ultimate goal to predict community and whole-ecosystem responses to such changes. Traits of reef corals have been explored to identify their life-history strategies1 and extinction risk2,3. Several palaeontological studies have examined trait-based extinction using the fossil record4,5,6. Changes in ancient reef ecosystems can provide valuable insights into drivers of reef proliferation and decline. Understanding why fossil reefs collapsed and how they recovered may help to inform conservation activities in modern coral reefs in light of anthropogenic climate change. Many reefs of the Phanerozoic eon (the last 540 million years) can be considered analogues of modern tropical coral reefs with regards to reef architecture and environmental controls7. Phanerozoic reef-building is not restricted to scleractinian corals but also a multitude of other sessile hypercalcifying animals (animals with a large skeletal to biomass ratio) such as extinct coral clades, calcifying sponges and rudist bivalves.
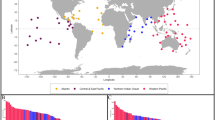
In the first release of this database, we focus on scleractinian corals, which have a rich fossil record from the Middle Triassic (~245 Ma) and became the dominant reef builders in the Late Triassic (~225 million years ago)8. Scleractinian corals have an extensive fossil record due to their calcified skeletons. Accordingly, morphological traits are well preserved and several of those traits have been shown to be linked to species extinction risk: examples are corallite integration, corallite diameter, growth rate (linked to morphology or measured directly from growth bands), and colony longevity (linked to size)3,9,10,11,12. Spatial and environmental traits are also important predictors of extinction risk: habitat breadth and maximum water depth are two key parameters linked to climate change vulnerability in corals9.
The traits of extant reef corals have been compiled in the openly accessible Coral Traits Database (CTD)13,14, which in addition to morphological traits, also contains data on physiology, biology and reproduction. However, there is no single, exhaustive resource for fossil reef building organisms including extinct scleractinian corals and the traits of extinct corals are currently scattered in online repositories and research publications. Some trait compilations that cover only one type of trait (i.e., corallite integration) and/or span one period (i.e., Triassic) have previously been published15 but are not openly available. The online resource Corallosphere (www.corallosphere.org) contains a description of genus-specific traits for both extinct and extant scleractinian corals, while the Paleobiology Database (PBDB; www.paleobiodb.org) contains information for a limited number of traits such as inferred symbiotic status or preferred environment. The database on Neogene Marine Biota of Tropical America (NMITA) mostly contains trait information of Pleistocene to Holocene scleractinian corals from the Caribbean region16,17. Therefore, there is a need for an updated, comprehensive and openly accessible compilation of fossil coral traits.
Here, we present the Ancient Reef Traits Database (ARTD) which fills this gap. ARTD is a unique, specimen-based compilation of reef builders’ traits. At the initial stage, the database contains 15 traits of scleractinian corals and covers the time period from the mid-Triassic until the mid-Holocene. The value of ARTD lies not just in its data coverage but in its interlinkage with other databases. ARTD is compatible with CTD and is also designed in a way that provides easy integration with the biggest resource of fossil occurrence data, namely the PBDB.
Methods
ARTD has a similar structure as the CTD and is designed to contain specimen-level traits for identified species and genera (Fig. 1). The basic unit of entry is that of the trait of a single specimen, e.g. a coral specimen, that is also accompanied by contextual characteristics such as the geological stage(s), the present-day geographic region and the present-day coordinates of the locality in decimal degrees, in which the specimen was found. Reconstructed palaeo-coordinates were computed from the present-day coordinates using the rotation file supplied by C. Scotese in the ‘PALEOMAP PaleoAtlas for GPlates’ package18. These metadata are crucial for analyses of traits in a spatial, temporal and environmental context. For example, the corallite diameter or colony size of a species may vary due to genealogical trends or environmental factors19. A specimen can be linked to a number of observed traits as per the source of the data. In the case where there are multiple observations for a specimen or group of specimens (usually reported as such in the primary source), text-based information is entered and separated by “-“, e.g. trabecular-substyliform for columella structure. We also employ a hierarchical taxonomic structure where any inheritable trait (i.e. symbiotic status or corallite integration) of a taxon, is automatically applied to all the lower taxonomic levels and specimens of that particular taxon. For example, if a genus’ corallite integration is qualified as cerioid all its containing species and specimens are also assigned to a cerioid trait, unless exceptions are known. In the case of growth forms, heritability was assigned only to genera where one growth form has been assigned, e.g. Isastraea (massive) as growth forms tend to vary among some groups due to factors such as environment20.
The taxonomic and contextual information are stored separately and linked to the “observation” table where each entry is provided with a unique identification number (id; Fig. 1). This observation table contains the respective specimen-level traits which contains information of the trait(s) being entered, the value (numeric or character depending on the trait being entered), the value type (whether raw, aggregated, or based on the opinion of experts), and the method used to obtain the measurements (e.g. observations made in the field or laboratory). This structure is used so that multiple specimens from one specific site and one taxon can be linked several times to the same traits. For example, published articles provide several measurements of corallite widths21. In the case when a range of values is provided for a measured trait, the mean value is entered, except for the traits “corallite width minimum” and “corallite width maximum” where the minimum and maximum values are entered, respectively. Traits are divided using the categories that are comparable to the CTD: morphological, reproductive, or physiological. However, it is clear that only morphological traits are directly accessible in fossils, whereas the latter categories need to be inferred. Currently the only inferred trait in our database is that of symbiotic status (trait_name: “Zooxanthellate”) which relies on morphological criteria of extinct coral species as defined by Kiessling and Kocsis22. The original source of the entered data is also included in the database (called the primary source), and if available, the reference number for this primary source in the PBDB which can then be used to integrate both databases. The taxonomic information is automatically validated against the PBDB before each database release to ensure that the most up-to-date information is available.
Data Records
Here, we provide a release of 15 traits of 505 genera and 1129 species, comprising a dataset of 17,841 trait values obtained from localities around the world (Fig. 2). This first release has broad taxonomic and temporal coverage and comprises more than 70% of all coral genera in the PBDB in each geological stage since the mid-Triassic (Figs. 2, 3a). The availability of traits per specimen or taxon also varies (Fig. 3b) depending for example on the preservation of the specimen being reported in the primary source, with some traits such as corallite diameter or corallite integration more readily available than others such as colony size (Fig. 3c).
A static release of the database is available directly from the ART platform (https://art.nat.fau.de) as well as Zenodo23. The data release is in the form of a compressed folder containing two files:
-
1.
data.csv: A csv-formatted file containing the contextual information and measurement of specimens.
-
2.
references.csv: A csv-formatted file containing the bibliographic information of data sources24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94,95,96,97,98,99,100,101,102,103,104,105,106,107,108,109,110,111,112,113,114.
The details and descriptions of the available trait variables are available in Table 1. Up-to-date data are directly available from the database. However, as data entry and validation (see Technical Validation) is ongoing, users are recommended to use the data made available through the static releases to maximise reproducibility of analyses and results. Both the static releases23 and direct downloads are accompanied by the primary sources24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94,95,96,97,98,99,100,101,102,103,104,105,106,107,108,109,110,111,112,113,114 which should be credited.
All data in the ARTD and included in this release are linked with published (e.g. peer-reviewed papers, taxonomic monographs, books) references24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94,95,96,97,98,99,100,101,102,103,104,105,106,107,108,109,110,111,112,113,114. The final dataset consists of one row per trait for each specimen entered. Each specimen is given a unique identification number (observation_id) which can be associated with various traits. The geographic data is available for each specimen, such as the country or region in which it was found (location_name), present-day coordinates (longitude, latitude), reconstructed palaeo-coordinates (paleolng, paleolat), and taxonomic information (identified_name: as entered in the database, accepted_name: based on updated taxonomic information, genus_name: genus of the specimen as per the updated taxonomic information, species_name: the species name as per the updated taxonomic information). The time period identified for the specimen (early_interval: the first interval in which the specimen was found, late_interval: the last interval in which it was found, min: the minimum identified age, max: the maximum identified age) is also provided. For each specimen, the available traits are entered. Each trait (trait_name) is assigned to a category (trait_class), and a trait entry for a specimen contains the trait value and unit (standard_unit) if applicable. Additional information (value_type) about any measurements such as whether the entered measurement is raw or an aggregated value (mean, minimum, maximum) based on expert opinion, model-derived, the unit for the measurements (standard_unit), and the methodology used to obtain the data (methodology_name) is also provided if available. Each entry contains the identification number of the data source (reference_no), whose bibliographic details are provided in the reference.csv table, which can then be used for citations purposes.
Technical Validation
The database is curated by the managerial board who undertake the tasks below. Curation and quality control of the data include:
Updating taxonomic information
All taxonomic data are cross validated taxonomic names against the PBDB to ensure that reliable taxonomic data is available.
Dealing with duplicates
All duplicated records identified based on the combination of data source, taxonomic information, location, time period, and measurement values are flagged and then removed.
Applying inherited traits
Any newly entered traits that are identified as inherited traits are applied to all species of the genus that contains that particular trait.
Standardising text-based fields
Text-based fields such as corallite integration or growth form are standardised (Table 1) to facilitate analysis on these traits.
Contributor approval
Anyone wishing to contribute to the database should become a formal contributor and any observations entered by them will be associated with their user account.
The database is hosted on the server of the Friedrich-Alexander-Universität Erlangen-Nürnberg (FAU) and will be maintained on the long-term by WK, MCK and other staff members of the FAU.
Usage Notes
ARTD offers many new opportunities for incorporating trait-based approaches in addressing macroevolutionary and macroecological questions using the fossil record of reef-building organisms. The correlation of certain traits with environmental conditions and their vulnerability or resilience to environmental change can be widely used to understand the evolution of these organisms and the reefs they build over time, their extinction risk in light of global warming and recovery after such an event. A trait-based framework allows the analysis of variation and evolution of traits within and across reef-building organisms over time and their responses to environmental change in the past. For example, the data from ARTD combined with the PBDB show that the diversity trajectories for colonial and solitary scleractinian corals were different (Spearman’s ρ = 0.241, p = 0.129 of first differences), with colonial corals showing an increase in diversity during the late Jurassic and Cretaceous but declining again in the Late Cretaceous (Fig. 4). On the other hand, the diversity of solitary corals remained relatively low compared to colonial corals.
Trait-based frameworks are also widely used in modern ecology to study the climate impacts of organisms and their extinction risk1,14,115,116,117,118. Focusing on such frameworks would therefore allow the integration of palaeontological and neontological data using similar concepts and methods to address urgent questions on biodiversity and extinction at multiple scales3,119,120. Such an integrated approach might contribute to the conservation of modern coral reefs, which are the most threatened ecosystems from climate change121.
Code availability
All the code used to generate the figures in this manuscript is available on the following GitHub repository: https://github.com/nussaibahrs/ARTD.
Change history
02 September 2022
A Correction to this paper has been published: https://doi.org/10.1038/s41597-022-01646-2
References
Darling, E. S., Alvarez-Filip, L., Oliver, T. A., McClanahan, T. R. & Côté, I. M. Evaluating life-history strategies of reef corals from species traits. Ecol. Lett. 15, 1378–1386 (2012).
Bridge, T. C. L. et al. Incongruence between life-history traits and conservation status in reef corals. Coral Reefs 39, 271–279 (2020).
Raja, N. B. et al. Morphological traits of reef corals predict extinction risk but not conservation status. Glob. Ecol. Biogeogr. 30, 1597–1608 (2021).
Orzechowski, E. A. et al. Marine extinction risk shaped by trait–environment interactions over 500 million years. Glob. Change Biol. 21, 3595–3607 (2015).
Pietsch, C., Mata, S. A. & Bottjer, D. J. High temperature and low oxygen perturbations drive contrasting benthic recovery dynamics following the end-Permian mass extinction. Palaeogeogr. Palaeoclimatol. Palaeoecol. 399, 98–113 (2014).
Wagner, P. J. & Estabrook, G. F. Trait-based diversification shifts reflect differential extinction among fossil taxa. Proc. Natl. Acad. Sci. 111, 16419–16424 (2014).
Kiessling, W. Geologic and Biologic Controls on the Evolution of Reefs. Annu. Rev. Ecol. Evol. Syst. 40, 173–192 (2009).
Kiessling, W. Reef expansion during the Triassic: Spread of photosymbiosis balancing climatic cooling. Palaeogeogr. Palaeoclimatol. Palaeoecol. 290, 11–19 (2010).
Foden, W. B. et al. Identifying the World’s Most Climate Change Vulnerable Species: A Systematic Trait-Based Assessment of all Birds, Amphibians and Corals. PLOS ONE 8, e65427 (2013).
Hughes, A. D. & Grottoli, A. G. Heterotrophic Compensation: A Possible Mechanism for Resilience of Coral Reefs to Global Warming or a Sign of Prolonged Stress? PLOS ONE 8, e81172 (2013).
Stanley, G. D. Jr & Helmle, K. P. Middle Triassic Coral Growth Bands and Their Implication for Photosymbiosis. PALAIOS 25, 754–763 (2010).
van Woesik, R. et al. Hosts of the Plio-Pleistocene past reflect modern-day coral vulnerability. Proc. R. Soc. B Biol. Sci. 279, 2448–2456 (2012).
Madin, J. S. et al. The Coral Trait Database, a curated database of trait information for coral species from the global oceans. Sci. Data 3, 160017 (2016).
Madin, J. S. et al. A Trait-Based Approach to Advance Coral Reef Science. Trends Ecol. Evol. 31, 419–428 (2016).
Riedel, P. Korallen in der Trias der Tethys:. Stratigraphische Reichweiten, Diversitätsmuster, Entwicklungstrends und Bedeutung als Rifforganismen. Mitteilungen Ges. Geol.- Bergbaustud. Österr. 37, 97–118 (1991).
Budd, A. F., Adrain, T. S., Park, J. W., Klaus, J. S. & Johnson, K. G. The Neogene Marine Biota of Tropical America (“NMITA”) Database: Integrating Data from the Dominican Republic Project. in Evolutionary Stasis and Change in the Dominican Republic Neogene (eds. Nehm, R. H. & Budd, A. F.) 301–310, https://doi.org/10.1007/978-1-4020-8215-3_13 (Springer Netherlands, 2008).
Budd, A. F., Foster, C. T., Dawson, J. P. & Johnson, K. G. The Neogene Marine Biota of Tropical America (“NMITA”) database: Accounting for biodiversity in paleontology. J. Paleontol. 75, 743–751 (2001).
Scotese, C. R. PALEOMAP PaleoAtlas for GPlates and the PaleoData Plotter Program. https://www.earthbyte.org/paleomap-paleoatlas-for-gplates/ (2016).
Johnson, K. G., Budd, A. F. & Stemann, T. A. Extinction selectivity and ecology of Neogene Caribbean reef corals. Paleobiology 21, 52–73 (1995).
Pinzón, J. H. et al. Blind to morphology: genetics identifies several widespread ecologically common species and few endemics among Indo-Pacific cauliflower corals (Pocillopora, Scleractinia). J. Biogeogr. 40, 1595–1608 (2013).
Lathuilière, B. Coraux constructeurs du Bajocien inférieur de France: 2ème partie. Geobios 33, 153–181 (2000).
Kiessling, W. & Kocsis, Á. T. Biodiversity dynamics and environmental occupancy of fossil azooxanthellate and zooxanthellate scleractinian corals. Paleobiology 41, 402–414 (2015).
Raja, N. B., Dimitrijević, D., Krause, M. C. & Kiessling, W. Ancient Reef Traits Database. Zenodo https://doi.org/10.5281/zenodo.5717611 (2022).
Mannani, M. Late Triassic scleractinian corals from Nayband Formation, southwest Ardestan, Central Iran. Bol. Soc. Geológica Mex. 72, A090619 (2020).
Löser, H., Stemann, T. A. & Mitchell, S. Oldest scleractinian fauna from Jamaica (Hauterivian, Benbow Inlier). J. Paleontol. 83, 333–349 (2009).
Löser, H. Morphology, Taxonomy and Distribution of the Cretaceous coral genus Aulastraeopora (Late Barremian-Early Cenomanian; Scleractinia). Riv. Ital. Paleontol. E Stratigr. 114, (2008).
Löser, H. Revision of Actinastrea, the most common Cretaceous coral genus. Paläontol. Z. 86, 15–22 (2012).
Löser, H., Werner, W. & Darga, R. A Middle Cenomanian coral fauna from the Northern Calcareous Alps (Bavaria, Southern Germany) – new insights into the evolution of Mid-Cretaceous corals. Zitteliana 53, 37–76 (2013).
Löser, H. & Bilotte, M. Taxonomy of a platy coral association from the Late Cenomanian of the southern Corbières (Aude, France). Ann. Paléontol. 103, 3–17 (2017).
Löser, H., Steuber, T. & Löser, C. Early Cenomanian coral faunas from Nea Nikopoli (Kozani, Greece; Cretaceous). Carnets Géologie Noteb. Geol. 18, 23–121 (2018).
Löser, H. Early evolution of the family Siderastraeidae (Scleractinia; Cretaceous-extant). Paläontol. Z. 90, 1–17 (2016).
Kiessling, W. et al. Massive corals in Paleocene siliciclastic sediments of Chubut (Argentina). Facies 51, 233–241 (2005).
Stolarski, J. & Vertino, A. First Mesozoic record of the scleractinian Madrepora from the Maastrichtian siliceous limestones of Poland. Facies 53, 67–78 (2007).
Yabe, H. & Sugiyama, T. 5. Younger Cenozoic Reef-corals from the Nabire Beds of Nabire, Dutch New Guinea. Proc. Imp. Acad. 18, 16–23 (1942).
Wilson, M. A., Vinn, O. & Palmer, T. J. Bivalve borings, bioclaustrations and symbiosis in corals from the Upper Cretaceous (Cenomanian) of southern Israel. Palaeogeogr. Palaeoclimatol. Palaeoecol. 414, 243–245 (2014).
Tomás, S., Löser, H. & Salas, R. Low-light and nutrient-rich coral assemblages in an Upper Aptian carbonate platform of the southern Maestrat Basin (Iberian Chain, eastern Spain). Cretac. Res. 29, 509–534 (2008).
Baron-Szabo, R. C. Scleractinian corals from the upper Berriasian of central Europe and comparison with contemporaneous coral assemblages. Zootaxa 4383, 1 (2018).
Kiessling, W., Roniewicz, E., Villier, L., Leonide, P. & Struck, U. An early Hettangian coral reef in southern France: Implications for the end-Triassic reef crisis. PALAIOS 24, 657–671 (2009).
Stanley, G. D. & Beauvais, L. Middle Jurassic corals from the Wallowa terrane, west-central Idaho. J. Paleontol. 64, 352–362 (1990).
Gretz, M., Lathuilière, B., Martini, R. & Bartolini, A. The Hettangian corals of the Isle of Skye (Scotland): An opportunity to better understand the palaeoenvironmental conditions during the aftermath of the Triassic–Jurassic boundary crisis. Palaeogeogr. Palaeoclimatol. Palaeoecol. 376, 132–148 (2013).
Reolid, M., Molina, J. M., Löser, H., Navarro, V. & Ruiz-Ortiz, P. A. Coral biostromes of the Middle Jurassic from the Subbetic (Betic Cordillera, southern Spain): facies, coral taxonomy, taphonomy, and palaeoecology. Facies 55, 575–593 (2009).
Pandey, D. K., Lathuilière, B., Fürsich, F. T. & Kuldeep, S. The oldest Jurassic cyathophorid coral (Scleractinia) from siliciclastic environments of the Kachchh Basin, western India. Paläontol. Z. 76, 347–356 (2002).
Löser, H. & Heinrich, M. New coral genera and species from the Rußbach and Gosau area (Upper Cretaceous; Austria). Palaeodiversity 11, 127–149 (2018).
Stanley, G. D. & Whalen, M. T. Triassic corals and spongiomorphs from Hells Canyon, Wallowa terrane, Oregon. J. Paleontol. 63, 800–819 (1989).
Gill, G. A., Santantonio, M. & Lathuilière, B. The depth of pelagic deposits in the Tethyan Jurassic and the use of corals: an example from the Apennines. Sediment. Geol. 166, 311–334 (2004).
Baron-Szabo, R. C., Hamedani, A. & Senowbari-Daryan, B. Scleractinian corals from lower cretaceous deposits north of Esfahan (central Iran). Facies 48, 199–215 (2003).
Lathuilière, B., Baron-Szabo, R. C., Charbonnier, S. & Pacaud, J.-M. The Mesozoic scleractinian genus Adelocoenia (Stylinidae) and its Jurassic species. Carnets Géologie Noteb. Geol. 20, 367–406 (2020).
Roniewicz, E. & Stanley, G. D. Middle Triassic cnidarians from the New Pass Range, Central Nevada. J. Paleontol. 72, 246–256 (1998).
Shepherd, H. M. E., Stanley, G. D. & Amirhassankhani, F. Norian to Rhaetian scleractinian corals in the Ferdows Patch Reef (Nayband Formation, east central Iran). J. Paleontol. 86, 801–812 (2012).
Budd, A. F. & Wallace, C. C. First record of the Indo-Pacific reef coral genus Isopora in the Caribbean Region: two new species from the Neogene of Curaçao, Netherlands Antilles. Palaeontology 51, 1387–1401 (2008).
Pandolfi, J. M. A new, extinct pleistocene reef coral from the Montastraea “annularis” species complex. J. Paleontol. 81, 472–482 (2007).
El-Asa’ad, G. M. A. Oxfordian hermatypic corals from Central Saudi Arabia. Geobios 24, 267–287 (1991).
Masse, J.-P., Morycowa, E. & Fenerci-Masse, M. Valanginian-Hauterivian scleractinian coral communities from the Marseille region (SE France). Cretac. Res. 30, 178–192 (2009).
El-Sorogy, A. S. & Al-Kahtany, K. M. Contribution to the scleractinian corals of Hanifa Formation, Upper Jurassic, Jabal Al-Abakkayn, central Saudi Arabia. Hist. Biol. 27, 90–102 (2015).
Beauvais, L. & Stump, T. E. Corals, molluscs, and paleogeography of late Jurassic strata of the Cerro Pozo Serna, Sonora, Mexico. Palaeogeogr. Palaeoclimatol. Palaeoecol. 19, 275–301 (1976).
Roniewicz, E., Stanley, G. D., da Costa Monteiro, F. & Grant-Mackie, J. A. Late Triassic (Carnian) corals from Timor-Leste (East Timor): their identity, setting, and biogeography. Alcheringa Australas. J. Palaeontol. 29, 287–303 (2005).
Stanley, G. D. & Onoue, T. Upper Triassic reef corals from the Sambosan Accretionary Complex, Kyushu, Japan. Facies 61, 1 (2015).
Melnikova, G. K. & Roniewicz, E. Early Jurassic corals with dominating solitary growth forms from the Kasamurg Mountains, Central Asia. Palaeoworld 26, 124–148 (2017).
Stanley, G. D. & Beauvais, L. Corals from an Early Jurassic coral reef in British Columbia: refuge on an oceanic island reef. Lethaia 27, 35–47 (1994).
Caruthers, A. H. & Stanley, G. D. Systematic analysis of Upper Triassic silicified scleractinian corals from Wrangellia and the Alexander Terrane, Alaska and British Columbia. J. Paleontol. 82, 470–491 (2008).
Roniewicz, E. & Stanley, G. D. Upper Triassic corals from Nevada, western North America, and the implications for paleoecology and paleogeography. J. Paleontol. 87, 934–964 (2013).
Lathuilière, B. Coraux constructeurs du Bajocien inférieur de France. 1ere partie. Geobios 33, 51–72 (2000).
Morycowa, E. Supplemental data on Triassic (Anisian) corals from Upper Silesia (Poland). Ann. Soc. Geol. Pol. https://doi.org/10.14241/asgp.2018.001 (2018).
Budd, A. F. & Bosellini, F. R. Revision of Oligocene Mediterranean meandroid corals in the scleractinian families Mussidae, Merulinidae and Lobophylliidae. J. Syst. Palaeontol. 14, 771–798 (2016).
Roniewicz, E. Early Norian (Triassic) Corals from the Northern Calcareous Alps, Austria, and the Intra-Norian Faunal Turnover. Acta Palaeontol. Pol. 56, 401–428 (2011).
Budd, A. F., Adrain, T. S., Park, J. W., Klaus, J. S. & Johnson, K. G. The Neogene Marine Biota of Tropical America (“NMITA”) Database: Integrating Data from the Dominican Republic Project. in Evolutionary Stasis and Change in the Dominican Republic Neogene (eds. Nehm, R. H. & Budd, A. F.) vol. 30 301–310 (Springer Netherlands, 2008).
Mielnikova, G. Monstroseris, a new Upper Triassic scleractinian coral from Iran. Acta Palaeontol. Pol. 34, 71–74 (1989).
Löser, H. Taxonomy, stratigraphic distribution and palaeobiogeography of the Early Cretaceous coral genus Holocystis. Rev. Mex. Cienc. Geológicas 23, 288–301 (2006).
Löser, H. Corals from the Maastrichtian Ocozocoautla Formation (Chiapas, Mexico)-a closer look. Rev. Mex. Cienc. Geológicas 29, 534–550 (2012).
Löser, H. The Barremian coral fauna of the Serre de Bleyton mountain range (Drôme, SE France). Ann. Naturhistorischen Mus. Wien Ser. Für Mineral. Petrogr. Geol. Paläontol. Anthropol. Prähistorie 112, 575–612 (2010).
Löser, H., García-Barrera, P., Mendoza-Rosales, C. C. & Ortega-Hernández, J. Corals from the Early Cretaceous (Barremian - Early Albian) of Puebla (Mexico) - Introduction and Family Stylinidae. Rev. Mex. Cienc. Geológicas 30, 385–403 (2013).
Morycowa, E., Masse, J.-P., Arias, C. & Minondo, L. V. Montlivaltia multiformis Toula (Scleractinia) from the Aptian of the Prebetic domain (SE Spain). Span. J. Palaeontol. 16, 131–144 (2001).
Morycowa, E. & Masse, J.-P. Actinaraeopsis ventosiana, a new scleractinian species from the Lower Cretaceous of Provence (SE France). Ann. Soc. Geol. Pol. 77, 141–145 (2007).
Stolarski, J. & Taviani, M. Oligocene scleractinian corals from CRP- 3 drillhole, McMurdo Sound (Victoria Land Basin, Antarctica). Terra Antarct. 8, 1–4 (2001).
Morycowa, E. & Marcopoulou-Diacantoni, A. Albian corals from the Subpelagonian zone of Central Greece (Agrostylia, Parnassos region). Ann. Soc. Geol. Pol. 72, 1–65 (2002).
Morycowa, E. & Roniewicz, E. Revision of the genus Cladophyllia and description of Apocladophyllia gen. n.(Cladophylliidae fam. n., Scleractinia). Acta Palaeontol. Pol. 35, 165–190 (1990).
Morycowa, E. & Masse, J.-P. Lower Cretaceous Microsolenina (Scleractinia) from Provence (southern France). Ann. Soc. Geol. Pol. 79, 97–140 (2009).
Squires, R. L. & Demetrion, R. A. Paleontology of the Eocene Bateque Formation, Baja California Sur, Mexico. Contrib. Sci. 434, 1–55 (1992).
Wells, J. W. Cretaceous, Tertiary, and Recent Corals, a Sponge, and an Alga from Venezuela. J. Paleontol. 18, 429–447 (1944).
Morycowa, E. & Decrouez, D. Early Aptian scleractinian corals from the Upper Schrattenkalk of Hergiswil (Lucerne region, Helvetic Zone of central Switzerland). Rev. Paléobiol. 25, 791 (2006).
Stolarski, J. Paleogene corals from Seymour Island, Antarctic Peninsula. Palaeontol. Pol. 55, 1–63 (1996).
Vaughan, T. W. New Corals: One Recent, Alaska; Three Eocene, Alabama and Louisiana. J. Paleontol. 15, 280–284 (1941).
Stolarski, J. & Russo, A. Microstructural diversity of the stylophyllid [Scleractinia] skeleton. Acta Palaeontol. Pol. 47, (2002).
Roniewicz, E. Jurassic scleractinian coral Thamnoseris Etallon, 1864 (Scleractinia), and its homeomorphs. Acta Palaeontol. Pol. 24, 51–70 (1979).
Lathuilière, B., Charbonnier, S. & Pacaud, J.-M. Nomenclatural and taxonomic acts and remarks for the revision of Jurassic corals. Zitteliana 89, 133–150 (2017).
Roniewicz, E. Upper Kimmeridgian Scleractinia of Pomerania (Poland). Ann. Soc. Geol. Pol. 47, 613–622 (1977).
Roniewicz, E. Scleractinia from the Upper Portlandian of Tisbury, Wiltshire, England. Acta Palaeontol. Pol. 15, 519–541 (1970).
Roniewicz, E. Kimmeridgian-Valanginian reef corals from the Moesian platform from Bulgaria. Ann. Soc. Geol. Pol. 78, 91–134 (2008).
Ricci, C., Lathuiliere, B. & Rusciadelli, G. Coral communities, zonation and paleoecology of an Upper Jurassic reef complex (Ellipsactinia Limestones, Central Apennines, Italy). Riv. Ital. Paleontol. E Stratigr. 124, 433–508 (2018).
Pandey, D. K. et al. Jurassic corals from southern Tunisia. Zitteliana A45, 3–34 (2005).
Pandey, D. K. et al. Jurassic corals from the Shemshak Formation of the Alborz Mountains, Iran. Zitteliana A46, 41–74 (2006).
Pandey, D. K. & Fürsich, F. T. Contributions to the Jurassic of Kachchh, Western India I. The coral fauna. Beringeria 8, 3–69.
Morycowa, E. & Mišík, M. Upper Jurassic shallow-water scleractinian corals from the Pieniny Klippen Belt (Western Carpathians, Slovakia). Geol. Carpathica 56, (2005).
Pandey, D. K. et al. Lower Cretaceous corals from the Koppeh Dagh, NE-Iran. Zitteliana A47, 3–52 (2007).
Morycowa, E. Corals from the Tithonian carbonate complex in the Dąbrowa Tarnowska–Szczucin area (Polish Carpathian Foreland). Ann. Soc. Geol. Pol. 82, 1–38 (2012).
Baron-Szabo, R. Corals of the Theresienstein reef (Upper Turonian-Coniacian, Salzburg, Austria). Proc. Biol. Soc. Wash. 10, 257–268 (2001).
Morycova, E. Middle Triassic Scleractinia from the Cracow-Silesia region, Poland. Acta Palaeontol. Pol. 33, 91–121 (1988).
El-Asa’ad, G. M. A. Callovian colonial corals from the Tuwaiq Mountain Limestone of Saudi Arabia. Paleontology 32, 675–684 (1989).
Roniewicz, E. & Michalik, J. Rhaetian scleractinian corals in the Western Carpathians. Geol. Carpathica 49, 391–399 (1998).
Roniewicz, E. & Michalik, J. Carnian corals from the Male Karpaty Mountains, Western Carpathians, Slovakia. Geol. Carpathica 53, 149–157 (2002).
Roniewicz, E. Rhaetian corals of the Tatra Mts. Acta Geol. Pol. 24, 97–116 (1974).
Turnšek, D. et al. Contributions to the fauna (corals, brachiopods) and stable isotopes of the Late Triassic Steinplatte reef/basin-complex, Northern Calcareous Alps, Austria. Abh. Geol. Bundensanstalt 56, 121–142 (1999).
Roniewicz, E. Upper Triassic Solitary Corals from the Gosaukamm and other North Alpine Regions. Sitzungsberichte Biol. Wiss. Erdwissenschaften 3–41 (1995).
Wells, J. W. & Jenks, W. F. Mesozoic invertebrate faunas of Peru. Part 3, Lower Jurassic corals from the Arequipa region. Am. Mus. Novit. 1631 (1953).
Turnšek, D. & Senowbari-Daryan, B. Upper Triassic (Carnian-Lowermost Norian) Corals from the Pantokrator Limestone of Hydra (Greece). AbhGeolB-A 50, (1994).
Wells, J. W. Jurassic Corals from the Smackover Limestone, Arkansas. J. Paleontol. 16, 126–129 (1942).
Turnšek, D., Buser, S. & Debeljak, I. Liassic coral patch reef above the” Lithiotid limestone” on Trnovski gozd plateau, west Slovenia: Liasni koralni kopasti greben na” litiotidnem apnencu” v Trnovskem gozdu, zahodna Slovenija. Razpr. IV Razreda SAZU XLIV–1, 285–331 (2003).
Turnšek, D. & Košir, A. Early Jurassic corals from Krim Mountain, Slovenia. Razpr. IV Razreda SAZU XLI–1, 81–113 (2000).
Roniewicz, E. Triassic scleractinian corals of the Zlambach Beds, Northern Calcareous Alps, Austria. Denkschr Osterr Akad Wiss Math Nat K1 126, 1–152 (1989).
Roniewicz, E. Les scléractiniaires du Jurassique supérieur de la Dobrogea centrale, Roumanie. Palaeontol. Pol. 34, 17–121 (1976).
Kiessling, W., Kumar Pandey, D., Schemm-Gregory, M., Mewis, H. & Aberhan, M. Marine benthic invertebrates from the Upper Jurassic of northern Ethiopia and their biogeographic affinities. J. Afr. Earth Sci. 59, 195–214 (2011).
Lathuilière, B. Coraux constructeurs du Bajocien inférieur de France: 2ème partie. Geobios 33, 153–181 (2000).
Baron‐Szabo, R. C. Corals of the K/T‐boundary: Scleractinian corals of the suborders Astrocoeniina, Faviina, Rhipidogyrina and Amphiastraeina. J. Syst. Palaeontol. 4, 1–108 (2006).
Filkorn, H. F. & Pantoja-Alor, J. NOMENCLATURAL NOTES Mexican Cretaceous coral species (Cnidaria, Anthozoa, Scleractinia) described as new by Filkorn & Pantoja-Alor (2009), but deemed ‘unpublished’ under the International Code of Zoological Nomenclature: republication of data necessary for nomenclatural availability. Bull. Zool. Nomencl. 72, 93–101 (2015).
Olden, J. D., Poff, N. L. & Bestgen, K. R. Trait Synergisms and the Rarity, Extirpation, and Extinction Risk of Desert Fishes. Ecology 89, 847–856 (2008).
Schleuning, M. et al. Trait-Based Assessments of Climate-Change Impacts on Interacting Species. Trends Ecol. Evol. 35, 319–328 (2020).
Solan, M., Aspden, R. J. & Paterson, D. M. Marine Biodiversity and Ecosystem Functioning: Frameworks, Methodologies, and Integration. (OUP Oxford, 2012).
Suding, K. N. et al. Scaling environmental change through the community-level: a trait-based response-and-effect framework for plants. Glob. Change Biol. 14, 1125–1140 (2008).
Finnegan, S. et al. Paleontological baselines for evaluating extinction risk in the modern oceans. Science https://doi.org/10.1126/science.aaa6635 (2015).
Yasuhara, M. & Deutsch, C. A. Paleobiology provides glimpses of future ocean. Science https://doi.org/10.1126/science.abn2384 (2022).
Cooley, S. et al. Ocean and coastal ecosystems and their services. in Climate change 2022: Impacts, adaptation and vulnerability. Contribution of Working Group II to the Sixth Assessment Report of the Intergovernmental Panel of Climate Change (IPCC) (eds. Pörtner, H.-O. et al.) (Cambridge University Press, 2022).
Acknowledgements
We would like to thank J. Madin for his suggestions and feedback in the initial stage of this project. Our thanks also go to the following people for their help in data entry: K. Frühbeißer, M. A. Gomez Correa, T. M. Khan, J. Machleidt, A. El Manharawy, I. Smith. This work was supported by the Deutsche Forschungsgemeinschaft (KI 806/17–1) and is embedded in the Research Unit TERSANE (FOR 2332: Temperature-related stressors as a unifying principle in ancient extinctions).
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
N.B.R. and W.K. designed the database. N.B.R. and M.C.K. programmed the database and user interface. N.B.R. and D.D. organised and carried out the data collection. N.B.R. drafted the manuscript with input from all the authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Raja, N.B., Dimitrijević, D., Krause, M.C. et al. Ancient Reef Traits, a database of trait information for reef-building organisms over the Phanerozoic. Sci Data 9, 425 (2022). https://doi.org/10.1038/s41597-022-01486-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41597-022-01486-0
This article is cited by
-
A dynamic and collaborative database for morphogeometric information of trilobites
Scientific Data (2023)