Abstract
Background/objectives
To assess the methodological quality of Clinical Practice Guidelines (CPG) for the diagnosis and management of Retinal Vein Occlusion (RVO).
Methods
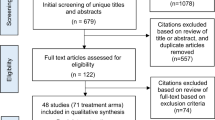
A systematic review of CPGs for the diagnosis and management of RVO was carried out with a search in databases, metasearch engines, CPG development institutions, ophthalmology associations and CPG repositories until April 2022. Search update was performed on April 2023, with no new record available. Five CPGs published in the last 10 years in English/Spanish were selected, and 5 authors evaluated them independently, using the Appraisal of Guidelines for Research and Evaluation (AGREE-II) instrument. An individual assessment of each CPG by domain (AGREE-II), an overall assessment of the guide, and its use with or without modifications were performed. Additionally, a meta-synthesis of the recommendations for the most relevant outcomes was carried out.
Results
The lowest score (mean 18.8%) was for domain 5 ‘applicability’, and the highest score (mean 62%) was for domain 4 ‘clarity of presentation’. The 2019 American guideline (PPP) presented the best score (40.4%) in domain 3 ‘rigour of development’. When evaluating the overall quality of the CPGs analysed, all CPGs could be recommended with modifications. In the meta-synthesis, anti-VEGF therapy is the first-choice therapy for macular oedema associated with RVO, but there is no clear recommendation about the type of anti-VEGF therapy to choose. Recommendations for diagnosis and follow-up are similar among the CPGs appraised.
Conclusion
Most CPGs for the diagnosis and management of RVO have a low methodological quality assessed according to the AGREE-II. PPP has the higher score in the domain ‘rigour of development’. Among the CPGs appraised, there is no clear recommendation on the type of anti-VEGF therapy to choose.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 18 print issues and online access
$259.00 per year
only $14.39 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Song P, Xu Y, Zha M, Zhang Y, Rudan I. Global epidemiology of retinal vein occlusion: a systematic review and meta-analysis of prevalence, incidence, and risk factors. J Glob Health. 2019;9:010427.
Ørskov M, Vorum H, Larsen TB, Lip GYH, Bek T, Skjøth F. Similarities and differences in systemic risk factors for retinal artery occlusion and retinal vein occlusion: a nationwide case-control study. Int Ophthalmol. 2023;43:817–24.
Institute of Medicine. Clinical practice guidelines we can trust. Washington DC: The National Academies Press; 2011.
Yao L, Brignardello-Petersen R, Guyatt GH. Developing trustworthy guidelines using GRADE. Can J Ophthalmol. 2020;55:349–51.
AGREE Next Steps Consortium. The AGREE II Instrument [Internet]. 2017. http://www.agreetrust.org.
Brouwers MC, Kerkvliet K, Spithoff K, AGREE Next Steps Consortium. The AGREE Reporting Checklist: a tool to improve reporting of clinical practice guidelines. BMJ. 2016;352:i1152.
Ou Y, Goldberg I, Migdal C, Lee PP. A critical appraisal and comparison of the quality and recommendations of glaucoma clinical practice guidelines. Ophthalmology. 2011;118:1017–23.
Wu AM, Wu CM, Young BK, Wu DJ, Chen A, Margo CE, et al. Evaluation of primary open-angle glaucoma clinical practice guidelines. Can J Ophthalmol. 2015;50:192–6.
Michaelov E, Armstrong JJ, Nguyen M, Instrum B, Lam T, Denstedt J, et al. Assessing the methodological quality of glaucoma clinical practice guidelines and their recommendations on microinvasive glaucoma surgery: a systematic review. J Glaucoma. 2018;27:6.
Lingham G, Thakur S, Safi S, Gordon I, Evans JR, Keel S. A systematic review of clinical practice guidelines for childhood glaucoma. BMJ Open Ophthalmol. 2022;7:e000933.
Chan VF, Yong AC, Azuara-Blanco A, Gordon I, Safi S, Lingham G, et al. A systematic review of clinical practice guidelines for infectious and non-infectious conjunctivitis. Ophthalmic Epidemiol. 2022;29:473–82.
Zhang JH, Ramke J, Lee CN, Gordon I, Safi S, Lingham G, et al. A systematic review of clinical practice guidelines for cataract: evidence to support the development of the WHO package of eye care interventions. Vision. 2022;6:36.
Evans JR, Lawrenson JG, Ramke J, Virgili G, Gordon I, Lingham G, et al. Identification and critical appraisal of evidence for interventions for refractive error to support the development of the WHO package of eye care interventions: a systematic review of clinical practice guidelines. Ophthalmic Physiol Opt J Br Coll Ophthalmic Opt Optom. 2022;42:526–33.
Lingham G, McGuinness MB, Safi S, Gordon I, Evans JR, Keel S. Clinical practice guidelines for the detection and treatment of amblyopia: a systematic literature review. J Binocul Vis Ocul Motil. 2022;72:77–85.
Wu AM, Wu CM, Young BK, Wu DJ, Margo CE, Greenberg PB. Critical Appraisal Of Clinical Practice Guidelines For Age-related Macular Degeneration. J Ophthalmol. 2015;2015:710324.
Han X, Chen Y, Gordon I, Safi S, Lingham G, Evans J, et al. A Systematic Review Of Clinical Practice Guidelines For Age-related Macular Degeneration. Ophthalmic Epidemiol. 2022;1-8.
Vargas-Peirano M, Verdejo C, Vergara-Merino L, Loézar C, Hoehmann M, Pérez-Bracchiglione J. Intravitreal antivascular endothelial growth factor in diabetic macular oedema: scoping review of clinical practice guidelines recommendations. Br J Ophthalmol. 2021; bjophthalmol-2021-319504.
Chen Y, Han X, Gordon I, Safi S, Lingham G, Evans J, et al. A systematic review of clinical practice guidelines for myopic macular degeneration. J Glob Health. 2022;12:04026.
Johnston A, Kelly SE, Hsieh SC, Skidmore B, Wells GA. Systematic reviews of clinical practice guidelines: a methodological guide. J Clin Epidemiol. 2019;108:64–76.
Keel S, Evans JR, Block S, Bourne R, Calonge M, Cheng CY, et al. Strengthening the integration of eye care into the health system: methodology for the development of the WHO package of eye care interventions. BMJ Open Ophthalmol. 2020;5:e000533.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;29:n71.
Royal College of Ophthalmologists. Retinal Vein Occlusion (RVO) Guidelines. Royal College of Ophthalmologists; 2022. https://www.rcophth.ac.uk/resources-listing/retinal-vein-occlusion-rvo-guidelines/. Accessed 30 April 2022.
Schmidt-Erfurth U, Garcia-Arumi J, Gerendas BS, Midena E, Sivaprasad S, Tadayoni R, et al. Guidelines foR The Management Of Retinal Vein Occlusion by the European Society of Retina Specialists (EURETINA). Ophthalmologica. 2019;242:123–62.
Flaxel CJ, Adelman RA, Bailey ST, Fawzi A, Lim JI, Vemulakonda GA, et al. Retinal vein occlusions preferred practice Pattern®. Ophthalmology. 2020;127:P288–320.
Gómez-Ulla F, Abraldes MJ 2015 guía 5. Manejo de las oclusiones venosas de la retina. Guías de Práctica Clínica de la SERV. Sociedad Española de Retina y Vítreo; 2021. https://serv.es/publicaciones/guias-practica-clinica/. Accessed 30 April 2022.
Berger AR, Cruess AF, Altomare F, Chaudhary V, Colleaux K, Greve M, et al. Optimal treatment of retinal vein occlusion: Canadian Expert Consensus. Ophthalmologica. 2015;234:6–25.
Baker A, Young K, Potter J, Madan I. A review of grading systems for evidence-based guidelines produced by medical specialties. Clin Med. 2010;10:358–63.
Scottish Intercollegiate Guidelines Network (SIGN). New developments. 2021. https://www.sign.ac.uk/what-we-do/methodology/new-developments/.
Granholm A, Alhazzani W, Møller MH. Use of the GRADE approach in systematic reviews and guidelines. Br J Anaesth. 2019;123:554–9.
Grading of Recommendations Assessment, Development and Evaluation (short GRADE) Working Group. 2024. https://www.gradeworkinggroup.org/.
Thornton J, Alderson P, Tan T, Turner C, Latchem S, Shaw E, et al. Introducing GRADE across the NICE clinical guideline program. J Clin Epidemiol. 2013;66:124–31.
Young BK, Wu CM, Wu AM, Margo CE, Greenberg PB. Are clinical practice guidelines for cataract and glaucoma trustworthy? Am J Med Qual. 2015;30:188–90.
Salviat F, Guedj M, Bodaghi B, Sahel JA, Tubach F, Dechartres A. Quality of evidence in ophthalmology: an overview of cochrane reviews. Ophthalmology. 2021;128:330–2.
Golozar A, Chen Y, Lindsley K, Rouse B, Musch DC, Lum F, et al. Identification and description of reliable evidence for 2016 American Academy of Ophthalmology preferred practice pattern guidelines for cataract in the adult eye. JAMA Ophthalmol. 2018;136:514–23.
Lindsley K, Li T, Ssemanda E, Virgili G, Dickersin K. Interventions for age-related macular degeneration. Ophthalmology. 2016;123:884–97.
Venus C, Jamrozik E. Transparency in clinical practice guidelines: the problem of consensus‐based recommendations and practice points. Intern Med J. 2021;51:291–4.
Ford N, Thomas R, Grove J. Transparency: a central principle underpinning trustworthy guidelines. J Clin Epidemiol. 2022;142:246–8.
García LM, Sanabria AJ, Álvarez EG, Trujillo-Martín MM, Etxeandia-Ikobaltzeta I, Kotzeva A, et al. The validity of recommendations from clinical guidelines: a survival analysis. CMAJ. 2014;186:1211–9.
Becker M, Neugebauer EAM, Eikermann M. Partial updating of clinical practice guidelines often makes more sense than full updating: a systematic review on methods and the development of an updating procedure. J Clin Epidemiol. 2014;67:33–45.
Álvarez-Vargas ML, Galvez-Olortegui JK, Galvez-Olortegui TV, Sosa-Rosado JM, Camacho-Saavedra LA. Clinical practice guidelines in hypertension: a review. Medwave. 2015;15:e6290.
Alshowaeir D, Almasoud N, Aldossari S, Alsirhy EY, Osman E, Turjoman A, et al. Primary open angle glaucoma management in a tertiary eye care center in Saudi Arabia: a best practice implementation pilot project. JBI Evid Implement. 2021;19:208–16.
El-Assal K, Foulds J, Dobson S, Sanders R. A comparative study of glaucoma referrals in Southeast Scotland: effect of the new general ophthalmic service contract, eyecare integration pilot programme and NICE guidelines. BMC Ophthalmol. 2015;15:172.
Cabrera-Aguas M, Kerdraon Y, Symes RJ, McCluskey P, Samarawickrama C, Rawlinson W, et al. Development, implementation, and evaluation of treatment guidelines for herpes simplex keratitis in Sydney, Australia. Cornea. 2020;39:834–40.
Kewcharoen J, Tom ES, Wiboonchutikula C, Trongtorsak A, Wittayalikit C, Vutthikraivit W, et al. Prevalence of atrial fibrillation in patients with retinal vessel occlusion and its association: a systematic review and meta-analysis. Curr Eye Res. 2019;44:1337–44.
Posch-Pertl L, List W, Michelitsch M, Pinter-Hausberger S, Posch F, Innauer F, et al. Role of the ABO blood groups as a risk factor for retinal vein occlusion. Ophthalmic Res. 2023;66:164–9.
Ørskov M, Vorum H, Bjerregaard Larsen T, Vestergaard N, Lip GYH, Bek T, et al. A review of risk factors for retinal vein occlusions. Expert Rev Cardiovasc Ther. 202;20:761–72.
Schreiberova Z, Rehak J, Babkova B, Sin M, Rybarikova M, Paskova B, et al. Hypertension, hyperlipidaemia and thrombophilia as the most common risk factors for retinal vein occlusion in patients under 50 years. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2022. https://doi.org/10.5507/bp.2022.036. Online ahead of print.
Wang Y, Wu S, Wen F, Cao Q. Diabetes mellitus as a risk factor for retinal vein occlusion: a meta-analysis. Medicine. 2020;99:e19319.
Valeriani E, Paciullo F, Porfidia A, Pignatelli P, Candeloro M, Di Nisio M, et al. Antithrombotic treatment for retinal vein occlusion: a systematic review and meta-analysis. J Thromb Haemost. 2023;21:284–93.
Guirado-Torrecillas L, Salazar-Rosa V. Retinal vein occlusion, a great unknown and a challenge in venous thromboembolic disease. Rev Clínica Esp Engl Ed. 2023;223:96–7.
Galvez-Olortegui J, Burgueño-Montañes C, Zavaleta-Mercado M, Galvez-Olortegui T. Recommendations for systemic diagnosis and management in clinical practice guidelines for retinal vein occlusion (RVO). Rev Clínica Esp Engl Ed. 2023;223:520–21.
Chatziralli I, Kazantzis D, Kroupis C, Machairoudia G, Dimitriou E, Theodossiadis G, et al. The impact of laboratory findings and optical coherence tomography biomarkers on response to intravitreal anti-VEGF treatment in patients with retinal vein occlusion. Int Ophthalmol. 2022;42:3449–57.
Hunter A, Williams M. Long-term outcomes for patients treated for macular oedema secondary to retinal vein occlusion: a systematic review. BMJ Open Ophthalmol. 2022;7:e001010.
Nanji K, Khan M, Khalid MF, Xie JS, Sarohia GS, Phillips M, et al. Treat‐and‐extend regimens of anti‐vascular endothelial growth factor therapy for retinal vein occlusions: a systematic review and meta‐analysis. Acta Ophthalmol. 2022;100:e1199–e1208.
Patil NS, Dhoot AS, Nichani PAH, Popovic MM, Muni RH, Kertes PJ. Safety and efficacy of a treat-and-extend regimen of anti–vascular endothelial growth factor agents for diabetic macular edema or macular edema secondary to retinal vein occlusion: a systematic review and meta-analysis. Ophthalmic Surg Lasers Imaging Retina. 2023;54:131–8.
Wang N, Hunt A, Nguyen V, Shah J, Fraser‐Bell S, McAllister I, et al. One‐year real‐world outcomes of bevacizumab for the treatment of macular oedema secondary to retinal vein occlusion. Clin Exp Ophthalmol. 2022;50:1038–46.
Adedokun L, Burke C. Cost-effectiveness of ranibizumab versus aflibercept for macular edema secondary to branch retinal vein occlusion: a UK healthcare perspective. Adv Ther. 2016;33:116–28.
Qian T, Zhao M, Wan Y, Li M, Xu X. Comparison of the efficacy and safety of drug therapies for macular edema secondary to central retinal vein occlusion. BMJ Open. 2018;8:e022700.
Gao S, Zhang Y, Li X, Ge G, Duan J, Lei C, et al. Comparative efficacy of pharmacotherapy for macular edema secondary to retinal vein occlusion: a network meta-analysis. Front Pharmacol. 2021;12:752048.
Sangroongruangsri S, Ratanapakorn T, Wu O, Anothaisintawee T, Chaikledkaew U. Comparative efficacy of bevacizumab, ranibizumab, and aflibercept for treatment of macular edema secondary to retinal vein occlusion: a systematic review and network meta-analysis. Expert Rev Clin Pharmacol. 2018;11:903–16.
Regnier SA, Larsen M, Bezlyak V, Allen F. Comparative efficacy and safety of approved treatments for macular oedema secondary to branch retinal vein occlusion: a network meta-analysis. BMJ Open. 2015;5:e007527.
Nanji K, Sarohia GS, Kennedy K, Ceyhan T, McKechnie T, Phillips M, et al. The 12- and 24-month effects of intravitreal ranibizumab, aflibercept, and bevacizumab on intraocular pressure. Ophthalmology. 2022;129:498–508.
Hattenbach LO, Abreu F, Arrisi P, Basu K, Danzig CJ, Guymer R, et al. BALATON and COMINO: phase 3 randomized clinical trials of faricimab for retinal vein occlusion: study design and rationale. Ophthalmol Sci. 2023;3:100302.
Chakraborty D, Mondal S, Boral S, Das A. Intravitreal injection of brolucizumab for recalcitrant macular edema due to central retinal vein occlusion: a small case series. Case Rep Ophthalmol. 2022;13:912–9.
Garretón R, Gonzalez R. Aflibercept versus dexamethasone for macular edema secondary to central retinal vein occlusion. Medwave. 2019;19:e7738.
Zhang W, Liu Y, Sang A. Efficacy and effectiveness of anti-VEGF or steroids monotherapy versus combination treatment for macular edema secondary to retinal vein occlusion: a systematic review and meta-analysis. BMC Ophthalmol. 2022;22:472.
Namvar E, Yasemi M, Nowroozzadeh MH, Ahmadieh H. Intravitreal injection of anti-vascular endothelial growth factors combined with corticosteroids for the treatment of macular edema secondary to retinal vein occlusion: a systematic review and meta-analysis. Semin Ophthalmol. 2024;39:109–19.
Ford JA, Shyangdan D, Uthman OA, Lois N, Waugh N. Drug treatment of macular oedema secondary to central retinal vein occlusion: a network meta-analysis. BMJ Open. 2014;4:e005292.
Pranata R, Vania A, Vania R, Victor AA. Intravitreal ranibizumab versus dexamethasone implant in macular edema due to branch retinal vein occlusion: Systematic review and meta-analysis. Eur J Ophthalmol. 2021;31:1907–14.
Cornish EE, Zagora SL, Spooner K, Fraser‐Bell S. Management of macular oedema due to retinal vein occlusion: an evidence‐based systematic review and meta‐analysis. Clin Experiment Ophthalmol. 2023;51:313–38.
Xiaodong L, Xuejun X. The efficacy and safety of dexamethasone intravitreal implant for diabetic macular edema and macular edema secondary to retinal vein occlusion: a meta-analysis of randomized controlled trials. J Ophthalmol. 2022;2022:4007002.
Hertzberg SN, Moe MC, Jørstad ØK, Petrovski BÉ, Burger E, Petrovski G. Healthcare expenditure of intravitreal anti‐vascular endothelial growth factor inhibitors compared with dexamethasone implant for diabetic macular oedema. Acta Ophthalmol. 2022;100:e1630–e1640.
Cho H, Choi KS, Lee JY, Lee D, Choi NK, Lee Y, et al. Healthcare resource use and costs of diabetic macular oedema for patients with antivascular endothelial growth factor versus a dexamethasone intravitreal implant in Korea: a population-based study. BMJ Open. 2019;9:e030930.
Ruiz-Moreno JM, Ruiz-Medrano J. Early-switch versus late-switch in patients with diabetic macular edema: a cost-effectiveness study. Graefes Arch Clin Exp Ophthalmol. 2023;261:941–9.
Zhong P, He M, Yu H, Wu Q, Peng Q, Huang M, et al. A meta-analysis of cardiovascular events associated with intravitreal anti-VEGF treatment in patients with retinal vein occlusion. Curr Eye Res. 2020;45:615–22.
Billioti de Gage S, Bertrand M, Grimaldi S, Zureik M. Risk of myocardial infarction, stroke, or death in new users of intravitreal aflibercept versus ranibizumab: a nationwide cohort study. Ophthalmol Ther. 2022;11:587–602.
Zou W, Du Y, Ji X, Zhang J, Ding H, Chen J, et al. Comparison of the efficiency of anti-VEGF drugs intravitreal injections treatment with or without retinal laser photocoagulation for macular edema secondary to retinal vein occlusion: a systematic review and meta-analysis. Front Pharmacol. 2022;13:948852.
Scott IU, VanVeldhuisen PC, Oden NL, Ip MS, Blodi BA. Month 60 outcomes after treatment initiation with anti-vascular endothelial growth factor therapy for macular edema due to central retinal or hemiretinal vein occlusion. Am J Ophthalmol. 2022;240:330–41.
Ang JL, Ah-Moye S, Kim LN, Nguyen V, Hunt A, Barthelmes D, et al. A systematic review of real-world evidence of the management of macular oedema secondary to branch retinal vein occlusion. Eye. 2020;34:1770–96.
Funding
Self-funded.
Author information
Authors and Affiliations
Contributions
JGO: Conceptualisation, Validation, Formal analysis, Investigation, Data Curation, Writing - Original Draft, Writing - Review & Editing. RBE: Validation, Formal analysis, Investigation, Data Curation, Writing - Original Draft, Writing - Review & Editing. ISO: Validation, Formal analysis, Investigation, Data Curation, Writing - Review & Editing. HPH: Validation, Formal analysis, Investigation, Data Curation, Writing - Review & Editing. MCM: Conceptualisation, Writing - Review & Editing. CBM: Validation, Investigation, Data Curation, Writing - Review & Editing. TGO: Conceptualisation, Investigation, Writing - Review & Editing.
Corresponding author
Ethics declarations
Competing interests
JGO was peer reviewer of CPG, CPG critical appraisal tools, and SR of CPG, for Annals of Internal Medicine, Journal of Evidence Based Medicine and Journal of Affective Disorders. JGO has received speaker fees from Novelty Technology Care and Angelini. JGO has received Travel and meeting support from Esteve, Bausch and Lomb, Thea, Equipsa, Alcon, Zeiss, Angelini. RBE has received Travel and meeting support from Novartis Spain, Angelini, Thea, Roche, Alcon, Esteve and AbbVie. CBM has received Travel and meeting support from Bausch. ISO, HPH, MCM, MZM and TGO have nothing to disclose.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Galvez-Olortegui, J., Bouchikh-El Jarroudi, R., Silva-Ocas, I. et al. Systematic review of clinical practice guidelines for the diagnosis and management of retinal vein occlusion. Eye 38, 1722–1733 (2024). https://doi.org/10.1038/s41433-024-03008-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-024-03008-1