Abstract
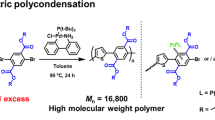
In this work, various types of chain-transfer agents (CTAs) were investigated in the reversible addition-fragmentation chain-transfer (RAFT) polymerization of isopropenyl boronate pinacol ester (IPBpin) toward not only precise synthesis of poly(IPBpin) but also elucidation of its feature as a monomer. CTAs suitable for conjugated monomers were found to be more compatible than those for nonconjugated monomers, likely due to the moderate stabilization of the growing radical by boron. This trend was supported by density functional theory-based calculations for energy balance between the growing IPBpin radical and the chain-transfer species to the CTA. The CTA-derived group at the ω-terminus of the obtained poly(IPBpin) was quantitatively converted into olefin under cobalt catalysis leading to improved stability, enabling evaluation of its well-defined structure by MALDI-TOF-MS and its thermal properties by DSC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Rudin A, Choi P. The elements of polymer science & engineering. 3rd ed. Boston: Elsevier, Academic Press; 2013.
Chiefari J, Chong YK, Ercole F, Krstina J, Jeffery J, Le TPT, et al. Living free-radical polymerization by reversible addition-fragmentation chain transfer: the RAFT process. Macromolecules. 1998;31:5559–62.
Moad G, Rizzardo E, Thang SH. Radical addition—fragmentation chemistry in polymer synthesis. Polymer. 2008;49:1079–131.
Keddie DJ, Moad G, Rizzardo E, Thang SH. RAFT agent design and synthesis. Macromolecules. 2012;45:5321–42.
Stenzel MH, Cummins L, Roberts GE, Davis TP, Vana P, Barner-Kowollik C. Xanthate mediated living polymerization of vinyl acetate: a systematic variation in MADIX/RAFT agent structure. Macromol Chem Phys. 2003;204:1160–8.
Destarac M, Franck X, Charmot D, Zard SZ. Dithiocarbamates as universal reversible addition-fragmentation chain transfer agents. Macromol Rapid Commun. 2000;21:1035–9.
Harrisson S, Liu X, Ollagnier J-N, Coutelier O, Marty JD, Destarac M. RAFT polymerization of vinyl esters: synthesis and applications. Polymers. 2014;6:1437–88.
Nakabayashi K, Mori H. Recent progress in controlled radical polymerization of N-vinyl monomers. Eur Polym J. 2013;49:2808–38.
Mori H, Ishikawa K, Abiko Y, Maki Y, Onuma A, Morishima M. Synthesis of triazole-based amphiphilic block copolymers containing carbazole moiety By RAFT polymerization. Macromol Chem Phys. 2012;213:1803–14.
Nakabayashi K, Abiko Y, Mori H. RAFT polymerization of S-vinyl sulfide derivatives and synthesis of block copolymers having two distinct optoelectronic functionalities. Macromolecules. 2013;46:5998–6012.
Mori H, Ookuma H, Nakano S, Endo T. Xanthate-mediated controlled radical polymerization of N-vinylcarbazole. Macromol Chem Phys. 2006;207:1005–17.
Wan WM, Baggett AW, Cheng F, Lin H, Liu SY, Jäkle F. Synthesis by free radical polymerization and properties of BN-polystyrene and BN-poly(vinylbiphenyl). Chem Commun. 2016;52:13616–9.
Thiedemann B, Gliese PJ, Hoffmann J, Lawrence PG, Sönnichsen FD, Staubitz A. High molecular weight poly(N-methyl-B-vinylazaborine)—a semi-inorganic B-N polystyrene analogue. Chem Commun. 2017;53:7258–61.
van de Wouw HL, Lee JY, Klausen RS. Gram-scale free radical polymerization of an azaborine vinyl monomer. Chem Commun. 2017;53:7262–5.
Nishikawa T, Ouchi M. An alkenyl boronate as a monomer for radical polymerizations: boron as a guide for chain growth and as a replaceable side chain for post-polymerization transformation. Angew Chem Int Ed. 2019;58:12435–9.
van de Wouw HL, Lee JY, Awuyah EC, Klausen RS. A BN aromatic ring strategy for tunable hydroxy content in polystyrene. Angew Chem Int Ed. 2018;57:1673–7.
van de Wouw HL, Awuyah EC, Baris JI, Klausen RS. An organoborane vinyl monomer with styrene-like radical reactivity: reactivity ratios and role of aromaticity. Macromolecules. 2018;51:6359–68.
Makino H, Nishikawa T, Ouchi M. Elucidating monomer character of an alkenyl boronate through radical copolymerization leads to copolymer synthesis beyond the limitation of copolymerizability by side-chain replacement. ACS Macro Lett. 2020;9:788–93.
Maki Y, Mori H, Endo T. Xanthate-mediated controlled radical polymerization of N-vinylindole derivatives. Macromolecules. 2007;40:6119–30.
Sugihara S, Kawamoto Y, Maeda Y. Direct radical polymerization of vinyl ethers: reversible addition—fragmentation chain transfer polymerization of hydroxy-functional vinyl ethers. Macromolecules. 2016;49:1563–74.
Sugihara S, Kawakami R, Maeda Y. Poly[di(ethylene glycol) vinyl ether]-stabilized poly(vinyl acetate) nanoparticles with various morphologies via RAFT aqueous emulsion polymerization of vinyl acetate. Polym J. 2021;53:309–21.
Abreu CMR, Mendonca PV, Serra AC, Coelho JFJ, Popov AV, Gryn’ova G, et al. Reversible addition-fragmentation chain transfer polymerization of vinyl chloride. Macromolecules. 2012;45:2200–8.
Oliveira M, Barbosa SB, Nele M, Pinto JC. Reversible addition-fragmentation chain transfer polymerization of vinyl acetate in bulk and suspension systems. Macromol React Eng. 2014;8:493–502.
Sugihara S, Yoshida A, Kono T, Takayama T, Maeda Y. Controlled radical homopolymerization of representative cationicallypolymerizable vinyl ethers. J Am Chem Soc. 2019;141:13954–61.
Coote ML, Henry DJ. Effect of substituents on radical stability in reversible addition fragmentation chain transfer polymerization: an ab initio study. Macromolecules. 2005;38:1415–33.
Mori H, Yahagi M, Endo T. RAFT polymerization of N-vinylimidazolium salts and synthesis of thermoresponsive ionic liquid block copolymers. Macromolecules. 2009;42:8082–92.
Soeriyadi AH, Boyer C, Burns J, Becer CR, Whittaker MR, Haddleton DM, et al. High fidelity vinyl terminated polymers by combining RAFT and cobaltcatalytic chain transfer (CCT) polymerization methods. Chem Commun. 2010;46:6338–40.
Andrews RJ, Grulke EA. Glass transition temperatures of polymers. In: Brandrup J, Immergut EH, Eric A Grulke EA, editors. Polymer handbook. 4th ed. New York: John Wiley and Sons; 1999.
Acknowledgements
We appreciate Mr. Takehiro Itami and Dr. Takaya Terashima for their kind advice. This work was partially supported by JSPS [KAKENHI grants 19K15622 (TN) and 17H06453 (MO)], the Mazda Foundation (TN), the Kyoto Technoscience Center (TN), the Masuya Memorial Foundation of the Promotion of Basic Research (TN), and the Hattori Hokokai Foundation (TN).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Kanazawa, T., Nishikawa, T. & Ouchi, M. RAFT polymerization of isopropenyl boronate pinacol ester and subsequent terminal olefination: precise synthesis of poly(alkenyl boronate)s and evaluation of their thermal properties. Polym J 53, 1167–1174 (2021). https://doi.org/10.1038/s41428-021-00498-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41428-021-00498-8