Abstract
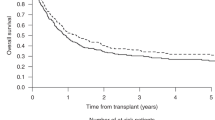
Reduced intensity conditioning (RIC) and nonmyeloablative (NMA) conditioning regimens have expanded use of allogeneic hematopoietic cell transplantation (HCT) in AML to include older and medically less-fit patients, but relative efficacies and toxicities remain poorly defined. Here, we analyzed outcomes from 343 adults transplanted in remission after RIC (n = 137) or NMA (n = 206) conditioning between 2006 and 2021. The characteristics of RIC and NMA HCT patients were similar except that RIC patients were younger and their time between most recent remission achievement and allografting was shorter. There were no significant differences in relapse risk, relapse-free survival (RFS), overall survival (OS), and non-relapse mortality (NRM) between RIC and NMA HCT patients, both overall (relapse: hazard ratio [HR] = 0.80, P = 0.27; RFS: HR = 0.93, P = 0.61; OS: HR = 0.93, P = 0.66; NRM: HR = 1.13, P = 0.59) and when patients were stratified by pre-HCT measurable residual disease (MRD) status. After multivariable adjustment, there was no statistically significant association between conditioning intensity and relapse (HR = 0.69, P = 0.088), RFS (HR = 0.86, P = 0.37), OS (HR = 0.89, P = 0.49), or NRM (HR = 1.37, P = 0.19). In this non-randomized cohort of adults undergoing allografting for AML in first or second remission at our center, we could not detect statistically significant differences in outcomes between those assigned to RIC and those assigned to NMA conditioning.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Döhner H, Wei AH, Appelbaum FR, Craddock C, Di Nardo CD, Dombret H, et al. Diagnosis and management of AML in adults: 2022 recommendations from an international expert panel on behalf of the ELN. Blood. 2022;140:1345–77.
Pingali SR, Champlin RE. Pushing the envelope-nonmyeloablative and reduced intensity preparative regimens for allogeneic hematopoietic transplantation. Bone Marrow Transpl. 2015;50:1157–67.
Jethava YS, Sica S, Savani B, Socola F, Jagasia M, Mohty M, et al. Conditioning regimens for allogeneic hematopoietic stem cell transplants in acute myeloid leukemia. Bone Marrow Transpl. 2017;52:1504–11.
Lipof JJ, Loh KP, O’Dwyer K, Liesveld JL. Allogeneic hematopoietic cell transplantation for older adults with acute myeloid leukemia. Cancers 2018;10:179.
Magliano G, Bacigalupo A. Allogeneic hematopoietic stem cell transplantation for acute myeloid leukemia of the elderly: review of literature and new perspectives. Mediterr J Hematol Infect Dis. 2020;12:e2020081.
Luger SM, Ringdén O, Zhang MJ, Pérez WS, Bishop MR, Bornhäuser M, et al. Similar outcomes using myeloablative vs reduced-intensity allogeneic transplant preparative regimens for AML or MDS. Bone Marrow Transpl. 2012;47:203–11.
Martino R, de Wreede L, Fiocco M, van Biezen A, von dem Borne PA, Hamladji RM. et al. Comparison of conditioning regimens of various intensities for allogeneic hematopoietic SCT using HLA-identical sibling donors in AML and MDS with <10% BM blasts: a report from EBMT. Bone Marrow Transpl. 2013;48:761–70.
Heinicke T, Labopin M, Polge E, Niederwieser D, Platzbecker U, Sengelov H, et al. Fludarabine/busulfan versus fludarabine/total-body-irradiation (2 Gy) as conditioning prior to allogeneic stem cell transplantation in patients (>/=60 years) with acute myelogenous leukemia: a study of the acute leukemia working party of the EBMT. Bone Marrow Transpl. 2020;55:729–39.
Blaise D, Tabrizi R, Boher JM, Le Corroller-Soriano AG, Bay JO, Fegueux N, et al. Randomized study of 2 reduced-intensity conditioning strategies for human leukocyte antigen-matched, related allogeneic peripheral blood stem cell transplantation: prospective clinical and socioeconomic evaluation. Cancer. 2013;119:602–11.
Arber DA, Orazi A, Hasserjian R, Thiele J, Borowitz MJ, Le Beau MM, et al. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood. 2016;127:2391–405.
Morsink LM, Othus M, Bezerra ED, Wood BL, Fang M, Sandmaier BM, et al. Impact of pre-transplant measurable residual disease on outcome of allogeneic hematopoietic cell transplantation in adult monosomal karyotype AML. Leukemia. 2020;34:1577–87.
Morsink LM, Sandmaier BM, Othus M, Palmieri R, Granot N, Bezerra ED et al. Conditioning intensity, pre-transplant flow cytometric measurable residual disease, and outcome in adults with acute myeloid leukemia undergoing allogeneic hematopoietic cell transplantation. Cancers. 2020;12:2339.
Paras G, Morsink LM, Othus M, Milano F, Sandmaier BM, Zarling LC, et al. Conditioning intensity and peritransplant flow cytometric MRD dynamics in adult AML. Blood. 2022;139:1694–706.
Zarling LC, Othus M, Sandmaier BM, Milano F, Schoch G, Davis C, et al. Utility of the Treatment-Related Mortality (TRM) score to predict outcomes of adults with acute myeloid leukemia undergoing allogeneic hematopoietic cell transplantation. Leukemia. 2022;36:1563–74.
Orvain C, Wilson JA, Fang M, Sandmaier BM, Rodríguez-Arbolí E, Wood BL et al. Relative impact of residual cytogenetic abnormalities and flow cytometric measurable residual disease on outcome after allogeneic hematopoietic cell transplantation in adult acute myeloid leukemia. Haematologica. 2022. https://doi.org/10.3324/haematol.2022.281585 Online ahead of print.
Rodríguez-Arbolí E, Orvain C, Othus M, Walter RB. Significance of measurable residual disease in adults with secondary acute myeloid leukemia undergoing allogeneic hematopoietic cell transplantation. Bone Marrow Transplant. 2022;57:1732-4.
Rodríguez-Arbolí E, Othus M, Orvain C, Zarling LC, Sandmaier BM, Milano F, et al. Contribution of measurable residual disease status to prediction accuracy of relapse and survival in adults with acute myeloid leukemia undergoing allogeneic hematopoietic cell transplantation. Haematologica. 2022. https://doi.org/10.3324/haematol.2022.281631 Online ahead of print.
Orvain C, Byelykh M, Othus M, Sandmaier BM, Schoch G, Davis C, et al. Relationship between pre-transplant nutritional status and outcomes of adults with acute myeloid leukemia undergoing allogeneic hematopoietic cell transplantation. Transplant Cell Ther. 2022;28:846.e1–9.
Sorror ML, Maris MB, Storb R, Baron F, Sandmaier BM, Maloney DG, et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood. 2005;106:2912–9.
Walter RB, Othus M, Borthakur G, Ravandi F, Cortes JE, Pierce SA, et al. Prediction of early death after induction therapy for newly diagnosed acute myeloid leukemia with pretreatment risk scores: a novel paradigm for treatment assignment. J Clin Oncol. 2011;29:4417–23.
Breems DA, Van Putten WLJ, De Greef GE, Van Zelderen-Bhola SL, Gerssen-Schoorl KBJ, Mellink CHM, et al. Monosomal karyotype in acute myeloid leukemia: a better indicator of poor prognosis than a complex karyotype. J Clin Oncol. 2008;26:4791–7.
Leisenring WM, Martin PJ, Petersdorf EW, Regan AE, Aboulhosn N, Stern JM, et al. An acute graft-versus-host disease activity index to predict survival after hematopoietic cell transplantation with myeloablative conditioning regimens. Blood. 2006;108:749–55.
Jagasia MH, Greinix HT, Arora M, Williams KM, Wolff D, Cowen EW, et al. National Institutes of Health Consensus Development Project on criteria for clinical trials in chronic graft-versus-host disease: I. The 2014 Diagnosis and Staging Working Group report. Biol Blood Marrow Transpl. 2015;21:389–401 e381.
Craddock C, Jackson A, Loke J, Siddique S, Hodgkinson A, Mason J, et al. Augmented reduced-intensity regimen does not improve postallogeneic transplant outcomes in acute myeloid leukemia. J Clin Oncol. 2021;39:768–78.
Walter RB, Gooley TA, Wood BL, Milano F, Fang M, Sorror ML, et al. Impact of pretransplantation minimal residual disease, as detected by multiparametric flow cytometry, on outcome of myeloablative hematopoietic cell transplantation for acute myeloid leukemia. J Clin Oncol. 2011;29:1190–7.
Walter RB, Buckley SA, Pagel JM, Wood BL, Storer BE, Sandmaier BM, et al. Significance of minimal residual disease before myeloablative allogeneic hematopoietic cell transplantation for AML in first and second complete remission. Blood. 2013;122:1813–21.
Walter RB, Gyurkocza B, Storer BE, Godwin CD, Pagel JM, Buckley SA, et al. Comparison of minimal residual disease as outcome predictor for AML patients in first complete remission undergoing myeloablative or nonmyeloablative allogeneic hematopoietic cell transplantation. Leukemia. 2015;29:137–44.
Araki D, Wood BL, Othus M, Radich JP, Halpern AB, Zhou Y, et al. Allogeneic hematopoietic cell transplantation for acute myeloid leukemia: is it time to move toward a minimal residual disease-based definition of complete remission. J Clin Oncol. 2016;34:329–36.
Zhou Y, Othus M, Araki D, Wood BL, Radich JP, Halpern AB, et al. Pre- and post-transplant quantification of measurable (‘minimal’) residual disease via multiparameter flow cytometry in adult acute myeloid leukemia. Leukemia. 2016;30:1456–64.
Wood BL. Acute myeloid leukemia minimal residual disease detection: the difference from normal approach. Curr Protoc Cytom. 2020;93:e73.
Walter RB, Sandmaier BM, Storer BE, Godwin CD, Buckley SA, Pagel JM, et al. Number of courses of induction therapy independently predicts outcome after allogeneic transplantation for acute myeloid leukemia in first morphological remission. Biol Blood Marrow Transpl. 2015;21:373–8.
Hoffmann AP, Besch AL, Othus M, Morsink LM, Wood BL, Mielcarek M, et al. Early achievement of measurable residual disease (MRD)-negative complete remission as predictor of outcome after myeloablative allogeneic hematopoietic cell transplantation in acute myeloid leukemia. Bone Marrow Transpl. 2020;55:669–72.
Morsink LM, Bezerra ED, Othus M, Wood BL, Fang M, Sandmaier BM, et al. Comparative analysis of total body irradiation (TBI)-based and non-TBI-based myeloablative conditioning for acute myeloid leukemia in remission with or without measurable residual disease. Leukemia. 2020;34:1701–5.
Gyurkocza B, Storb R, Storer BE, Chauncey TR, Lange T, Shizuru JA, et al. Nonmyeloablative allogeneic hematopoietic cell transplantation in patients with acute myeloid leukemia. J Clin Oncol. 2010;28:2859–67.
Krakow EF, Gyurkocza B, Storer BE, Chauncey TR, McCune JS, Radich JP, et al. Phase I/II multisite trial of optimally dosed clofarabine and low-dose TBI for hematopoietic cell transplantation in acute myeloid leukemia. Am J Hematol. 2020;95:48–56.
Scott BL, Pasquini MC, Logan BR, Wu J, Devine SM, Porter DL, et al. Myeloablative versus reduced-intensity hematopoietic cell transplantation for acute myeloid leukemia and myelodysplastic syndromes. J Clin Oncol. 2017;35:1154–61.
Scott BL, Pasquini MC, Fei M, Fraser R, Wu J, Devine SM, et al. Myeloablative versus reduced-Intensity conditioning for hematopoietic cell transplantation in acute myelogenous leukemia and myelodysplastic syndromes-long-term follow-up of the BMT CTN 0901 clinical trial. Transpl Cell Ther. 2021;27:483 e481–483 e486.
Hourigan CS, Dillon LW, Gui G, Logan BR, Fei M, Ghannam J, et al. Impact of conditioning intensity of allogeneic transplantation for acute myeloid leukemia with genomic evidence of residual disease. J Clin Oncol. 2020;38:1273–83.
Buckley SA, Wood BL, Othus M, Hourigan CS, Ustun C, Linden MA, et al. Minimal residual disease prior to allogeneic hematopoietic cell transplantation in acute myeloid leukemia: a meta-analysis. Haematologica. 2017;102:865–73.
Hourigan CS, Gale RP, Gormley NJ, Ossenkoppele GJ, Walter RB. Measurable residual disease testing in acute myeloid leukaemia. Leukemia. 2017;31:1482–90.
Acknowledgements
Research reported in this publication was supported by grants P01-CA078902, P01-CA018029, and P30-CA015704 from the National Cancer Institute/National Institutes of Health (NCI/NIH), Bethesda, MD, USA. The authors acknowledge the excellent care provided by the physicians advanced practice providers, dietitians, pharmacists, and nurses of the HCT teams, the staff in the Long-Term Follow-up office at the Fred Hutchinson Cancer Center, the Hematopathology Laboratory at the University of Washington, and the patients for participating in our research protocols.
Author information
Authors and Affiliations
Contributions
RBW, BMS, and RS conceptualized and designed this study and participated in data analysis and interpretation and drafting of the manuscript. BMS, MUO, HJD, and RS contributed to the provision of study material, patient recruitment, and acquisition of data. MO conducted all statistical analyses and participated in data interpretation. CO, ERA, GS, and CD contributed to the collection and assembly of data. All authors revised the manuscript critically and gave final approval to submit for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Walter, R.B., Sandmaier, B.M., Othus, M. et al. Comparison of reduced intensity and nonmyeloablative conditioning for adults with acute myeloid leukemia undergoing allogeneic hematopoietic cell transplantation in first or second remission. Bone Marrow Transplant 58, 377–385 (2023). https://doi.org/10.1038/s41409-022-01909-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-022-01909-x