Key Points
-
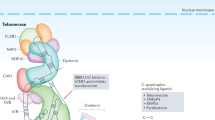
Telomerase activation or the cancer-specific, telomerase-independent alternative lengthening of telomeres (ALT) mechanism are two telomere maintenance mechanisms in human cells. Most prostate cancers activate telomerase and a subset of lethal metastases use ALT
-
Substantial telomere shortening is common in prostate cancers and in the precursor lesion prostatic intraepithelial neoplasia (PIN). Moderate telomere shortening has also been observed in cancer-associated stroma
-
The mechanisms for telomere shortening in prostate cancer and PIN are not fully understood; in addition to replication-associated telomere loss, inflammation and reactive oxygen species might be contributors
-
Telomere length assessment might be useful in prostate cancer diagnosis and in current prognostic tools to more reliably predict whether organ-confined prostate cancer will progress to lethal metastatic disease
-
Telomerase-targeted single-agent treatments for solid cancers have, to date, been ineffective in clinical trials; these therapies have yet to be tested in prostate cancer and might potentially be useful in combination with established androgen receptor (AR)-targeted treatments
-
Disruption of AR function in AR-positive prostate cancer cells activates the DNA damage response (DDR) at telomeres; thus, DDR inhibitors might potentiate the effects of androgen deprivation therapy
Abstract
Aberrations in telomere biology are among the earliest events in prostate cancer tumorigenesis and continue during tumour progression. Substantial telomere shortening occurs in prostate cancer cells and high-grade prostatic intraepithelial neoplasia. Not all mechanisms of telomere shortening are understood, but oxidative stress from local inflammation might accelerate prostatic telomere loss. Critically short telomeres can drive the accumulation of tumour-promoting genomic alterations; however, continued telomere erosion is unsustainable and must be mitigated to ensure cancer cell survival and unlimited replication potential. Prostate cancers predominantly maintain telomeres by activating telomerase, but alternative mechanisms of telomere extension can occur in metastatic disease. Telomerase activity and telomere length assessment might be useful in prostate cancer diagnosis and prognosis. Telomere shortening in normal stromal cells has been associated with prostate cancer, whereas variable telomere lengths in prostate cancer cells and telomere shortening in cancer-associated stromal cells correlated with lethal disease. Single-agent telomerase-targeted treatments for solid cancers were ineffective in clinical trials but have not been investigated in prostate cancer and might be useful in combination with established regimens. Telomere-directed strategies have not been explored as extensively. Telomere deprotection strategies have the advantage of being effective in both telomerase-dependent and telomerase-independent cancers. Disruption of androgen receptor function in prostate cancer cells results in telomere dysfunction, indicating telomeres and telomerase as potential therapeutic targets in prostate cancer.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Murray, A. W. & Szostak, J. W. Construction of artificial chromosomes in yeast. Nature 305, 189–193 (1983).
Hsiao, C. L. & Carbon, J. High-frequency transformation of yeast by plasmids containing the cloned yeast ARG4 gene. Proc. Natl Acad. Sci. USA 76, 3829–3833 (1979).
Stinchcomb, D. T., Struhl, K. & Davis, R. W. Isolation and characterisation of a yeast chromosomal replicator. Nature 282, 39–43 (1979).
Clarke, L. & Carbon, J. Isolation of a yeast centromere and construction of functional small circular chromosomes. Nature 287, 504–509 (1980).
Stinchcomb, D. T., Mann, C. & Davis, R. W. Centromeric DNA from Saccharomyces cerevisiae. J. Mol. Biol. 158, 157–190 (1982).
Szostak, J. W. & Blackburn, E. H. Cloning yeast telomeres on linear plasmid vectors. Cell 29, 245–255 (1982).
Blackburn, E. H. & Challoner, P. B. Identification of a telomeric DNA sequence in Trypanosoma brucei. Cell 36, 447–457 (1984).
Moyzis, R. K. et al. A highly conserved repetitive DNA sequence, (TTAGGG)n, present at the telomeres of human chromosomes. Proc. Natl Acad. Sci. USA 85, 6622–6626 (1988).
Samassekou, O., Gadji, M., Drouin, R. & Yan, J. Sizing the ends: normal length of human telomeres. Ann. Anat. 192, 284–291 (2010).
de Lange, T. Shelterin: the protein complex that shapes and safeguards human telomeres. Genes Dev. 19, 2100–2110 (2005).
Zhong, Z., Shiue, L., Kaplan, S. & de Lange, T. A mammalian factor that binds telomeric TTAGGG repeats in vitro. Mol. Cell. Biol. 12, 4834–4843 (1992).
Chong, L. et al. A human telomeric protein. Science 270, 1663–1667 (1995).
Bilaud, T. et al. Telomeric localization of TRF2, a novel human telobox protein. Nat. Genet. 17, 236–239 (1997).
Broccoli, D., Smogorzewska, A., Chong, L. & de Lange, T. Human telomeres contain two distinct Myb-related proteins, TRF1 and TRF2. Nat. Genet. 17, 231–235 (1997).
Kim, S. H., Kaminker, P. & Campisi, J. TIN2, a new regulator of telomere length in human cells. Nat. Genet. 23, 405–412 (1999).
Li, B., Oestreich, S. & de Lange, T. Identification of human Rap1: implications for telomere evolution. Cell 101, 471–483 (2000).
Houghtaling, B. R., Cuttonaro, L., Chang, W. & Smith, S. A dynamic molecular link between the telomere length regulator TRF1 and the chromosome end protector TRF2. Curr. Biol. 14, 1621–1631 (2004).
Liu, D. et al. PTOP interacts with POT1 and regulates its localization to telomeres. Nat. Cell Biol. 6, 673–680 (2004).
Ye, J. Z. et al. POT1-interacting protein PIP1: a telomere length regulator that recruits POT1 to the TIN2/TRF1 complex. Genes Dev. 18, 1649–1654 (2004).
Jones, M. et al. The shelterin complex and hematopoiesis. J. Clin. Invest. 126, 1621–1629 (2016).
Griffith, J. D. et al. Mammalian telomeres end in a large duplex loop. Cell 97, 503–514 (1999).
Hayflick, L. The limited in vitro lifetime of human diploid cell strains. Exp. Cell Res. 37, 614–636 (1965).
Hayflick, L. & Moorhead, P. S. The serial cultivation of human diploid cell strains. Exp. Cell Res. 25, 585–621 (1961).
Watson, J. D. Origin of concatemeric T7 DNA. Nat. New Biol. 239, 197–201 (1972).
Olovnikov, A. M. A theory of marginotomy. The incomplete copying of template margin in enzymic synthesis of polynucleotides and biological significance of the phenomenon. J. Theor. Biol. 41, 181–190 (1973).
Levy, M. Z., Allsopp, R. C., Futcher, A. B., Greider, C. W. & Harley, C. B. Telomere end-replication problem and cell aging. J. Mol. Biol. 225, 951–960 (1992).
Fumagalli, M. et al. Telomeric DNA damage is irreparable and causes persistent DNA-damage-response activation. Nat. Cell Biol. 14, 355–365 (2012).
von Zglinicki, T., Saretzki, G., Ladhoff, J., d' Adda di Fagagna, F. & Jackson, S. P. Human cell senescence as a DNA damage response. Mech. Ageing Dev. 126, 111–117 (2005).
Zou, Y., Sfeir, A., Gryaznov, S. M., Shay, J. W. & Wright, W. E. Does a sentinel or a subset of short telomeres determine replicative senescence? Mol. Biol. Cell 15, 3709–3718 (2004).
Blasco, M. A. et al. Telomere shortening and tumor formation by mouse cells lacking telomerase RNA. Cell 91, 25–34 (1997).
McClintock, B. The stability of broken ends of chromosomes in Zea Mays. Genetics 26, 234–282 (1941).
Counter, C. M. et al. Telomere shortening associated with chromosome instability is arrested in immortal cells which express telomerase activity. EMBO J. 11, 1921–1929 (1992).
Chin, L. et al. p53 deficiency rescues the adverse effects of telomere loss and cooperates with telomere dysfunction to accelerate carcinogenesis. Cell 97, 527–538 (1999).
Hackett, J. A., Feldser, D. M. & Greider, C. W. Telomere dysfunction increases mutation rate and genomic instability. Cell 106, 275–286 (2001).
Shay, J. W. & Bacchetti, S. A survey of telomerase activity in human cancer. Eur. J. Cancer 33, 787–791 (1997).
Dunham, M. A., Neumann, A. A., Fasching, C. L. & Reddel, R. R. Telomere maintenance by recombination in human cells. Nat. Genet. 26, 447–450 (2000).
Henson, J. D., Neumann, A. A., Yeager, T. R. & Reddel, R. R. Alternative lengthening of telomeres in mammalian cells. Oncogene 21, 598–610 (2002).
Haffner, M. C. et al. Tracking the clonal origin of lethal prostate cancer. J. Clin. Invest. 123, 4918–4922 (2013).
Sommerfeld, H. J. et al. Telomerase activity: a prevalent marker of malignant human prostate tissue. Cancer Res. 56, 218–222 (1996).
Greider, C. W. & Blackburn, E. H. The telomere terminal transferase of Tetrahymena is a ribonucleoprotein enzyme with two kinds of primer specificity. Cell 51, 887–898 (1987).
Podlevsky, J. D., Bley, C. J., Omana, R. V., Qi, X. & Chen, J. J. The telomerase database. Nucleic Acids Res. 36, D339–343 (2008).
Greider, C. W. & Blackburn, E. H. A telomeric sequence in the RNA of Tetrahymena telomerase required for telomere repeat synthesis. Nature 337, 331–337 (1989).
Shippen-Lentz, D. & Blackburn, E. H. Functional evidence for an RNA template in telomerase. Science 247, 546–552 (1990).
Batista, L. F. Telomere biology in stem cells and reprogramming. Prog. Mol. Biol. Transl Sci. 125, 67–88 (2014).
Allshire, R. C., Dempster, M. & Hastie, N. D. Human telomeres contain at least three types of G-rich repeat distributed non-randomly. Nucleic Acids Res. 17, 4611–4627 (1989).
Berges, R. R. et al. Implication of cell kinetic changes during the progression of human prostatic cancer. Clin. Cancer Res. 1, 473–480 (1995).
Meeker, A. K. et al. Telomere shortening is an early somatic DNA alteration in human prostate tumorigenesis. Cancer Res. 62, 6405–6409 (2002).
Kyprianou, N., Tu, H. & Jacobs, S. C. Apoptotic versus proliferative activities in human benign prostatic hyperplasia. Hum. Pathol. 27, 668–675 (1996).
Helpap, B. Cell kinetic studies on prostatic intraepithelial neoplasia (PIN) and atypical adenomatous hyperplasia (AAH) of the prostate. Pathol. Res. Pract. 191, 904–907 (1995).
De Marzo, A. M., Nelson, W. G., Meeker, A. K. & Coffey, D. S. Stem cell features of benign and malignant prostate epithelial cells. J. Urol. 160, 2381–2392 (1998).
Heatfield, B. M., Sanefuji, H. & Trump, B. F. Studies on carcinogenesis of human prostate. III. Long-term explant culture of normal prostate and benign prostatic hyperplasia: transmission and scanning electron microscopy. J. Natl Cancer Inst. 69, 757–766 (1982).
Merchant, D. J., Clarke, S. M., Ives, K. & Harris, S. Primary explant culture: an in vitro model of the human prostate. Prostate 4, 523–542 (1983).
Bonkhoff, H., Stein, U. & Remberger, K. The proliferative function of basal cells in the normal and hyperplastic human prostate. Prostate 24, 114–118 (1994).
Bostwick, D. G. & Brawer, M. K. Prostatic intra-epithelial neoplasia and early invasion in prostate cancer. Cancer 59, 788–794 (1987).
Rohr, H. P. & Bartsch, G. Human benign prostatic hyperplasia: a stromal disease? New perspectives by quantitative morphology. Urology 16, 625–633 (1980).
McNeal, J. E., Haillot, O. & Yemoto, C. Cell proliferation in dysplasia of the prostate: analysis by PCNA immunostaining. Prostate 27, 258–268 (1995).
Rane, J. K. et al. Telomerase activity and telomere length in human benign prostatic hyperplasia stem-like cells and their progeny implies the existence of distinct basal and luminal cell lineages. Eur. Urol. 69, 551–554 (2016).
Bostwick, D. G. Prostatic intraepithelial neoplasia (PIN): current concepts. J. Cell Biochem. Suppl 16H, 10–19 (1992).
Mostofi, F. K., Sesterhenn, I. A. & Davis, C. J. Jr. Prostatic intraepithelial neoplasia (PIN): morphological clinical significance. Prostate Suppl. 4, 71–77 (1992).
Koeneman, K. S. et al. Telomerase activity, telomere length, and DNA ploidy in prostatic intraepithelial neoplasia (PIN). J. Urol. 160, 1533–1539 (1998).
Zhang, W., Kapusta, L. R., Slingerland, J. M. & Klotz, L. H. Telomerase activity in prostate cancer, prostatic intraepithelial neoplasia, and benign prostatic epithelium. Cancer Res. 58, 619–621 (1998).
National Cancer Institute (2016).
Thompson, S. J. et al. P53 and Ki-67 immunoreactivity in human prostate cancer and benign hyperplasia. Br. J. Urol. 69, 609–613 (1992).
Shay, J. W. & Wright, W. E. Hayflick, his limit, and cellular ageing. Nat. Rev. Mol. Cell Biol. 1, 72–76 (2000).
Capper, R. et al. The nature of telomere fusion and a definition of the critical telomere length in human cells. Genes Dev. 21, 2495–2508 (2007).
Muller, F. The nature and mechanism of superoxide production by the electron transport chain: its relevance to aging. J. Am. Aging Assoc. 23, 227–253 (2000).
Kryston, T. B., Georgiev, A. B., Pissis, P. & Georgakilas, A. G. Role of oxidative stress and DNA damage in human carcinogenesis. Mutat. Res. 711, 193–201 (2011).
Halliwell, B. Superoxide-dependent formation of hydroxyl radicals in the presence of iron salts. Its role in degradation of hyaluronic acid by a superoxide-generating system. FEBS Lett. 96, 238–242 (1978).
Liochev, S. I. & Fridovich, I. The role of O2.- in the production of HO.: in vitro and in vivo. Free Radic. Biol. Med. 16, 29–33 (1994).
McCord, J. M. & Day, E. D. Jr. Superoxide-dependent production of hydroxyl radical catalyzed by iron-EDTA complex. FEBS Lett. 86, 139–142 (1978).
Burrows, C. J. & Muller, J. G. Oxidative nucleobase modifications leading to strand scission. Chem. Rev. 98, 1109–1152 (1998).
Gajewski, E., Rao, G., Nackerdien, Z. & Dizdaroglu, M. Modification of DNA bases in mammalian chromatin by radiation-generated free radicals. Biochemistry 29, 7876–7882 (1990).
Kawanishi, S. & Oikawa, S. Mechanism of telomere shortening by oxidative stress. Ann. NY Acad. Sci. 1019, 278–284 (2004).
Fortini, P. et al. 8-Oxoguanine DNA damage: at the crossroad of alternative repair pathways. Mutat. Res. 531, 127–139 (2003).
Wilson, D. M., 3rd & Bohr, V. A. The mechanics of base excision repair, and its relationship to aging and disease. DNA Repair (Amst.) 6, 544–559 (2007).
Sitte, N., Saretzki, G. & von Zglinicki, T. Accelerated telomere shortening in fibroblasts after extended periods of confluency. Free Radic. Biol. Med. 24, 885–893 (1998).
Petersen, S., Saretzki, G. & von Zglinicki, T. Preferential accumulation of single-stranded regions in telomeres of human fibroblasts. Exp. Cell Res. 239, 152–160 (1998).
Kruk, P. A., Rampino, N. J. & Bohr, V. A. DNA damage and repair in telomeres: relation to aging. Proc. Natl Acad. Sci. USA 92, 258–262 (1995).
Webb, C. J., Wu, Y. & Zakian, V. A. DNA repair at telomeres: keeping the ends intact. Cold Spring Harb Perspect. Biol. 5, a012666 (2013).
Rhodes, D. & Lipps, H. J. G-Quadruplexes and their regulatory roles in biology. Nucleic Acids Res. 43, 8627–8637 (2015).
Sfeir, A. et al. Mammalian telomeres resemble fragile sites and require TRF1 for efficient replication. Cell 138, 90–103 (2009).
von Zglinicki, T. Oxidative stress shortens telomeres. Trends Biochem. Sci. 27, 339–344 (2002).
Mittal, M., Siddiqui, M. R., Tran, K., Reddy, S. P. & Malik, A. B. Reactive oxygen species in inflammation and tissue injury. Antioxid. Redox Signal 20, 1126–1167 (2014).
Mantovani, A., Allavena, P., Sica, A. & Balkwill, F. Cancer-related inflammation. Nature 454, 436–444 (2008).
De Marzo, A. M. et al. Inflammation in prostate carcinogenesis. Nat. Rev. Cancer 7, 256–269 (2007).
Sfanos, K. S. & De Marzo, A. M. Prostate cancer and inflammation: the evidence. Histopathology 60, 199–215 (2012).
Gurel, B. et al. Chronic inflammation in benign prostate tissue is associated with high-grade prostate cancer in the placebo arm of the prostate cancer prevention trial. Cancer Epidemiol. Biomarkers Prev. 23, 847–856 (2014).
Vidal, A. C. et al. Aspirin, NSAIDs, and risk of prostate cancer: results from the REDUCE study. Clin. Cancer Res. 21, 756–762 (2015).
Stimac, G. et al. Aggressiveness of inflammation in histological prostatitis—correlation with total and free prostate specific antigen levels in men with biochemical criteria for prostate biopsy. Scott. Med. J. 54, 8–12 (2009).
Fujita, K. et al. Prostatic inflammation detected in initial biopsy specimens and urinary pyuria are predictors of negative repeat prostate biopsy. J. Urol. 185, 1722–1727 (2011).
Delongchamps, N. B. et al. Evaluation of prostatitis in autopsied prostates—is chronic inflammation more associated with benign prostatic hyperplasia or cancer? J. Urol. 179, 1736–1740 (2008).
Nickel, J. C., Downey, J., Young, I. & Boag, S. Asymptomatic inflammation and/or infection in benign prostatic hyperplasia. BJU Int. 84, 976–981 (1999).
Nickel, J. C. et al. The relationship between prostate inflammation and lower urinary tract symptoms: examination of baseline data from the REDUCE trial. Eur. Urol. 54, 1379–1384 (2008).
Sfanos, K. S., Isaacs, W. B. & De Marzo, A. M. Infections and inflammation in prostate cancer. Am. J. Clin. Exp. Urol. 1, 3–11 (2013).
Sfanos, K. S., Wilson, B. A., De Marzo, A. M. & Isaacs, W. B. Acute inflammatory proteins constitute the organic matrix of prostatic corpora amylacea and calculi in men with prostate cancer. Proc. Natl Acad. Sci. USA 106, 3443–3448 (2009).
De Marzo, A. M., Marchi, V. L., Epstein, J. I. & Nelson, W. G. Proliferative inflammatory atrophy of the prostate: implications for prostatic carcinogenesis. Am. J. Pathol. 155, 1985–1992 (1999).
Feneley, M. R., Young, M. P., Chinyama, C., Kirby, R. S. & Parkinson, M. C. Ki-67 expression in early prostate cancer and associated pathological lesions. J. Clin. Pathol. 49, 741–748 (1996).
Ruska, K. M., Sauvageot, J. & Epstein, J. I. Histology and cellular kinetics of prostatic atrophy. Am. J. Surg. Pathol. 22, 1073–1077 (1998).
van Leenders, G. J. et al. Intermediate cells in human prostate epithelium are enriched in proliferative inflammatory atrophy. Am. J. Pathol. 162, 1529–1537 (2003).
Putzi, M. J. & De Marzo, A. M. Morphologic transitions between proliferative inflammatory atrophy and high-grade prostatic intraepithelial neoplasia. Urology 56, 828–832 (2000).
Aizer, A. A. et al. Lack of reduction in racial disparities in cancer-specific mortality over a 20-year period. Cancer 120, 1532–1539 (2014).
Eastham, J. A. et al. Clinical characteristics and biopsy specimen features in African-American and white men without prostate cancer. J. Natl Cancer Inst. 90, 756–760 (1998).
Peto, J. Cancer epidemiology in the last century and the next decade. Nature 411, 390–395 (2001).
Hsu, A., Bray, T. M. & Ho, E. Anti-inflammatory activity of soy and tea in prostate cancer prevention. Exp. Biol. Med. (Maywood) 235, 659–667 (2010).
Heaphy, C. M. et al. Prostate cancer cell telomere length variability and stromal cell telomere length as prognostic markers for metastasis and death. Cancer Discov. 3, 1130–1141 (2013).
De Nunzio, C., Presicce, F. & Tubaro, A. Inflammatory mediators in the development and progression of benign prostatic hyperplasia. Nat. Rev. Urol. 13, 613–626 (2016).
Cookson, M. S., Reuter, V. E., Linkov, I. & Fair, W. R. Glutathione S-transferase PI (GST-pi) class expression by immunohistochemistry in benign and malignant prostate tissue. J. Urol. 157, 673–676 (1997).
Lee, W. H. et al. Cytidine methylation of regulatory sequences near the pi-class glutathione S-transferase gene accompanies human prostatic carcinogenesis. Proc. Natl Acad. Sci. USA 91, 11733–11737 (1994).
Mian, O. Y. et al. GSTP1 Loss results in accumulation of oxidative DNA base damage and promotes prostate cancer cell survival following exposure to protracted oxidative stress. Prostate 76, 199–206 (2016).
Kanwal, R. et al. Protection against oxidative DNA damage and stress in human prostate by glutathione S-transferase P1. Mol. Carcinog. 53, 8–18 (2014).
Stephens, P. J. et al. Massive genomic rearrangement acquired in a single catastrophic event during cancer development. Cell 144, 27–40 (2011).
Malhotra, A. et al. Breakpoint profiling of 64 cancer genomes reveals numerous complex rearrangements spawned by homology-independent mechanisms. Genome Res. 23, 762–776 (2013).
Zack, T. I. et al. Pan-cancer patterns of somatic copy number alteration. Nat. Genet. 45, 1134–1140 (2013).
Storchova, Z. & Kloosterman, W. P. The genomic characteristics and cellular origin of chromothripsis. Curr. Opin. Cell Biol. 40, 106–113 (2016).
Rausch, T. et al. Genome sequencing of pediatric medulloblastoma links catastrophic DNA rearrangements with TP53 mutations. Cell 148, 59–71 (2012).
Molenaar, J. J. et al. Sequencing of neuroblastoma identifies chromothripsis and defects in neuritogenesis genes. Nature 483, 589–593 (2012).
Wu, C. et al. Poly-gene fusion transcripts and chromothripsis in prostate cancer. Genes Chromosomes Cancer 51, 1144–1153 (2012).
Maciejowski, J., Li, Y., Bosco, N., Campbell, P. J. & de Lange, T. Chromothripsis and kataegis induced by telomere crisis. Cell 163, 1641–1654 (2015).
Kovtun, I. V., Murphy, S. J., Johnson, S. H., Cheville, J. C. & Vasmatzis, G. Chromosomal catastrophe is a frequent event in clinically insignificant prostate cancer. Oncotarget 6, 29087–29096 (2015).
Tu, L. et al. Widespread telomere instability in prostatic lesions. Mol. Carcinog. 55, 842–852 (2016).
Feijoo, P., Dominguez, D., Tusell, L. & Genesca, A. Telomere-dependent genomic integrity: evolution of the fusion-bridge-breakage cycle concept. Curr. Pharm. Des. 20, 6375–6385 (2014).
Vukovic, B. et al. Correlating breakage-fusion-bridge events with the overall chromosomal instability and in vitro karyotype evolution in prostate cancer. Cytogenet. Genome Res. 116, 1–11 (2007).
Xi, L. & Cech, T. R. Inventory of telomerase components in human cells reveals multiple subpopulations of hTR and hTERT. Nucleic Acids Res. 42, 8565–8577 (2014).
Cohen, S. B. et al. Protein composition of catalytically active human telomerase from immortal cells. Science 315, 1850–1853 (2007).
Kilian, A. et al. Isolation of a candidate human telomerase catalytic subunit gene, which reveals complex splicing patterns in different cell types. Hum. Mol. Genet. 6, 2011–2019 (1997).
Nakamura, T. M. et al. Telomerase catalytic subunit homologs from fission yeast and human. Science 277, 955–959 (1997).
Bodnar, A. G. et al. Extension of life-span by introduction of telomerase into normal human cells. Science 279, 349–352 (1998).
Counter, C. M. et al. Telomerase activity is restored in human cells by ectopic expression of hTERT (hEST2), the catalytic subunit of telomerase. Oncogene 16, 1217–1222 (1998).
Vaziri, H. & Benchimol, S. Reconstitution of telomerase activity in normal human cells leads to elongation of telomeres and extended replicative life span. Curr. Biol. 8, 279–282 (1998).
Nieto, C. M., Rider, L. C. & Cramer, S. D. Influence of stromal-epithelial interactions on androgen action. Endocr. Relat. Cancer 21, T147–160 (2014).
Meeker, A. K., Sommerfeld, H. J. & Coffey, D. S. Telomerase is activated in the prostate and seminal vesicles of the castrated rat. Endocrinology 137, 5743–5746 (1996).
Ravindranath, N. et al. Androgen depletion activates telomerase in the prostate of the nonhuman primate, Macaca mulatta. Prostate 49, 79–89 (2001).
Moehren, U. et al. Wild-type but not mutant androgen receptor inhibits expression of the hTERT telomerase subunit: a novel role of AR mutation for prostate cancer development. FASEB J. 22, 1258–1267 (2008).
Guo, C., Armbruster, B. N., Price, D. T. & Counter, C. M. In vivo regulation of hTERT expression and telomerase activity by androgen. J. Urol. 170, 615–618 (2003).
Cho, S. D. et al. Methyl selenium metabolites decrease prostate-specific antigen expression by inducing protein degradation and suppressing androgen-stimulated transcription. Mol. Cancer Ther. 3, 605–611 (2004).
Liu, S. et al. Telomerase as an important target of androgen signaling blockade for prostate cancer treatment. Mol. Cancer Ther. 9, 2016–2025 (2010).
Renaud, S. et al. Dual role of DNA methylation inside and outside of CTCF-binding regions in the transcriptional regulation of the telomerase hTERT gene. Nucleic Acids Res. 35, 1245–1256 (2007).
Killela, P. J. et al. TERT promoter mutations occur frequently in gliomas and a subset of tumors derived from cells with low rates of self-renewal. Proc. Natl Acad. Sci. USA 110, 6021–6026 (2013).
Vinagre, J. et al. Frequency of TERT promoter mutations in human cancers. Nat. Commun. 4, 2185 (2013).
Stoehr, R. et al. Frequency of TERT Promoter Mutations in Prostate Cancer. Pathobiology 82, 53–57 (2015).
Gurel, B. et al. Nuclear MYC protein overexpression is an early alteration in human prostate carcinogenesis. Mod. Pathol. 21, 1156–1167 (2008).
Lapointe, J. et al. Gene expression profiling identifies clinically relevant subtypes of prostate cancer. Proc. Natl Acad. Sci. USA 101, 811–816 (2004).
Dhanasekaran, S. M. et al. Molecular profiling of human prostate tissues: insights into gene expression patterns of prostate development during puberty. FASEB J. 19, 243–245 (2005).
Varambally, S. et al. Integrative genomic and proteomic analysis of prostate cancer reveals signatures of metastatic progression. Cancer Cell 8, 393–406 (2005).
Tomlins, S. A. et al. Integrative molecular concept modeling of prostate cancer progression. Nat. Genet. 39, 41–51 (2007).
Yu, Y. P. et al. Gene expression alterations in prostate cancer predicting tumor aggression and preceding development of malignancy. J. Clin. Oncol. 22, 2790–2799 (2004).
Latil, A. et al. htert expression correlates with MYC over-expression in human prostate cancer. Int. J. Cancer 89, 172–176 (2000).
Wu, K. J. et al. Direct activation of TERT transcription by c-MYC. Nat. Genet. 21, 220–224 (1999).
Kyo, S. et al. Sp1 cooperates with c-Myc to activate transcription of the human telomerase reverse transcriptase gene (hTERT). Nucleic Acids Res. 28, 669–677 (2000).
Jenkins, R. B., Qian, J., Lieber, M. M. & Bostwick, D. G. Detection of c-myc oncogene amplification and chromosomal anomalies in metastatic prostatic carcinoma by fluorescence in situ hybridization. Cancer Res. 57, 524–531 (1997).
Nupponen, N. N., Kakkola, L., Koivisto, P. & Visakorpi, T. Genetic alterations in hormone-refractory recurrent prostate carcinomas. Am. J. Pathol. 153, 141–148 (1998).
Koh, C. M. et al. MYC and Prostate Cancer. Genes Cancer 1, 617–628 (2010).
Andriole, G. L. et al. Mortality results from a randomized prostate-cancer screening trial. N. Engl. J. Med. 360, 1310–1319 (2009).
Schroder, F. H. et al. Screening and prostate-cancer mortality in a randomized European study. N. Engl. J. Med. 360, 1320–1328 (2009).
Morin, G. B. The human telomere terminal transferase enzyme is a ribonucleoprotein that synthesizes TTAGGG repeats. Cell 59, 521–529 (1989).
Kim, N. W. et al. Specific association of human telomerase activity with immortal cells and cancer. Science 266, 2011–2015 (1994).
Zhou, X. & Xing, D. Assays for human telomerase activity: progress and prospects. Chem. Soc. Rev. 41, 4643–4656 (2012).
Fizazi, K. et al. High detection rate of circulating tumor cells in blood of patients with prostate cancer using telomerase activity. Ann. Oncol. 18, 518–521 (2007).
Xu, T., Lu, B., Tai, Y. C. & Goldkorn, A. A cancer detection platform which measures telomerase activity from live circulating tumor cells captured on a microfilter. Cancer Res. 70, 6420–6426 (2010).
Goldkorn, A. et al. Circulating tumor cell telomerase activity as a prognostic marker for overall survival in SWOG 0421: a phase III metastatic castration resistant prostate cancer trial. Int. J. Cancer 136, 1856–1862 (2015).
Heaphy, C. M. et al. Prostate stromal cell telomere shortening is associated with risk of prostate cancer in the placebo arm of the Prostate Cancer Prevention Trial. Prostate 75, 1160–1166 (2015).
Loeb, S. et al. Overdiagnosis and overtreatment of prostate cancer. Eur. Urol. 65, 1046–1055 (2014).
Shay, J. W., Reddel, R. R. & Wright, W. E. Cancer. Cancer and telomeres—an ALTernative to telomerase. Science 336, 1388–1390 (2012).
Shay, J. W., Zou, Y., Hiyama, E. & Wright, W. E. Telomerase and cancer. Hum. Mol. Genet. 10, 677–685 (2001).
Williams, S. C. No end in sight for telomerase-targeted cancer drugs. Nat. Med. 19, 6 (2013).
Jafri, M. A., Ansari, S. A., Alqahtani, M. H. & Shay, J. W. Roles of telomeres and telomerase in cancer, and advances in telomerase-targeted therapies. Genome Med. 8, 69 (2016).
Chiappori, A. A. et al. A randomized phase II study of the telomerase inhibitor imetelstat as maintenance therapy for advanced non-small-cell lung cancer. Ann. Oncol. 26, 354–362 (2015).
Baerlocher, G. M. et al. Telomerase inhibitor imetelstat in patients with essential thrombocythemia. N. Engl. J. Med. 373, 920–928 (2015).
Rousseau, P. & Autexier, C. Telomere biology: Rationale for diagnostics and therapeutics in cancer. RNA Biol. 12, 1078–1082 (2015).
Marian, C. O., Wright, W. E. & Shay, J. W. The effects of telomerase inhibition on prostate tumor-initiating cells. Int. J. Cancer 127, 321–331 (2010).
Kawashima, T. et al. Telomerase-specific replication-selective virotherapy for human cancer. Clin. Cancer Res. 10, 285–292 (2004).
Nemunaitis, J. et al. A phase I study of telomerase-specific replication competent oncolytic adenovirus (telomelysin) for various solid tumors. Mol. Ther. 18, 429–434 (2010).
Middleton, G. et al. Gemcitabine and capecitabine with or without telomerase peptide vaccine GV1001 in patients with locally advanced or metastatic pancreatic cancer (TeloVac): an open-label, randomised, phase 3 trial. Lancet Oncol. 15, 829–840 (2014).
Robinson, D. et al. Integrative clinical genomics of advanced prostate cancer. Cell 161, 1215–1228 (2015).
Artandi, S. E. & DePinho, R. A. Telomeres and telomerase in cancer. Carcinogenesis 31, 9–18 (2010).
Smith, L. L., Coller, H. A. & Roberts, J. M. Telomerase modulates expression of growth-controlling genes and enhances cell proliferation. Nat. Cell Biol. 5, 474–479 (2003).
Park, J. I. et al. Telomerase modulates Wnt signalling by association with target gene chromatin. Nature 460, 66–72 (2009).
Choi, J. et al. TERT promotes epithelial proliferation through transcriptional control of a Myc- and Wnt-related developmental program. PLoS Genet. 4, e10 (2008).
Jagadeesh, S. & Banerjee, P. P. Telomerase reverse transcriptase regulates the expression of a key cell cycle regulator, cyclin D1. Biochem. Biophys. Res. Commun. 347, 774–780 (2006).
Beck, S. et al. Telomerase activity-independent function of TERT allows glioma cells to attain cancer stem cell characteristics by inducing EGFR expression. Mol. Cells 31, 9–15 (2011).
Liu, Z. et al. Telomerase reverse transcriptase promotes epithelial-mesenchymal transition and stem cell-like traits in cancer cells. Oncogene 32, 4203–4213 (2013).
Imamura, Y. & Sadar, M. D. Androgen receptor targeted therapies in castration-resistant prostate cancer: bench to clinic. Int. J. Urol. 23, 654–65 (2016).
Hu, J. et al. Antitelomerase therapy provokes ALT and mitochondrial adaptive mechanisms in cancer. Cell 148, 651–663 (2012).
Rothkamm, K. et al. DNA damage foci: meaning and significance. Environ. Mol. Mutag. 56, 491–504 (2015).
Cesare, A. J. et al. Spontaneous occurrence of telomeric DNA damage response in the absence of chromosome fusions. Nat. Struct. Mol. Biol. 16, 1244–1251 (2009).
Silvestre, D. C. et al. Alternative lengthening of telomeres in human glioma stem cells. Stem Cells 29, 440–451 (2011).
Flynn, R. L. et al. Alternative lengthening of telomeres renders cancer cells hypersensitive to ATR inhibitors. Science 347, 273–277 (2015).
Kim, S. H. et al. Androgen receptor interacts with telomeric proteins in prostate cancer cells. J. Biol. Chem. 285, 10472–10476 (2010).
Hayashi, M. T., Cesare, A. J., Rivera, T. & Karlseder, J. Cell death during crisis is mediated by mitotic telomere deprotection. Nature 522, 492–496 (2015).
Xu, T., He, K., Wang, L. & Goldkorn, A. Prostate tumor cells with cancer progenitor properties have high telomerase activity and are rapidly killed by telomerase interference. Prostate 71, 1390–1400 (2011).
Muller, S. & Rodriguez, R. G-Quadruplex interacting small molecules and drugs: from bench toward bedside. Expert Rev. Clin. Pharmacol. 7, 663–679 (2014).
Rizzo, A., Salvati, E. & Biroccio, A. Methods of studying telomere damage induced by quadruplex-ligand complexes. Methods 57, 93–99 (2012).
Reddy, V. et al. ATM Inhibition Potentiates Death of Androgen Receptor-inactivated Prostate Cancer Cells with Telomere Dysfunction. J. Biol. Chem. 290, 25522–25533 (2015).
Kim, S. H. et al. Telomere dysfunction and cell survival: roles for distinct TIN2-containing complexes. J. Cell Biol. 181, 447–460 (2008).
Zhou, J. et al. Structural and functional association of androgen receptor with telomeres in prostate cancer cells. Aging (Albany NY) 5, 3–17 (2013).
Fan, X. et al. hTERT gene amplification and increased mRNA expression in central nervous system embryonal tumors. Am. J. Pathol. 162, 1763–1769 (2003).
Zhang, A. et al. Amplification of the telomerase reverse transcriptase (hTERT) gene in cervical carcinomas. Genes Chromosomes Cancer 34, 269–275 (2002).
Zhu, C. Q. et al. Amplification of telomerase (hTERT) gene is a poor prognostic marker in non-small-cell lung cancer. Br. J. Cancer 94, 1452–1459 (2006).
Totoki, Y. et al. Trans-ancestry mutational landscape of hepatocellular carcinoma genomes. Nat. Genet. 46, 1267–1273 (2014).
Peifer, M. et al. Telomerase activation by genomic rearrangements in high-risk neuroblastoma. Nature 526, 700–704 (2015).
Valentijn, L. J. et al. TERT rearrangements are frequent in neuroblastoma and identify aggressive tumors. Nat. Genet. 47, 1411–1414 (2015).
Castelo-Branco, P. et al. Methylation of the TERT promoter and risk stratification of childhood brain tumours: an integrative genomic and molecular study. Lancet Oncol. 14, 534–542 (2013).
Bethel, C. R. et al. Decreased NKX3.1 protein expression in focal prostatic atrophy, prostatic intraepithelial neoplasia, and adenocarcinoma: association with gleason score and chromosome 8p deletion. Cancer Res. 66, 10683–10690 (2006).
Bernhardt, S. L. et al. Telomerase peptide vaccination of patients with non-resectable pancreatic cancer: a dose escalating phase I/II study. Br. J. Cancer 95, 1474–1482 (2006).
Huang, P. et al. Direct and distant antitumor effects of a telomerase-selective oncolytic adenoviral agent, OBP-301, in a mouse prostate cancer model. Cancer Gene Ther. 15, 315–322 (2008).
Drygin, D. et al. Anticancer activity of CX-3543: a direct inhibitor of rRNA biogenesis. Cancer Res. 69, 7653–7661 (2009).
Balasubramanian, S., Hurley, L. H. & Neidle, S. Targeting G-quadruplexes in gene promoters: a novel anticancer strategy? Nat. Rev. Drug Discov. 10, 261–275 (2011).
Hasegawa, D. et al. G-Quadruplex ligand-induced DNA damage response coupled with telomere dysfunction and replication stress in glioma stem cells. Biochem. Biophys. Res. Commun. 471, 75–81 (2016).
Grand, C. L. et al. The cationic porphyrin TMPyP4 down-regulates c-MYC and human telomerase reverse transcriptase expression and inhibits tumor growth in vivo. Mol. Cancer Ther. 1, 565–573 (2002).
Salvati, E. et al. Telomere damage induced by the G-quadruplex ligand RHPS4 has an antitumor effect. J. Clin. Invest. 117, 3236–3247 (2007).
Acknowledgements
The authors wish to thank Dr Christopher Michael Heaphy and Dr Karen Sandell Sfanos for critical reading of the manuscript. The authors' research work was supported by NIH research grants R01CA172380 to A. M., NIH Training in Areas Fundamental to Cancer Research 5T32CA009110-38 to M. K. G., and the Prostate Cancer Foundation Young Investigators Award (granted to C. M. Heaphy and supported M. K. G.).
Author information
Authors and Affiliations
Contributions
Both authors researched data for the article and made substantial contributions to discussion of its content. Both authors wrote and reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- T-Loop
-
A structure stabilized by shelterin proteins at the end of telomeres, where the telomere double-stranded DNA loops onto itself to form a partial overlap between the 3′ G-rich telomere overhang and the complementary C-rich telomere strand upstream of the overhang.
- End replication problem
-
During DNA replication, synthesis on the lagging DNA strand of linear templates is incomplete, resulting in the loss of ∼50 terminal nucleotides in each round of cellular division.
- Replicative senescence
-
In normal cells, cessation of cell division owing to substantial telomere shortening following ∼50 cell divisions (Hayflick limit).
- BPH
-
Noncancerous enlargement of the prostate owing to hyperproliferation of epithelial and/or stromal cells in the prostate.
- Prostatic intraepithelial neoplasia
-
(PIN). A noncancerous lesion in the prostate with abnormal acinar architecture, observed as overcrowding of luminal cells with enlarged nuclei.
- High-grade PIN
-
(HGPIN). Considered a precursor lesion of prostate cancer, featuring cancer-like morphological abnormalities (for example, nuclear pleomorphism and prominent nucleoli), but no evidence of invasion.
- Reactive oxygen species
-
(ROS). Highly reactive, oxygen-containing free radicals that can damage cellular RNA, DNA, and proteins.
- 8-Oxoguanine
-
The best-characterized and highly abundant DNA lesion arising from the oxidation of guanine through reactive oxygen species.
- Base excision repair
-
(BER). The DNA repair pathway that employs specialized DNA glycosylases, N-glycosylase/DNA lyase and adenine DNA glycosylase, to repair 8-oxoguanine.
- G-Quadruplexes
-
Nucleic acid secondary structures arising from Hoogsteen base pairing (an alternative form of base pairing) interactions of guanine residues.
- Fragile sites
-
Unstable regions in the genome that are prone to break under replication stress.
- Prostatic inflammatory atrophy
-
Prostatic lesions characterized by increased proliferation and atrophic morphology of prostatic luminal epithelial cells, associated with local inflammatory cells.
- Chromothripsis
-
Multiple translocation events occurring in a single catastrophic event leading to imperfect rearrangement and repair of one or a few shattered chromosomes.
- Overdiagnosis and overtreatment
-
Diagnosing patients with a disease that will not give rise to symptoms or cause death, often leading to treatment that might have no benefit and might even be harmful to the patient.
- Fluorescence in situ hybridization
-
(FISH). A technique using fluorophore-conjugated oligonucleotide probes that bind to specific DNA sequences via complementary Watson–Crick base pairing, enabling detection of sequences of interest in intact cells or chromosomes by fluorescence microscopy.
- Prostate Cancer Prevention Trial
-
A study conducted from 1994–2003 to investigate if the 5α-reductase inhibitor finasteride reduces prostate cancer development in men ≥55 years of age.
- Peptide vaccine
-
A peptide conjugated with a vaccine adjuvant to stimulate an immune response against a target antigen that shares the same amino acid sequence of the peptide.
- Epithelial–mesenchymal transition
-
The biological process in which epithelial cells acquire characteristics more consistent with mesenchymal cells, including loss of cell polarity and adhesion, and enhanced migration and invasiveness.
- Telomere deprotection
-
Telomeres partially or completely unprotected by shelterin proteins, resulting in the activation of DDR.
Rights and permissions
About this article
Cite this article
Graham, M., Meeker, A. Telomeres and telomerase in prostate cancer development and therapy. Nat Rev Urol 14, 607–619 (2017). https://doi.org/10.1038/nrurol.2017.104
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrurol.2017.104
This article is cited by
-
Prognostic and therapeutic potential of senescent stromal fibroblasts in prostate cancer
Nature Reviews Urology (2023)
-
The role of testosterone in men’s health: is it time for a new approach?
International Urology and Nephrology (2022)
-
TGF-β controls stromal telomere length through epigenetic modifications
3 Biotech (2022)
-
TAR30, a homolog of the canonical plant TTTAGGG telomeric repeat, is enriched in the proximal chromosome regions of peanut (Arachis hypogaea L.)
Chromosome Research (2022)
-
Prognostic value of TERF1 expression in prostate cancer
Journal of the Egyptian National Cancer Institute (2021)