Abstract
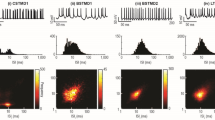
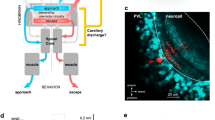
How active membrane conductance dynamics tunes neurons for specific time-varying stimuli remains poorly understood. We studied the biophysical mechanisms by which spike frequency adaptation shapes visual stimulus selectivity in an identified visual interneuron of the locust. The lobula giant movement detector (LGMD) responds preferentially to objects approaching on a collision course with the locust. Using calcium imaging, pharmacology and modeling, we show that spike frequency adaptation in the LGMD is mediated by a Ca2+-dependent potassium conductance closely resembling those associated with 'small-conductance' (SK) channels. Intracellular block of this conductance minimally affected the LGMD's response to approaching stimuli, but substantially increased its response to translating ones. Thus, spike frequency adaptation contributes to the neuron's tuning by selectively decreasing its responses to nonpreferred stimuli. Our results identify a new mechanism by which spike frequency adaptation may tune visual neurons to behaviorally relevant stimuli.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
O'Shea, M. & Williams, J.L.D. The anatomy and output connection of a locust visual interneurone: the lobular giant movement detector (LGMD) neurone. J. Comp. Physiol. 91, 257–266 (1974).
Krapp, H.G. & Gabbiani, F. Spatial distribution of inputs and local receptive field properties of a wide-field, looming sensitive neuron. J. Neurophysiol. 93, 2240–2253 (2005).
Burrows, M. The Neurobiology of an Insect Brain (Oxford University Press, Oxford, UK, 1996).
Schlotterer, G.R. Response of the locust descending movement detector neuron to rapidly approaching and withdrawing visual stimuli. Can. J. Zool. 55, 1372–1376 (1977).
Rind, F.C. & Simmons, P.J. Orthopteran DCMD neuron: a reevaluation of responses to moving objects. I. Selective responses to approaching objects. J. Neurophysiol. 68, 1654–1666 (1992).
Simmons, P.J. & Rind, F.C. Orthopteran DCMD neuron: a reevaluation of responses to moving objects. II. Critical cues for detecting approaching objects. J. Neurophysiol. 68, 1667–1682 (1992).
Judge, S. & Rind, F. The locust DCMD, a movement-detecting neurone tightly tuned to collision trajectories. J. Exp. Biol. 200, 2209–2216 (1997).
Gabbiani, F., Krapp, H.G. & Laurent, G. Computation of object approach by a wide-field, motion-sensitive neuron. J. Neurosci. 19, 1122–1141 (1999).
Matheson, T., Rogers, S.M. & Krapp, H.G. Plasticity in the visual system is correlated with a change in lifestyle of solitarious and gregarious locusts. J. Neurophysiol. 91, 1–12 (2004).
Fotowat, H. & Gabbiani, F. Relationship between the phases of sensory and motor activity during a looming-evoked multistage escape behavior. J. Neurosci. 27, 10047–10059 (2007).
Yamamoto, K., Nakata, M. & Nakagawa, H. Input and output characteristics of collision avoidance behavior in the frog Rana catesbeiana. Brain Behav. Evol. 62, 201–211 (2003).
Sun, H. & Frost, B.J. Computation of different optical variables of looming objects in pigeon nucleus rotundus neurons. Nat. Neurosci. 1, 296–303 (1998).
Preuss, T., Osei-Bonsu, P.E., Weiss, S.A., Wang, C. & Faber, D.S. Neural representation of object approach in a decision-making motor circuit. J. Neurosci. 26, 3454–3464 (2006).
Rowell, C.H.F. & O'Shea, M. The neuronal basis of a sensory analyser, the acridid movement detector system. IV. The preference for small field stimuli. J. Exp. Biol. 68, 157–185 (1977).
Gabbiani, F., Cohen, I. & Laurent, G. Time-dependent activation of feed-forward inhibition in a looming-sensitive neuron. J. Neurophysiol. 94, 2150–2161 (2005).
Gabbiani, F. & Krapp, H.G. Spike-frequency adaptation and intrinsic properties of an identified, looming-sensitive neuron. J. Neurophysiol. 96, 2951–2962 (2006).
Peron, S.P., Krapp, H.G. & Gabbiani, F. Influence of electrotonic structure and synaptic mapping on the receptive field properties of a collision-detecting neuron. J. Neurophysiol. 97, 159–177 (2007).
Santer, R.D., Yamawaki, Y., Rind, F.C. & Simmons, P.J. Preparing for escape: an examination of the role of the DCMD neuron in locust escape jumps. J. Comp. Physiol. A Neuroethol. Sens. Neural Behav. Physiol. 194, 69–77 (2008).
Rowell, C.H. The orthopteran descending movement detector (DMD) neurones: a characterisation and review. Z. Vergl. Physiol. 73, 167–194 (1971).
Tsien, R.Y. New calcium indicators and buffers with high selectivity against magnesium and protons: design, synthesis, and properties of prototype structures. Biochemistry 19, 2396–2404 (1980).
Stocker, M. Ca2+-activated K+ channels: molecular determinants and function of the SK family. Nat. Rev. Neurosci. 5, 758–770 (2004).
Sobel, E.C. & Tank, D.W. In Vivo Ca2+ Dynamics in a Cricket Auditory Neuron: An Example of Chemical Computation. Science 263, 823–826 (1994).
Wang, X.J. Calcium coding and adaptive temporal computation in cortical pyramidal neurons. J. Neurophysiol. 79, 1549–1566 (1998).
Faber, E.S. & Sah, P. Calcium-activated potassium channels: multiple contributions to neuronal function. Neuroscientist 9, 181–194 (2003).
Scott, R.H., Sutton, K.G., Griffin, A., Stapleton, S.R. & Currie, K.P. Aspects of calcium-activated chloride currents: a neuronal perspective. Pharmacol. Ther. 66, 535–565 (1995).
Bond, C.T., Maylie, J. & Adelman, J.P. SK channels in excitability, pacemaking and synaptic integration. Curr. Opin. Neurobiol. 15, 305–311 (2005).
Wicher, D., Walther, C. & Wicher, C. Non-synaptic ion channels in insects–basic properties of currents and their modulation in neurons and skeletal muscles. Prog. Neurobiol. 64, 431–525 (2001).
Heidel, E. & Pflüger, H.J. Ion currents and spiking properties of identified subtypes of locust octopaminergic dorsal unpaired median neurons. Eur. J. Neurosci. 23, 1189–1206 (2006).
Gu, N., Vervaeke, K. & Storm, J.F. BK potassium channels facilitate high-frequency firing and cause early spike frequency adaptation in rat CA1 hippocampal pyramidal cells. J. Physiol. (Lond.) 580, 859–882 (2007).
Killmann, F. & Schürmann, F.W. Both electrical and chemical transmission between the lobula giant movement detector and the descending contralateral movement detector neurons of locusts are supported by electron microscopy. J. Neurocytol. 14, 637–652 (1985).
Neher, E. & Sakaba, T. Multiple roles of calcium ions in the regulation of neurotransmitter release. Neuron 59, 861–872 (2008).
Peracchia, C. Chemical gating of gap junction channels; roles of calcium, pH and calmodulin. Biochim. Biophys. Acta 1662, 61–80 (2004).
Gabbiani, F., Krapp, H.G., Koch, C. & Laurent, G. Multiplicative computation in a visual neuron sensitive to looming. Nature 420, 320–324 (2002).
Rall, W. Theoretical significance of dendritic trees for neuronal input-output relations. in Neural Theory and Modeling (ed. R.F. Reiss) (Stanford University Press, Palo Alto, CA, 1964).
Koch, C., Poggio, T. & Torre, V. Retinal ganglion cells: a functional interpretation of dendritic morphology. Phil. Trans. R. Soc. Lond. B 298, 227–263 (1982).
Bhattacharjee, A. & Kaczmarek, L.K. For K+ channels, Na+ is the new Ca2+. Trends Neurosci. 28, 422–428 (2005).
Wittekindt, O.H. et al. An Apamin and Scyllatoxin-Insensitive Isoform of the Human SK3 Channel. Mol. Pharmacol. 65, 788–801 (2004).
Teagarden, M., Atherton, J.F., Bevan, M.D. & Wilson, C.J. Accumulation of cytoplasmic calcium, but not apamin-sensitive afterhyperpolarization current, during high frequency firing in rat subthalamic nucleus cells. J. Physiol. (Lond.) 586, 817–833 (2008).
Sah, P. Ca2+-activated K+ currents in neurones: types, physiological roles and modulation. Trends Neurosci. 19, 150–154 (1996).
Hirschberg, B., Maylie, J., Adelman, J.P. & Marrion, N.V. Gating of recombinant small-conductance Ca-activated K+ channels by calcium. J. Gen. Physiol. 111, 565–581 (1998).
Haag, J. & Borst, A. Spatial distribution and characteristics of voltage-gated calcium signals within visual interneurons. J. Neurophysiol. 83, 1039–1051 (2000).
Single, S. & Borst, A. Different mechanisms of calcium entry within different dendritic compartments. J. Neurophysiol. 87, 1616–1624 (2002).
Kurtz, R., Dürr, V. & Egelhaaf, M. Dendritic calcium accumulation associated with direction-selective adaptation in visual motion-sensitive neurons in vivo. J. Neurophysiol. 84, 1914–1923 (2000).
Harris, R.A., O'Carroll, D.C. & Laughlin, S.B. Contrast gain reduction in fly motion adaptation. Neuron 28, 595–606 (2000).
Ellis, L.D. et al. SK channels provide a novel mechanism for the control of frequency tuning in electrosensory neurons. J. Neurosci. 27, 9491–9502 (2007).
Sanchez-Vives, M.V., Nowak, L.G. & McCormick, D.A. Cellular mechanisms of long-lasting adaptation in visual cortical neurons in vitro. J. Neurosci. 20, 4286–4299 (2000).
Wang, X.J., Liu, Y., Sanchez-Vives, M.V. & McCormick, D.A. Adaptation and temporal decorrelation by single neurons in the primary visual cortex. J. Neurophysiol. 89, 3279–3293 (2003).
Migliore, M. & Shepherd, G.M. Emerging rules for the distributions of active dendritic conductances. Nat. Rev. Neurosci. 3, 362–370 (2002).
Acknowledgements
We would like to thank K. Josic and H. Krapp for comments. This work was supported by grants from the US National Institute of Mental Health. The use of the QNX 6 OS was made possible by QNX Software Systems' Educational Program.
Author information
Authors and Affiliations
Contributions
S.P. performed the experiments and simulations; S.P. and F.G. wrote the manuscript; F.G. supervised the project.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7, Supplementary Methods and Supplementary Pharmacology (PDF 2130 kb)
Rights and permissions
About this article
Cite this article
Peron, S., Gabbiani, F. Spike frequency adaptation mediates looming stimulus selectivity in a collision-detecting neuron. Nat Neurosci 12, 318–326 (2009). https://doi.org/10.1038/nn.2259
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2259
This article is cited by
-
Ultra-low power neuromorphic obstacle detection using a two-dimensional materials-based subthreshold transistor
npj 2D Materials and Applications (2023)
-
Modeling the grid cell activity based on cognitive space transformation
Cognitive Neurodynamics (2023)
-
Tonotopic Ca2+ dynamics and sound processing in auditory interneurons of the bush-cricket Mecopoda elongata
Journal of Comparative Physiology A (2023)
-
Visual antipredator effects of web flexing in an orb web spider, with special reference to web decorations
The Science of Nature (2023)
-
Approaching object acceleration differentially affects the predictions of neuronal collision avoidance models
Biological Cybernetics (2023)