Abstract
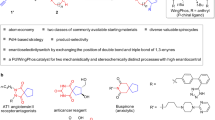
Building small-molecule libraries with structural and stereogenic diversity plays an important role in drug discovery. The development of switchable intermolecular cycloaddition reactions from identical substrates in different regioselective fashions would provide an attractive protocol. However, this also represents a challenge in organic chemistry, because it is difficult to control regioselectivity to afford the products exclusively and at the same time achieve high levels of stereoselectivity. Here, we report the diversified cycloadditions of α′-alkylidene-2-cyclopentenones catalysed by cinchona-derived primary amines. An asymmetric γ,β′-regioselective intermolecular [6+2] cycloaddition reaction with 3-olefinic (7-aza)oxindoles is realized through the in situ generation of formal 4-aminofulvenes, while a different β,γ-regioselective [2+2] cycloaddition reaction with maleimides to access fused cyclobutanes is disclosed. In contrast, an intriguing α,γ-regioselective [4+2] cycloaddition reaction is uncovered with the same set of substrates, by employing an unprecedented dual small-molecule catalysis of amines and thiols. All of the cycloaddition reactions exhibit excellent regio- and stereoselectivity, producing a broad spectrum of chiral architectures with high structural diversity and molecular complexity.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
MacMillan, D. W. C. The advent and development of organocatalysis. Nature 455, 304–308 (2008).
Dondoni, A. & Massi, A. Asymmetric organocatalysis: From infancy to adolescence. Angew. Chem. Int. Ed. 47, 4638–4660 (2008).
Melchiorre, P., Marigo, M., Carlone, A. & Bartoli, G. Asymmetric aminocatalysis—gold rush in organic chemistry. Angew. Chem. Int. Ed. 47, 6138–6171 (2008).
Bertelsen, S. & Jørgensen, K. A . Organocatalysis—after the gold rush. Chem. Soc. Rev. 38, 2178–2189 (2009).
Moyano, A. & Rios, R . Asymmetric organocatalytic cyclization and cycloaddition reactions. Chem. Rev. 111, 4703–4832 (2011).
Donslund, B. S., Johansen, T. K., Poulsen, P. H., Halskov, K. S. & Jørgensen, K. A. The diarylprolinol silyl ethers: ten years after. Angew. Chem. Int. Ed. 54, 13860–13874 (2015).
Harmata, M., Ghosh, S. K., Hong, X., Wacharasindhu, S. & Kirchhoefer, P. Asymmetric organocatalysis of 4+3 cycloaddition reactions. J. Am. Chem. Soc. 125, 2058–2059 (2003).
Burns, N. Z., Witten, M. R. & Jacobsen, E. N. Dual catalysis in enantioselective oxidopyrylium-based [5+2] cycloadditions. J. Am. Chem. Soc. 123, 14578–14581 (2011).
Orue, A., Uria Reyes, U. E., Carrillo, L. & Vicario, J. L. Catalytic enantioselective [5+2] cycloaddition between oxidopyrylium ylides and enals under dienamine activation. Angew. Chem. Int. Ed. 54, 3043–3046 (2015).
Dell'Amico, L . et al. Exploring the vinylogous reactivity of cyclohexenylidene malononitriles: switchable regioselectivity in the organocatalytic asymmetric addition to enals giving highly enantioenriched carbabicyclic structures. J. Am. Chem. Soc. 136, 11107–11114 (2014).
Guo, C., Sahoo, B., Daniliuc, C. G. & Glorius, F. N-heterocyclic carbene catalyzed switchable reactions of enals with azoalkenes: formal [4+3] and [4+1] annulations for the synthesis of 1,2-diazepines and pyrazoles. J. Am. Chem. Soc. 136, 17402–17405 (2014).
Guo, C., Fleige, M., Janssen-Müller, D., Daniliuc, C. G. & Glorius, F . Switchable selectivity in an NHC-catalysed dearomatizing annulation reaction. Nat. Chem. 7, 842–847 (2015).
Zhan, G. et al. Catalyst-controlled switch in chemo- and diastereoselectivities: annulations of Morita–Baylis–Hillman carbonates from isatins. Angew. Chem. Int. Ed. 55, 2147–2151 (2016).
Neuenschwander, M. in The Chemistry of Double-Bonded Functional Groups Supplement A (ed. Patai, S.) Vol. 2 1131–1268 (Wiley, 1989).
Barluenga, J., Martínez, S., Suárez-Sobrino, A. L. & Tomás, M. New reaction pathways for Fischer carbene complexes: [6+3] cycloaddition of chromium alkenyl carbene complexes with fulvenes. J. Am. Chem. Soc. 123, 11113–11114 (2001).
Barluenga, J., Martínez, S., Suárez-Sobrino, A. L. & Tomás, M . The [2+1] and [4+3] cyclization reactions of fulvenes with Fischer carbene complexes: new access to annulated cyclopentanones. J. Am. Chem. Soc. 124, 5948–5949 (2002).
Potowski, M., Antonchick, A. P. & Waldmann, H. Highly enantioselective catalytic [6+3] cycloadditions of azomethine ylides. Angew. Chem. Int. Ed. 51, 9512–9516 (2012).
He, Z.-L., Teng, H.-L. & Wang, C.-J. Fulvenes as effective dipolarophiles in copper(I)-catalyzed [6+3] cycloaddition of azomethine ylides: asymmetric construction of piperidine derivatives. Angew. Chem. Int. Ed. 52, 2934–2938 (2013).
Wu, T.-C. & Houk, K. N . Construction of linearly fused tricyclopentanoids by intramolecular [6+2] cycloadditions of fulvenes with enamines. J. Am. Chem. Soc. 107, 5308–5309 (1985).
Hayashi, Y . et al. Organocatalytic, enantioselective intramolecular [6+2] cycloaddition reaction for the formation of tricyclopentanoids and insight on its mechanism from a computational study. J. Am. Chem. Soc. 133, 20175–20185 (2011).
Hong, B.-C. et al. Novel [6+2] cycloaddition of fulvenes with alkenes: a facile synthesis of the anislactone and hirsutane framework. Org. Lett. 4, 2249–2252 (2002).
Li, J.-L., Liu, T.-Y. & Chen, Y.-C. Aminocatalytic asymmetric Diels–Alder reactions via HOMO activation. Acc. Chem. Res. 45, 1491–1500 (2012).
Kumar, I., Ramaraju, P. & Mir, N. A . Asymmetric trienamine catalysis: new opportunities in amine catalysis. Org. Biomol. Chem. 11, 709–716 (2013).
Arceo, E. & Melchiorre, P. Extending the aminocatalytic HOMO-raising activation strategy: where is the limit? Angew. Chem. Int. Ed. 51, 5290–5292 (2012).
Jiang, H., Albrecht, Ł. & Jørgensen, K. A . Aminocatalytic remote functionalization strategies. Chem. Sci. 4, 2287–2300 (2013).
Jurberg, I. D., Chatterjee, I., Tannert, R. & Melchiorre, P . When asymmetric aminocatalysis meets the vinylogy principle. Chem. Commun. 49, 4869–4883 (2013).
Jiang, K. & Chen, Y.-C. The development of asymmetric trienamine catalysis. Prog. Chem. 27, 137–145 (2015).
Takanami, T., Suda, K. & Ohmori, H . Utilization of PPh3-Ti(IV) reagents. An efficient α′-alkylidenation of cyclic enones. Tetrahedron Lett. 31, 677–680 (1990).
Melchiorre, P. Cinchona-based primary amine catalysis in the asymmetric functionalization of carbonyl compounds. Angew. Chem. Int. Ed. 51, 9748–9770 (2012).
Jiang, L. & Chen, Y.-C. Recent advances in asymmetric catalysis with cinchona alkaloid-based primary amines. Catal. Sci. Technol. 1, 354–365 (2011).
Halskov, K. S . et al. Cross-trienamines in asymmetric organocatalysis. J. Am. Chem. Soc. 134, 12943–12946 (2012).
Dieckmann, A., Breugst, M. & Houk, K. N . Zwitterions and unobserved intermediates in organocatalytic Diels–Alder reactions of linear and cross-conjugated trienamines. J. Am. Chem. Soc. 135, 3237–3242 (2013).
Lee, A., Michrowska, A., Sulzer-Mosse, S. & List, B. The catalytic asymmetric Knoevenagel condensation. Angew. Chem. Int. Ed. 50, 1707–1710 (2011).
Stiller, J., Kowalczyk, D., Jiang, H., Jørgensen, K. A. & Albrecht, Ł. Novel organocatalytic activation of unmodified Morita–Baylis–Hillman alcohols for the synthesis of bicyclic α-alkylidene-ketones. Chem. Eur. J. 20, 13108–13112 (2014).
Kanta De, C. & Seidel, D. Catalytic enantioselective desymmetrization of meso-diamines: a dual small-molecule catalysis approach. J. Am. Chem. Soc. 133, 14538–14541 (2011).
Birrell, J. A., Desrosiers, J.-N. & Jacobsen, E. N. Enantioselective acylation of silyl ketene acetals through fluoride anion-binding catalysis. J. Am. Chem. Soc. 133, 13872–13875 (2011).
Rahaman, H., Madarász, Á., Pápai, I. & Pihko, P. M. Dual hydrogen-bond/enamine catalysis enables a direct enantioselective three-component domino reaction. Angew. Chem. Int. Ed. 50, 6123–6127 (2011).
Aroyan, C. E. & Miller, S. J . Enantioselective Rauhut–Currier reactions promoted by protected cysteine. J. Am. Chem. Soc. 129, 256–257 (2007).
Aroyan, C. E., Dermenci, A. & Miller, S. J . Development of a cysteine-catalyzed enantioselective Rauhut–Currier reaction. J. Org. Chem. 75, 5784–5796 (2010).
Selig, P. S. & Miller, S. J. Ortho-acidic aromatic thiols as efficient catalysts of intramolecular Morita–Baylis–Hillman and Rauhut–Currier reactions. Tetrahedron Lett. 52, 2148–2151 (2011).
Mose, R., Jensen, M. E., Preegel, G. & Jørgensen, K. A. Direct access to multifunctional norcamphor scaffolds by asymmetric organocatalytic Diels–Alder reactions. Angew. Chem. Int. Ed. 54, 13630–13634 (2015).
Albrecht, Ł. et al. Asymmetric organocatalytic formal [2+2]-cycloadditions via bifunctional H-bond directing dienamine catalysis. J. Am. Chem. Soc. 134, 2543–2546 (2012).
Talavera, G., Reyes, E., Vicario, J. L. & Carrillo, L. Cooperative dienamine/hydrogen-bonding catalysis: enantioselective formal [2+2] cycloaddition of enals with nitroalkenes. Angew. Chem. Int. Ed. 51, 4104–4107 (2012).
Xu, Y., Conner, M. L. & Brown, M. K. Cyclobutane and cyclobutene synthesis: catalytic enantioselective [2+2] cycloadditions. Angew. Chem. Int. Ed. 54, 11918–11928 (2015).
Acknowledgements
The authors acknowledge financial support from the Natural Science Foundation of China (grant numbers 21125206, 21372160 and 21321061).
Author information
Authors and Affiliations
Contributions
Z.Z. and Y.C.C conceived and designed the research. Z.Z., Z.X.W., Y.C.Z. and W.X. performed the research. Q.O. performed the DFT computational calculation study. Y.C.C., Q.O., W.D. and Z.Z. co-wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 14020 kb)
Supplementary information
Crystallographic data for compound 3b. (CIF 326 kb)
Supplementary information
Crystallographic data for compound 4b. (CIF 643 kb)
Supplementary information
Crystallographic data for compound 9b. (CIF 379 kb)
Rights and permissions
About this article
Cite this article
Zhou, Z., Wang, ZX., Zhou, YC. et al. Switchable regioselectivity in amine-catalysed asymmetric cycloadditions. Nature Chem 9, 590–594 (2017). https://doi.org/10.1038/nchem.2698
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.2698
This article is cited by
-
Construction of a synthetic methodology-based library and its application in identifying a GIT/PIX protein–protein interaction inhibitor
Nature Communications (2022)
-
Catalytic chemodivergent annulations between α-diketones and alkynyl α-diketones
Science China Chemistry (2021)
-
N-Heterocyclic carbene-catalyzed enantioselective hetero-[10 + 2] annulation
Communications Chemistry (2020)