Abstract
Mesenchymal stem cells (MSCs) are ‘adult’ multipotent cells that promote regeneration of injured tissues in vivo. However, differences in oxygenation levels between normoxic culture conditions (21% oxygen) and both the MSC niche (2–8% oxygen) and ischemic injury-induced oxidative stress conditions in vivo have resulted in low efficacy of MSC therapies in both pre-clinical and clinical studies. To address this issue, we examined the effectiveness of hypoxia preconditioning (2% oxygen) for enhancing the bioactivity and tissue-regenerative potential of adipose-derived MSCs. Hypoxia preconditioning enhanced the proliferative potential of MSCs by promoting the expression of normal cellular prion protein (PrPC). In particular, hypoxia preconditioning-mediated MSC proliferation was regulated by PrPC-dependent JAK2 and STAT3 activation. In addition, hypoxia preconditioning-induced PrPC regulated superoxide dismutase and catalase activity, and inhibited oxidative stress-induced apoptosis via inactivation of cleaved caspase-3. In a murine hindlimb ischemia model, hypoxia preconditioning enhanced the survival and proliferation of transplanted MSCs, ultimately resulting in improved functional recovery of the ischemic tissue, including the ratio of blood flow perfusion, limb salvage, and neovascularization. These results suggest that Hypo-MSC offer a therapeutic strategy for accelerated neovasculogenesis in ischemic diseases, and that PrPC comprises a potential target for MSC-based therapies.
Similar content being viewed by others
Main
Human mesenchymal stem cells (MSCs) have numerous potential applications for regenerative medicine-based therapies for diseases such as cardiovascular disease, ischemic stroke, peripheral artery disease, spinal cord injury, liver disease, and anemia. However, various pathophysiological conditions, including ischemia, inflammation, and low nutrient levels, have inhibited the efficacy of MSC-based therapies.1 In particular, ischemia-mediated oxidative stress leads to the production of reactive oxygen species (ROS), resulting in DNA damage, cell apoptosis and death, and end-organ tissue damage.2 To address the low efficacy of transplanted MSCs, several studies have performed genetic modification of MSCs; however, the clinical application of this approach is limited owing to unexpected adverse effects, such as toxicity, immunogenicity, and oncogenicity.3, 4 Therefore, it is pertinent to search for novel approaches to promote transplanted cell survival to foster the success of stem cell-based therapies for ischemic diseases.
Ischemic tissues induce severe hypoxic conditions (O2 concentrations <0.1%),5 which often lead to low survival, inferior differentiation, and death of grafted cells.6, 7 Notably, however, previous studies demonstrated that hypoxia-induced apoptosis can be prevented in certain cell types by preconditioning the cells, via exposure to less severe hypoxic conditions (1–3% O2), for a period of time prior to exposing them to the severe ischemia at the injury site.8, 9, 10 Cultivation under hypoxic conditions (1–3% O2) may also be beneficial for MSCs, as this oxygen tension is similar to that present at the physiologic niche for MSCs in the bone marrow (2–7% O2). Although hypoxia preconditioning induced the upregulation of hypoxia-inducible factor-1 alpha (HIF-1α), CXCR4, c-Met, and/or TWIST, it is unclear how this treatment affects self-renewal, differentiation, and survival of various stem/progenitor cells in ischemic injured tissues.
The normal cellular isoform of prion protein (PrPC) is a highly conserved glycoprotein that is monomeric in structure, protease-sensitive, and tethered to the cell membrane by a glycosyl phosphatidyl inositol anchor.11 Meanwhile, the abnormal form of PrP is commonly multimeric, aggregated, and protease-resistant, and is associated with neurotoxicity and neurodegeneration.12 Although several studies investigated the molecular mechanisms underlying the role of the abnormal form of PrP in neuronal diseases, understanding the physiological role of PrPC remains a challenge due to its numerous functions13, 14 and its roles in cellular processes such as proliferation, self-renewal, and differentiation.15, 16, 17 In addition, a recent study indicated that cellular prion protein enhances neuronal survival and angioneurogenesis after post-ischemic injury.18 However, it is unknown whether MSCs express PrPC or whether PrPC affects MSCs bioactivity, including survival and proliferation, under ischemic conditions.
In this study, we investigated whether cultivation under hypoxic conditions could be utilized to enhance the efficacy of MSCs for ischemic therapy relative to normoxic culturing. We first attempted to establish a general protocol for achieving increased MSC proliferation under hypoxic culture conditions by determining the effects of hypoxic preconditioning on MSC proliferative potential. We subsequently evaluated the ability of preconditioned cells to promote therapeutic vasculogenesis in ischemic limbs. Moreover, we investigated the mechanism by which PrPC regulation affects the proliferation and survival of MSCs subjected to hypoxic preconditioning.
Results
Characterization of MSC differentiation and assessment of MSC proliferation under hypoxic culture conditions
There was no significant difference in MSC differentiation when cultivated under normoxic and hypoxic (2% O2) conditions, as determined by oil red O (adipogenesis), alkaline phosphate (osteogenesis), and safranin O (chondrogenesis) staining analyses, and fabp4 gene (adipogenesis), osteopontin gene (osteogenesis), and sox9 gene (chondrogenesis) expression did not change significantly (Figure 1a and b). To investigate their proliferative potential under hypoxic conditions, MSCs were evaluated using a single-cell expansion assay (Figure 1c). Notably, hypoxia-preconditioned MSCs (Hypo-MSC) exhibited significantly greater levels of expansion than MSCs cultivated under normoxic conditions (Nor-MSCs; Figure 1d and e). These data suggested that hypoxia preconditioning enhances MSC proliferation.
Effects of hypoxia preconditioning on the proliferation of mesenchymal stem cells (MSCs). (a) MSCs were differentiated into adipocytes, osteocytes, and chondrocytes under normoxic or hypoxic conditions in vitro. Adipogenic, osteogenic, and chondrogenic differentiation was examined by oil red O, alkaline phosphatase, and safranin O staining, respectively. Scale bar=200 μm. (b) Adipogenic, osteogenic, and chondrogenic differentiation was examined by fabp2, osteopontin, and sox9 gene expression using quantitative real-time PCR. (c) Schematic of single-cell assays using MSCs. Scale bar=200 μm. (d) Normoxic MSCs (Nor-MSC) and hypoxia-preconditioned MSCs (Hypo-MSC) were grown in single-cell cultures for 0, 5, and 10 days, stained with Giemsa (Scale bar=100 μm), and (e) the percentage of single MSCs undergoing at least one cell division after 1, 5, and 10 days of culture. Values represent means±S.E.M. **P<0.01 versus Nor-MSC
Hypoxia preconditioning induces HIF-1α-mediated PrPC expression
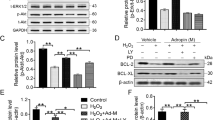
To explore whether hypoxia preconditioning induces PrPC expression in MSCs, we evaluated the expression levels of HIF-1α and PrPC in hypoxia-preconditioned cells by western blot analysis. Hypoxia preconditioning resulted in a time-dependent increase in HIF-1α expression (Figure 2a). Meanwhile, maximal levels of PrPC expression were observed after hypoxic stimulation for 12 h (Figure 2a). Notably, however, this hypoxia-induced expression of PrPC was attenuated by treatment of MSCs with HIF-1α-specific siRNAs (Figure 2b). These findings suggest that hypoxia preconditioning promotes the expression of PrPC via hypoxia-mediated increases in HIF-1α expression.
Hypoxia preconditioning increases cell proliferation through HIF-1α-mediated expression of cellular prion protein (PrPC). (a) Western blot analysis of HIF-1α and PrPC expression in mesenchymal stem cells (MSCs) subjected to hypoxia preconditioning for 0, 6, 12, and 24 h. (b) Western blot analysis of HIF-1α and PrPC expression in MSCs subjected to hypoxia preconditioning for 12 h and pretreated with HIF-1α-specific siRNA (si-HIF-1α). (c) Western blot analysis of PrPC expression in MSCs pretreated with PrPC-specific siRNA (si-PRNP). (d) Western blot analysis of JAK2 and STAT3 phosphorylation, and cyclin D1 and c-Myc expression in MSCs and MSCs treated with PrPC-specific siRNA after hypoxia preconditioning for 0, 6, 12, and 24 h. (e) Single-cell cultures of normoxic MSCs (Nor-MSC), hypoxia-preconditioned MSCs (Hypo-MSC), hypoxia-preconditioned MSCs transfected with PrPC-specific siRNA (si-PRNP Hypo-MSC), and hypoxia-preconditioned MSCs transfected with scrambled siRNA (Scr siRNA Hypo-MSC) were stained with Giemsa after 10 days of cultivation. Scale bar=100 μm. (f) Percentage of single MSCs undergoing at least one cell division after 10 days of culture. Values represent means±S.E.M. **P<0.01 versus Nor-MSC; ##P<0.01 versus Hypo-MSC; $$P<0.01 versus si-PRNP Hypo-MSC
PrPC regulates hMSC proliferation via the JAK2/STAT3 pathway
A previous study demonstrated that hypoxia preconditioning enhances cell proliferation through the JAK2/STAT3 signaling pathway.19 To confirm whether hypoxic conditions promote MSC proliferation through the JAK2/STAT3 pathway, hypoxia-induced phosphorylation of JAK2 and STAT3 were investigated by western blot analysis. Although hypoxia preconditioning for 12 h resulted in increased JAK2 and STAT3 phosphorylation, these effects were attenuated by treatment of MSCs with PrPC-specific siRNA molecules (Figure 2c and d). Likewise, we observed increased expression of cyclin D1 and c-Myc, which are encoded by STAT3 downstream genes, after hypoxic stimulation for 12 h, and decreased expression of these proteins upon siRNA-mediated knockdown of PrPC expression (Figure 2d).
To explore the role of enhanced PrPC expression on the proliferative potential of MSCs under hypoxic conditions, single-cell expansion assays were performed (Figure 2e). Notably, the increase in proliferation observed in Hypo-MSC, compared with that of MSCs cultivated under normoxic conditions, was inhibited by siRNA-mediated PrPC knockdown (Figure 2f). Together, these results suggest that hypoxia preconditioning enhances MSC proliferation potential through PrPC-mediated activation of JAK2/STAT3 pathway.
Hypoxia preconditioning-mediated increases in PrPC expression provide a protective effect against oxidative stress-induced apoptosis in MSCs
To examine the effects of increased PrPC expression on ROS-scavenging, the superoxide dismutase (SOD) and catalase activity of Hyp-MSCs were evaluated. Although hypoxia preconditioning had no significant effect on SOD activity, downregulation of PrPC resulted in significantly decreased SOD activity in Hypo-MSCs compared with normoxic control group (Figure 3a). Conversely, MSCs subjected to hypoxic preconditioning exhibited significantly increased catalase activity, compared with that of MSCs cultivated under normoxic conditions, and this enhancement was blocked by treatment with the PrPC-specific siRNA (Figure 3b). To investigate whether PrPC provides a protective effect against oxidative stress in MSCs, we assessed the levels of apoptosis in MSCs subjected to oxidative stress conditions, including long-term hypoxia (96 h) or treatment with H2O2 by western blot analysis and flow cytometry for detection of PI and Annexin V staining. In MSCs exposed to long-term hypoxia, siRNA-mediated downregulation of PrPC resulted in increased levels of phosphorylated-nuclear factor kappa B (p-NF-κB) and caspase-3 cleavage (Figure 3c and d), and increased numbers of apoptotic cells (Figure 3e and f). Similar results were observed in MSCs subjected to H2O2-induced oxidative stress conditions (Figure 3g and h). These data suggest that PrPC protects MSCs against induction of oxidative stress-induced apoptosis through ROS-scavenging activity.
Knockdown of cellular prion protein (PrPC) expression results in reduced ROS-scavenging activity and increased oxidative stress-induced apoptosis. (a and b) After hypoxia preconditioning for 12 h, we evaluated the superoxide dismutase (SOD) and catalase activities of normoxic MSCs (Nor-MSC), hypoxia-preconditioned MSCs (Hypo-MSC), hypoxia-preconditioned MSCs transfected with PrPC-specific siRNA (si-PRNP Hypo-MSC), and hypoxia-preconditioned MSCs transfected with scrambled siRNA (Scr siRNA Hypo-MSC). Values represent means±S.E.M. **P<0.01 versus Nor-MSC; ##P<0.01 versus Hypo-MSC; $$P<0.01 versus si-PRNP Hypo-MSC. (c) Western blot analysis of the levels of p-NF-κB and cleaved caspase-3 in MSCs and MSCs pretreated with PrPC-specific siRNA or scrambled siRNA after exposure to hypoxic conditions for 96 h. (d) Western blot analysis of the levels of p-NF-κB and cleaved caspase-3 in MSCs and MSCs pretreated with PrPC-specific siRNA or scrambled siRNA after exposure to H2O2 (10−3 M) for 3 h. (e) After exposure to hypoxia for 96 h, MSCs were treated with propidium iodide (PI) and an Annexin V stain, and the number of apoptotic cells was assessed by flow cytometry analysis. (f) Analysis of FACS experiments showing a significant apoptosis rate. **P<0.01 versus control; #P<0.05 versus hypoxia (96 h); $P<0.05 versus si-PRNP hypoxia (96 h). (g) After exposed to H2O2 (10−3 M) for 3 h, MSCs were treated with propidium iodide (PI) and an Annexin V stain, and the number of apoptotic cells was assessed by flow cytometry analysis. (h) Analysis of FACS experiments showing a significant apoptosis rate. **P<0.01 versus control; #P<0.05 versus H2O2 (10−3 M); $P<0.05 versus si-PRNP H2O2 (10−3 M)
Hypo-MSC yield improved functional recovery in a murine hindlimb ischemia model
To determine whether MSCs subjected to hypoxia preconditioning exhibit enhanced vascular repair in vivo, blood perfusion and tissue repair were investigated following transplantation of PBS, Nor-MSCs, Hypo-MSCs, sh-PRNP Hypo-MSCs, and Scr shRNA Hypo-MSC into ischemic thigh muscles of murine hindlimbs. We used PRNP shRNA for stable PrPC downregulation in MSCs. PrPC expression was confirmed by western blotting (Figure 4a).
Assessment of functional recovery in a murine hindlimb ischemia model. (a) Western blot analysis of PrPC expression in MSCs pretreated with PrPC-specific shRNA (sh-PRNP). (b) Blood flow recovery was evaluated via laser Doppler imaging analysis of the ischemic sites of mice injected with PBS, normoxic mesenchymal stem cells (Nor-MSC), hypoxia-preconditioned MSCs (Hypo-MSC), hypoxia-preconditioned MSCs transfected with PrPC-specific shRNA (sh-PRNP Hypo-MSCs), and hypoxia-preconditioned MSCs transfected with scrambled shRNA (Scr shRNA Hypo-MSCs) at 0, 5, 10, 15, 20, and 25 days post surgery. (c) The ratio of blood perfusion (blood flow in the left ischemic limb/blood flow in the right non-ischemic limb) was measured in each of the five treatment groups. Values represent means±S.E.M. **P<0.01 versus PBS; ##P<0.01 versus Nor-MSCs and sh-PRNP Hypo-MSCs. (d) Representative images illustrating the different outcomes (foot necrosis, toe loss, and limb salvage) observed for ischemic limbs in the five treatment groups at postoperative day 25. Distribution of the different outcomes in each group. (e) Necrosis of muscle tissues in the ischemic hindlimb was confirmed by hematoxylin and eosin staining. Scale bar=100 μm
Blood perfusion was assessed by LDPI at postoperative days 0, 5, 10, 15, 20, and 25 (Figure 4b). Notably, mice receiving Hypo-MSCs exhibited significantly increased blood perfusion ratios than those in the PBS, Nor-MSC, and sh-PRNP Hypo-MSC groups (Figure 4c). Meanwhile, transplantation of Hypo-MSCs also resulted in significantly reduced levels of toe loss and foot necrosis compared with that observed in the other mouse groups (Figure 4d and e). These data indicate that transplantation of Hypo-MSC yield enhanced functional recovery in the murine hindlimb ischemia model through improvement of blood flow.
Hypoxia preconditioning enhances MSC survival, proliferation, and neovascularization in ischemic tissues
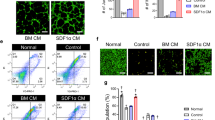
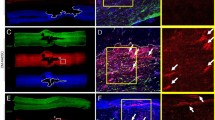
To explore whether hypoxia preconditioning augments the survival and proliferation of transplanted MSCs, ischemic tissues were harvested at postoperative day 3 and subjected to western blot analysis for p-NF-κB and cleaved caspase-3, and immunofluorescence staining for HNA, cleaved caspase-3, and Ki67 (Figure 5). The phosphorylation of NF-κB and expression of cleaved caspase-3 was significantly decreased in tissue lysates injected with hypo-MSCs compared within other groups (Figure 5a and b). The levels of apoptosis among the MSCs in the tissues of each group were then assessed by quantifying the number of HNA and cleaved caspase-3 double-positive cells (Figure 5c and d), whereas the levels of MSC proliferation were evaluated by quantifying the number of HNA and Ki67 double-positive cells (Figure 5e and f). There were significantly fewer apoptotic MSCs within ischemic sites transplanted with Hypo-MSCs than in those transplanted with Nor-MSCs or sh-PRNP Hypo-MSCs (Figure 5d). Meanwhile, ischemic tissues transplanted with Hypo-MSCs contained greater numbers of proliferating MSCs than those transplanted with Nor-MSCs or sh-PRNP Hypo-MSCs (Figure 5f). To confirm angiogenic cytokine expression after the transplantation of MSCs, we assessed the expression of human epidermal growth factor (hEGF), human vascular endothelial growth factor (hVEGF), human fibroblast growth factor (hFGF), and human hepatocyte growth factor (hHGF) at postoperative day 3. The expression levels of hEGF, hVEGF, hFGF, and hHGF were significantly higher in ischemic tissue injected with Hypo-MSCs than in tissues from other groups (Figure 6a). To evaluate the effects of Hypo-MSC on neovascularization, we quantified the number of CD31-positive capillaries and α-SMA-positive arterioles within the ischemic tissues of mice in each group at postoperative day 28 by immunofluorescence staining. There was a significantly greater density of capillaries and arterioles within the ischemic tissues of mice transplanted with Hypo-MSCs than in those transplanted with Nor-MSCs or sh-PRNP Hypo-MSCs (Figure 6b–e). These findings therefore imply that hypoxia preconditioning promotes the survival and proliferation of transplanted MSCs in ischemic tissues in vivo, thereby yielding increased transplanted MSC-mediated neovascularization.
Hypoxia preconditioning enhances the survival and proliferation of transplanted mesenchymal stem cells (MSCs). (a and b) Western blot analysis of p-NF-κB and cleaved caspase-3 expression in ischemic sites of mice injected with PBS, normoxic mesenchymal stem cells (Nor-MSC), hypoxia-preconditioned MSCs (Hypo-MSCs), hypoxia-preconditioned MSCs transfected with PrPC-specific shRNA (sh-PRNP Hypo-MSCs), and hypoxia-preconditioned MSCs transfected with scrambled shRNA (Scr shRNA Hypo-MSCs) at day 3. (c) The levels of MSC apoptosis were evaluated at 3 days after transplantation by immunostaining of ischemic limb tissues for human nuclear antigen (HNA) and cleaved caspase-3 expression. Scale bar=50 μm. (d) The levels of apoptosis of transplanted MSCs were quantified as the HNA and cleaved caspase-3 double-positive cells/HNA-positive cells. Values represent the mean±S.E.M. **P<0.01 versus PBS; ##P<0.01 versus Nor-MSC; $$P<0.01 versus sh-PRNP Hypo-MSCs. (e) MSC proliferation was evaluated by immunostaining for HNA and Ki67 expression in ischemic limb tissues at 3 days after hMSC transplantation. Scale bar=50 μm. (f) Proliferation of transplanted MSCs was quantified as the number of HNA and Ki67 double-positive cells. Values represent the mean±S.E.M. **P<0.01 versus PBS; ##P<0.01 versus Nor-MSC; $$P<0.01 versus sh-PRNP Hypo-MSCs
Hypoxia-preconditioned mesenchymal stem cells (Hypo-hMSCs) promote enhanced neovascularization in a murine hindlimb ischemia model. (a) The expression of hEGF, hVEGF, hFGF, and hHGF in ischemic limb tissue lysates was determined by ELISA. Values represent the mean±S.E.M. **P<0.01 versus PBS; ##P<0.01 versus Nor-MSCs; $P<0.05 versus sh-PRNP Hypo-MSCs $$P<0.01 versus sh-PRNP Hypo-MSCs. (b) Capillary density was evaluated in ischemic tissues at 25 days post MSC transplantation by immunostaining for CD31. Scale bar=50 μm. (c) Capillary density was quantified as the number of human CD31-positive cells. Values represent the mean±S.E.M. **P<0.01 versus PBS; ##P<0.01 versus Nor-MSC; $$P<0.01 versus sh-PRNP Hypo-MSC. (d) Arteriole density in ischemic tissues was evaluated at 25 days post MSC transplantation by immunostaining with an α-SMA antibody. Scale bar=50 μm. (e) Arteriole density was quantified as the number of α-SMA-positive cells. Values represent the mean±S.E.M. **P<0.01 versus PBS; ##P<0.01 versus Nor-MSC; $$P<0.01 versus sh-PRNP Hypo-MSCs
Discussion
MSCs have important roles in the maintenance and repair of bone and blood tissues. In principle, MSCs can be readily isolated from patient tissues, including bone marrow and adipose tissues, and then expanded in culture until the large numbers required for clinical application are obtained. However, the application of MSCs for stem cell-based therapies is hindered by several limitations, including low proliferative potential, restricted life span, and gradual loss of ‘stemness’ during ex vivo expansion.20, 21, 22 In addition, exposure to stressful conditions such as oxidative stress, ischemia, and inflammation can induce apoptosis and cell death, which has resulted in low efficacies of MSCs transplanted into injured tissues.20, 23, 24, 25 Notably, although the tissues from which MSCs are isolated are associated with variable oxygen levels (1–7 % in bone marrow, 10–15% in adipose tissue, and 2–8% at the MSC niche),26, 27 MSCs are typically cultured under normoxic conditions, resulting in the induction of oxidative stress and the alteration of DNA, biomolecule structures, and metabolism.28 To address this issue, several studies have demonstrated that hypoxia preconditioning of MSCs results in reduced apoptosis, chromosomal abnormality, and senescence, and enhanced paracrine activity and regenerative potential.20, 28, 29
Although there is increasing evidence highlighting the beneficial effects of hypoxia preconditioning on MSCs, the precise mechanism through which preconditioning affects these cells remains unclear. Likewise, although there is a consensus that PrPC enhances stem cell proliferation in vitro and in vivo,30 the molecular mechanism through which PrPC influences the proliferative potential of MSCs remains unclear. Here, we provide the first evidence that hypoxia treatment results in enhanced expression of PrPC, which in turn promotes MSC expansion via activation of the JAK2/STAT3 pathway. Previous reports revealed that STAT3 contributes to hypoxia-induced enhancement of self-renewal in embryonic and other adult stem cells.31, 32, 33 Furthermore, our previous study indicated that hypoxia increases the proliferation of endothelial colony-forming cells through the activation of JAK2/STAT3 pathway.19 These findings suggest that hypoxia-induced PrPC enhances MSC proliferation via JAK2/STAT3 pathway activation, and that PrPC is key regulator of JAK2/STAT3 signaling in MSCs.
Importantly, we also demonstrated that maintenance of PrPC expression protected MSCs against oxidative stress-induced apoptosis by promoting increased SOD and catalase activity. Our data are therefore in agreement with previous studies showing that PrPC confers enhanced resistance to oxidative stress by contributing to cellular SOD and catalase activity, whereas attenuated PrPC expression results in enhanced sensitivity to oxidative stress.34, 35, 36 Interestingly, PrPC has a pro-apoptotic role during ER stress, but an anti-apoptotic role during oxidative stress-induced cell death.37 Our data indicate that hypoxia preconditioning-mediated PrPC expression prevents oxidative stress-induced apoptosis and cell death through the regulation of SOD and catalase activity.
Although MSCs differentiate into smooth muscle, endothelial cells, or myocardial muscle in vivo, the predominant mechanism of MSC-mediated tissue repair occurs through angiogenic factor secretion. Accumulating evidence suggests that hypoxia enhances the secretion of VEGF, IGF-1, SDF-1, and EPO, indicating that hypoxic preconditioning improves the tissue repair potential of MSCs.38, 39, 40 However, successful cell engraftment is determined by several factors, including the rates of cell apoptosis and death, the extent of mechanical loss, and the rates at which the cells escape from the region of transplantation, influence the rates of cell engraftment and proliferation in ischemic sites.1 Our current study shows that hypoxia preconditioning yields enhanced functional recovery in a murine hindlimb ischemia model via augmentation of survival, proliferation, and neovascularization of transplanted MSCs. Moreover, the beneficial effects of hypoxia preconditioning on tissue repair and vascularization appears to be mediated by regulation of PrPC expression. Consistent with these findings, overexpression of PrPC was shown to provide a protective effect against focal cerebral ischemia via the activation of ERK, whereas downregulation of PrPC increased the exacerbation of ischemic brain injury through post-ischemic activation of caspase-3.41, 42 Moreover, PrPC was found to promote neuroprotection, neurogenesis, and angiogenesis in ischemic brain injuries.18
Conclusion
In this study, we investigated the efficacy of ex vivo hypoxic preconditioning for enhancing the proliferative potential of MSCs, as well as their resistance to ischemic stress-induced apoptosis. The optimized culture conditions examined here promoted robust cell survival, proliferation, and protection against oxidative stress, resulting in improved therapeutic efficacy of MSCs in a murine hindlimb ischemia model, suggesting that hypoxic preconditioning could comprise a new therapeutic strategy for overcoming ischemic diseases. In addition, our results indicate that PrPC might represent a novel target for MSC-based therapies for several ischemic diseases.
Materials and Methods
Cell cultures
Human adipose-derived MSCs were obtained from the American Type Culture Collection (ATCC; Manassas, VA, USA) and were pathogen- (HBV, HCV, HIV, and Syphilis) and mycoplasma-negative. The supplier certified that the MSCs exhibited expression of the cell surface markers CD73 and CD105, but not of CD31, as well as adipogenic and osteogenic differentiation potential when cultured in specific differentiation media. Cells were cultured in alpha-minimum essential medium (Thermo-Fisher Scientific, Waltham, MA, USA) supplemented with 10% (v/v) fetal bovine serum (Thermo-Fisher Scientific), 100 U/ml of penicillin, and 100 mg/ml streptomycin. Cells were growing a humidified incubator at 37 °C with 5% CO2.
MSC differentiation
For MSC differentiation, cells were grown in StemPro adipogenic, osteogenic, or chondrogenic culture medium (Thermo-Fisher Scientific). To detect adipocytes, cells were cultivated in adipogenic medium for 21 days and stained with oil red O (Sigma-Aldrich, St. Louis, MO, USA) for 15 min. To detect osteoblasts, cells were grown in osteogenic medium for 21 days and subjected to alkaline phosphatase staining with an alkaline phosphatase kit (Sigma-Aldrich). Chondrocytes were detected by staining cells grown in chondrogenic medium for 14 days with safranin O (Sigma-Aldrich).
Quantitative real-time PCR
qPCR analysis was performed using the Rotor-Gene 6000 real-time thermal cycling system (Corbett Research, Mortlake, NSW, Australia) with a QuantiMix SYBR Kit (Phile Korea Technology, Daejeon, Korea). PCR was performed under the following cycling conditions: 95 °C for 15 s, annealing at 60 °C for 30 s, and extension at 72 °C for 60 s for 40 cycles. The data were analyzed by the comparative threshold cycle (CT) method and normalized against Gapdh controls. Primer sequences were as follows: Gapdh forward, 5′-AGTATGACTCCACTCACGGCAA-3′; Gapdh reverse 5′-TCTCGCTCCTGGAAGATGGT-3′; fabp4 forward, 5′-TCGACTTTCCATCCCACTTC-3′; fabp4 reverse, 5′-AGTGACCTCTGTTCGAAGGT-3′; Osteopontin forward, 5′-ATGAGAGCCCTCAGACTCCTC-3′; Osteopontin reverse, 5′-CGCGCCGTAGAAGCGCCGATA-3′, Sox9 forward, 5′-CTGAACGAGAGCGAGAAG-3′; Sox9 reverse, 5′-TTCTTCACCGACTTCCTCC-3′.
Single-cell cultivation assay
A limited-dilution assay was used to aliquot single MSCs into individual wells of 96-well culture plates. In brief, cell suspensions containing 1 × 103 cells in 10 ml complete medium were diluted 1:10 (cells: complete medium), and 100 μl of the diluted sample (approximately 1 cell/100 μl) was seeded into 96-well plates. Cells were then cultured under normoxic or hypoxic conditions in a humidified incubator, and each well was examined for MSC grow that day 10.
Hypoxia preconditioning
MSCs were incubated in a modular incubator chamber (IB Science, Daejeon, Korea) maintaining a hypoxic gas mixture composed of a 2% O2, 5% CO2, and balanced N2 for 0, 6, 12, and 24 h at 37 °C.
siRNA transfection
hMSCs were grown to 70% confluence in culture dishes and were transfected for 48 h with SMART pool siRNAs (100 nM/l) specific to HIF-1α or PrPC mRNA using Lipofectamine 2000 reagent (Thermo-Fisher Scientific), according to the manufacturer’s protocols.
Western blot analysis
Cells and tissue homogenates (20 μg protein) were separated via 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred to nitrocellulose membranes for antibody probing. After washing with TBST (10 mM Tris–HCl (pH 7.6), 150 mM NaCl, 0.05% Tween-20), membranes were blocked with 5% skim milk for 2 h and then incubated with primary antibodies specific to HIF-1α, PrPC, janus kinase 2 (JAK2), phosphorylated-JAK2, signal transducer and activator of transcription 3 (STAT3), p-STAT3, cyclin D1, c-Myc, p-NF-κB, cleaved caspase-3, and β-actin (Santa Cruz Biotechnology, Dallas, TX, USA). After incubation of the membranes with peroxidase-conjugated secondary antibodies (Santa Cruz Biotechnology), bands were detected using enhanced chemiluminescence reagents (Amersham Biosciences, Little Chalfont, UK) in a dark room.
SOD activity assay
The protein concentration of cells lysate was determined using bicinchoninic acid assay (Sigma). The SOD activity assay is based on water soluble tetrazolium reduction by superoxide anion (produced from xanthine by xanthine oxidase) to a colored water soluble tetrazolium formazan product and was performed the total SOD activity measurement at 480 nm.
Catalase activity assay
Catalase activity was based on the rate of degradation of H2O2.The cells were removed from the culture dish by scraping with a policeman, and cells lysate protein extracts (40 μg) reaction sample contained the indicated quantities of catalase and 20 mM H2O2 in 0.1 M Tris–HCl, and incubated for 30 min, and 50 mM Amplex red reagent and 0.2 U/ml of horseradish peroxidase was added and incubated for 30 min at 37 °C. The decomposition of the substrate was recorded by the decrease in absorbance at 563 nm.
PI/Annexin V flow cytometry analysis
To examine the levels of cell apoptosis, cells were stained with Annexin V-FITC and propidium iodide (PI) (De Novo Software, Los Angeles, CA, USA) and evaluated using a Cyflow Cube 8 kit (Partec, Münster, Germany). Data were analyzed using standard FSC Express software (De Novo Software).
PrPC knockdown by shRNA
The lentiviral PRNP shRNA constructs were obtained from OriGene Technologies (Rockville, MD, USA). At 24 h after cell seeding, 293T cells (1.5 × 106) were transfected with a lentiviral vector encoding PRNP shRNA (3 μg) and packaging vector pGFP-C-shLenti (0.3 μg; OriGene Technologies) using Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA). Forty-eight hours after transfection, the supernatant harboring lentiviruses was collected and filtered using a sterilized 0.45-μm syringe filter. The PRNP shRNA lentiviral supernatant with polybrene (5 μg/ml) was added to the hMSCs for infection. After selection in growth medium containing puromycin (2 μg/ml) for 2 weeks, individual resistant cells were expanded in growth media without puromycin, and protein expression was evaluated via western blot analysis. PRNP shRNA No. 3 showed the most effective PrPC knockdown among the four PRNP shRNA clones (No.1–4). The corresponding knockdown clone was used for further studies. The PRNP shRNA No. 3 sequence was 5′-AACCAACATGAAGCACATGGCTGGTGCTG-3′.
Cell transplantation in a murine hindlimb ischemia model
Cell transplantation experiments were performed on 8-week-old nude male BALB/c mice (Biogenomics, Seoul, Korea) maintained under a 12-h light/dark cycle, and in accordance with the regulations of Soonchunhyang University, Seoul Hospital. All animal procedures were approved by the Institutional Animal Care and Use Committee of Soonchunhyang University, Seoul Hospital, Korea (IACUC2013-5). To induce ischemia and oxidative stress and to assess neovascularization, we utilized a hindlimb ischemia model, as previously described but with minor modifications.43, 44 Ischemia was induced by ligation of the proximal femoral artery and the boundary vessels of the mice. At no later than 6 h after surgery, PBS, Nor-MSCs, Hypo-MSC, Hypo-MSC transfected with PrPC-specific shRNA (sh-PRNP Hypo-MSCs), or Hypo-MSC transfected with scrambled shRNA (Scr shRNA Hypo-MSCs) were injected intramuscularly into the ischemic thigh area (5 × 105 cells/100 μl PBS per mouse; five mice per treatment group). Cells were injected into five distinct ischemic sites. Blood perfusion was assessed by measuring and comparing the ratio of blood flow in the ischemic (left) limb to that in the non-ischemic (right) limb via laser Doppler perfusion imaging analysis (Moor Instruments, Wilmington, DE, USA) on postoperative days 0, 5, 10, 15, 20, and 25.
Determination of human growth factors
The levels of hEGF, hVEGF, hFGF, and hHGF in the ischemic limb tissue (at 3 days post surgery) lysates were determined by ELISA using a commercially available ELISA kit (R&D Systems, Minneapolis, MN, USA) according to the manufacturer's recommendation. All proteins were quantified using a bicinchoninic acid assay (BCA protein assay; Thermo Scientific, Waltham, MA, USA). Growth factor expression levels were quantified by measuring the absorbance at 450 nm using a microplate reader (Tecan Group AG, Männedorf, Switzerland).
Immunohistochemistry
Ischemic thigh areas were removed at 3 or 25 days post MSC transplantation, fixed with 4% paraformaldehyde (Affymetrix, Santa Clara, CA, USA), embedded in paraffin, and sectioned. For histological analyses, sections were subjected to hematoxylin and eosin staining, followed by immunofluorescence staining in a dark room with primary antibodies against CD31, alpha-smooth muscle actin (α-SMA), cleaved caspase-3, Ki67 (Santa Cruz Biotechnology) or human nuclear antigen (HNA; Millipore, Billerica, MA, USA), and secondary antibodies conjugated to Alexa488 or Alexa594 (Thermo-Fisher Scientific). Nuclei were visualized by staining with 4′,6-diaminido-2-phenylindol (DAPI; Sigma-Aldrich). Immunostained slides were imaged by confocal microscopy (Olympus, Tokyo, Japan).
Statistical analyses
All data are presented as means±S.E.M. All experimental results were evaluated by analysis of variance, followed by a comparison of the treatment and control groups using the Bonferroni–Dunn test. P-values<0.05 were considered statistically significant.
Abbreviations
- MSCs:
-
mesenchymal stem cells
- PrPC:
-
cellular prion protein
- ROS:
-
reactive oxygen species
- HIF-1α:
-
hypoxia-inducible factor-1 alpha
- CXCR4:
-
C-X-C chemokine receptor type 4
- JAK2:
-
janus kinase 2
- STAT3:
-
signal transducer and activator of transcription 3
- NF-κB:
-
nuclear factor kappa B
- SOD:
-
superoxide dismutase
- hEGF:
-
human epidermal growth factor
- hVEGF:
-
human vascular endothelial growth factor
- hFGF:
-
human fibroblast growth factor
- hHGF:
-
human hepatocyte growth factor
- IGF-1:
-
insulin-like growth factor-1
- SDF-1:
-
stromal cell-derived factor-1
- EPO:
-
erythropoietin
- α-SMA:
-
alpha-smooth muscle actin
- HNA:
-
human nuclear antigen
- DAPI:
-
4′,6-diaminido-2-phenylindol
- FABP4:
-
fatty acid-binding protein 4
- GFP:
-
green fluorescent protein
- ERK:
-
extracellular signal-regulated protein kinase
- H2O2:
-
hydrogen peroxide
- ECL:
-
enhanced chemiluminescence
- HCl:
-
hydrochloric acid
- PI:
-
propidium iodide
- FITC:
-
fluorescein isothiocyanate
References
Wu KH, Mo XM, Han ZC, Zhou B . Stem cell engraftment and survival in the ischemic heart. Ann Thorac Surg 2011; 92: 1917–1925.
Monsel A, Zhu YG, Gennai S, Hao Q, Liu J, Lee JW . Cell-based therapy for acute organ injury: preclinical evidence and ongoing clinical trials using mesenchymal stem cells. Anesthesiology 2014; 121: 1099–1121.
Walther W, Stein U . Viral vectors for gene transfer: a review of their use in the treatment of human diseases. Drugs 2000; 60: 249–271.
Huang B, Tabata Y, Gao JQ . Mesenchymal stem cells as therapeutic agents and potential targeted gene delivery vehicle for brain diseases. J Control Release 2012; 162: 464–473.
Egred M, Al-Mohammad A, Waiter GD, Redpath TW, Semple SK, Norton M et al. Detection of scarred and viable myocardium using a new magnetic resonance imaging technique: blood oxygen level dependent (BOLD) MRI. Heart 2003; 89: 738–744.
Potier E, Ferreira E, Meunier A, Sedel L, Logeart-Avramoglou D, Petite H . Prolonged hypoxia concomitant with serum deprivation induces massive human mesenchymal stem cell death. Tissue Eng 2007; 13: 1325–1331.
Jiang S, Kh Haider H, Ahmed RP, Idris NM, Salim A, Ashraf M . Transcriptional profiling of young and old mesenchymal stem cells in response to oxygen deprivation and reparability of the infarcted myocardium. J Mol Cell Cardiol 2008; 44: 582–596.
Liu H, Liu S, Li Y, Wang X, Xue W, Ge G et al. The role of SDF-1-CXCR4/CXCR7 axis in the therapeutic effects of hypoxia-preconditioned mesenchymal stem cells for renal ischemia/reperfusion injury. PloS One 2012; 7: e34608.
Rosova I, Dao M, Capoccia B, Link D, Nolta JA . Hypoxic preconditioning results in increased motility and improved therapeutic potential of human mesenchymal stem cells. Stem Cells 2008; 26: 2173–2182.
Lee SH, Lee JH, Yoo SY, Hur J, Kim HS, Kwon SM . Hypoxia inhibits cellular senescence to restore the therapeutic potential of old human endothelial progenitor cells via the hypoxia-inducible factor-1alpha-TWIST-p21 axis. Arterioscler Thromb Vasc Biol 2013; 33: 2407–2414.
Caughey B, Baron GS . Prions and their partners in crime. Nature 2006; 443: 803–810.
Aguzzi A, Baumann F, Bremer J . The prion's elusive reason for being. Annu Rev Neurosci 2008; 31: 439–477.
Linden R, Martins VR, Prado MA, Cammarota M, Izquierdo I, Brentani RR . Physiology of the prion protein. Physiol Rev 2008; 88: 673–728.
Yusa S, Oliveira-Martins JB, Sugita-Konishi Y, Kikuchi Y . Cellular prion protein: from physiology to pathology. Viruses 2012; 4: 3109–3131.
Steele AD, Emsley JG, Ozdinler PH, Lindquist S, Macklis JD . Prion protein (PrPc) positively regulates neural precursor proliferation during developmental and adult mammalian neurogenesis. Proc Nati Acad Sci USA 2006; 103: 3416–3421.
Zhang CC, Steele AD, Lindquist S, Lodish HF . Prion protein is expressed on long-term repopulating hematopoietic stem cells and is important for their self-renewal. Proc Natl Acad Sci USA 2006; 103: 2184–2189.
Shi F, Yang Y, Wang T, Kouadir M, Zhao D, Hu S . Cellular prion protein promotes neuronal differentiation of adipose-derived stem cells by upregulating miRNA-124. J Mol Neurosci 2016; 59: 48–55.
Doeppner TR, Kaltwasser B, Schlechter J, Jaschke J, Kilic E, Bahr M et al. Cellular prion protein promotes post-ischemic neuronal survival, angioneurogenesis and enhances neural progenitor cell homing via proteasome inhibition. Cell Death Dis 2015; 6: e2024.
Lee SH, Lee JH, Han YS, Ryu JM, Yoon YM, Han HJ . Hypoxia accelerates vascular repair of endothelial colony-forming cells on ischemic injury via STAT3-BCL3 axis. Stem Cell Res Ther 2015; 6: 139.
Amiri F, Jahanian-Najafabadi A, Roudkenar MH . In vitro augmentation of mesenchymal stem cells viability in stressful microenvironments: in vitro augmentation of mesenchymal stem cells viability. Cell Stress Chaperones 2015; 20: 237–251.
Liu TM, Wu YN, Guo XM, Hui JH, Lee EH, Lim B . Effects of ectopic Nanog and Oct4 overexpression on mesenchymal stem cells. Stem Cells Dev 2009; 18: 1013–1022.
Yoon DS, Kim YH, Jung HS, Paik S, Lee JW . Importance of Sox2 in maintenance of cell proliferation and multipotency of mesenchymal stem cells in low-density culture. Cell Prolif 2011; 44: 428–440.
Wei H, Li Z, Hu S, Chen X, Cong X . Apoptosis of mesenchymal stem cells induced by hydrogen peroxide concerns both endoplasmic reticulum stress and mitochondrial death pathway through regulation of caspases, p38 and JNK. J Cell Biochemistry 2010; 111: 967–978.
Li Q, Wang Y, Deng Z . Pre-conditioned mesenchymal stem cells: a better way for cell-based therapy. Stem Cell Res Ther 2013; 4: 63.
Wang Y, Chen X, Cao W, Shi Y . Plasticity of mesenchymal stem cells in immunomodulation: pathological and therapeutic implications. Nat Immunol 2014; 15: 1009–1016.
Bizzarri A, Koehler H, Cajlakovic M, Pasic A, Schaupp L, Klimant I et al. Continuous oxygen monitoring in subcutaneous adipose tissue using microdialysis. Analytica Chimica Acta 2006; 573-574: 48–56.
Mohyeldin A, Garzon-Muvdi T, Quinones-Hinojosa A . Oxygen in stem cell biology: a critical component of the stem cell niche. Cell Stem Cell 2010; 7: 150–161.
Fehrer C, Brunauer R, Laschober G, Unterluggauer H, Reitinger S, Kloss F et al. Reduced oxygen tension attenuates differentiation capacity of human mesenchymal stem cells and prolongs their lifespan. Aging Cell 2007; 6: 745–757.
Hu X, Yu SP, Fraser JL, Lu Z, Ogle ME, Wang JA et al. Transplantation of hypoxia-preconditioned mesenchymal stem cells improves infarcted heart function via enhanced survival of implanted cells and angiogenesis. J Thorac Cardiovasc Surg 2008; 135: 799–808.
Lee YJ, Baskakov IV . The cellular form of the prion protein is involved in controlling cell cycle dynamics, self-renewal, and the fate of human embryonic stem cell differentiation. J Neurochem 2013; 124: 310–322.
Cartwright P, McLean C, Sheppard A, Rivett D, Jones K, Dalton S . LIF/STAT3 controls ES cell self-renewal and pluripotency by a Myc-dependent mechanism. Development 2005; 132: 885–896.
Barbosa HS, Fernandes TG, Dias TP, Diogo MM, Cabral JM . New insights into the mechanisms of embryonic stem cell self-renewal under hypoxia: a multifactorial analysis approach. PloS One 2012; 7: e38963.
Tu B, Du L, Fan QM, Tang Z, Tang TT . STAT3 activation by IL-6 from mesenchymal stem cells promotes the proliferation and metastasis of osteosarcoma. Cancer Lett 2012; 325: 80–88.
Brown DR, Besinger A . Prion protein expression and superoxide dismutase activity. Biochem J 1998; 334 (): 423–429.
Zanetti F, Carpi A, Menabo R, Giorgio M, Schulz R, Valen G et al. The cellular prion protein counteracts cardiac oxidative stress. Cardiovasc Res 2014; 104: 93–102.
Brown DR, Schulz-Schaeffer WJ, Schmidt B, Kretzschmar HA . Prion protein-deficient cells show altered response to oxidative stress due to decreased SOD-1 activity. Exp Neurol 1997; 146: 104–112.
Anantharam V, Kanthasamy A, Choi CJ, Martin DP, Latchoumycandane C, Richt JA et al. Opposing roles of prion protein in oxidative stress- and ER stress-induced apoptotic signaling. Free Radic Biol Med 2008; 45: 1530–1541.
Chen L, Tredget EE, Wu PY, Wu Y . Paracrine factors of mesenchymal stem cells recruit macrophages and endothelial lineage cells and enhance wound healing. PloS One 2008; 3: e1886.
Rehman J, Traktuev D, Li J, Merfeld-Clauss S, Temm-Grove CJ, Bovenkerk JE et al. Secretion of angiogenic and antiapoptotic factors by human adipose stromal cells. Circulation 2004; 109: 1292–1298.
Bronckaers A, Hilkens P, Martens W, Gervois P, Ratajczak J, Struys T et al. Mesenchymal stem/stromal cells as a pharmacological and therapeutic approach to accelerate angiogenesis. Pharmacol Ther 2014; 143: 181–196.
Weise J, Doeppner TR, Muller T, Wrede A, Schulz-Schaeffer W, Zerr I et al. Overexpression of cellular prion protein alters postischemic Erk1/2 phosphorylation but not Akt phosphorylation and protects against focal cerebral ischemia. Restor Neurol Neurosci 2008; 26: 57–64.
Weise J, Sandau R, Schwarting S, Crome O, Wrede A, Schulz-Schaeffer W et al. Deletion of cellular prion protein results in reduced Akt activation, enhanced postischemic caspase-3 activation, and exacerbation of ischemic brain injury. Stroke 2006; 37: 1296–1300.
Limbourg A, Korff T, Napp LC, Schaper W, Drexler H, Limbourg FP . Evaluation of postnatal arteriogenesis and angiogenesis in a mouse model of hind-limb ischemia. Nat Protoc 2009; 4: 1737–1746.
Niiyama H, Huang NF, Rollins MD, Cooke JP . Murine model of hindlimb ischemia. J Vis Exp 2009; 23: e1035.
Acknowledgements
This work was supported by the Soonchunhyang University Research Fund, a National Research Foundation grant funded by the Korean government (NRF-2011-0009610), NRF funded by the Ministry of Education (2016R1D1A3B01007727), a grant from the Korean Health Technology R&D Project, Ministry of Health and Welfare, Republic of Korea (HI14C2253). The funders had no role in the study design, data collection or analysis, the decision to publish, or preparation of the manuscript.
Author contributions
Yong-Seok Han: conception and design, collection and assembly of data. Jun Hee Lee: conception and design, data analysis and interpretation, manuscript writing. Yeo Min Yoon: collection and assembly of data. Chul Won Yun: collection and assembly of data. Hyunjin Noh: collection and assembly of data. Sang Hun Lee: conception and design, financial support, collection and assembly of data, data analysis and interpretation, manuscript writing, final approval of manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Edited by Y Shi
Rights and permissions
Cell Death and Disease is an open-access journal published by Nature Publishing Group. This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Han, YS., Lee, J., Yoon, Y. et al. Hypoxia-induced expression of cellular prion protein improves the therapeutic potential of mesenchymal stem cells. Cell Death Dis 7, e2395 (2016). https://doi.org/10.1038/cddis.2016.310
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cddis.2016.310
This article is cited by
-
First Clinical Experiences Using Preconditioning Approaches to Improve MSC-Based Therapies
Current Stem Cell Reports (2024)
-
Revisiting the role of mesenchymal stem cells in tuberculosis and other infectious diseases
Cellular & Molecular Immunology (2023)
-
Cellular Prion Protein Attenuates OGD/R-Induced Damage by Skewing Microglia toward an Anti-inflammatory State via Enhanced and Prolonged Activation of Autophagy
Molecular Neurobiology (2023)
-
Potential pre-activation strategies for improving therapeutic efficacy of mesenchymal stem cells: current status and future prospects
Stem Cell Research & Therapy (2022)
-
Adipose-derived mesenchymal stem cells with hypoxic preconditioning improve tenogenic differentiation
Journal of Orthopaedic Surgery and Research (2022)