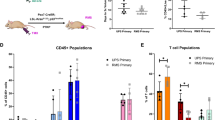
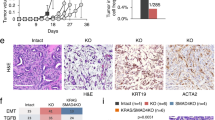
Abstract
Current mouse models do not reflect the sporadic nature of colon cancer and do not allow the analysis of antitumor immune response because of the lack of known tumor-specific antigens. Two transgenic mouse models with spontaneous tumor development were generated, directing the expression of SV40T antigen (Tag) either constitutively (Vil-Cre × LoxP-Tag-transgenic mice) or stochastically (Vil-Cre-ERT2 × LoxP-Tag-transgenic mice) into the putative stem cell region of the crypt of Lieberkühn. Tumor development and antitumor immune response were monitored. Vil-Cre × LoxP-Tag mice developed multiple adenocarcinomas of the small intestine and colon at an average age of 6 months. During the tumor development, Tag-specific immunoglobulin G (IgG) antibodies were induced in half of the mice, although they had developed neonatal cytotoxic T lymphocyte (CTL) tolerance. This model shows similarity to hereditary colon cancer but not to the sporadic tumor development. Therefore, the conditional Vil-Cre-ERT2 × LoxP-Tag mice were established, in which expression of the dormant Tag was induced by stochastic, tissue-specific activation of Cre recombinase. These mice spontaneously developed highly invasive, metastasizing colon carcinomas at an average age of 20 months. Colon carcinomas expressed epithelial and/or neuroendocrine markers depending on the grade of differentiation. Young Vil-Cre-ERT2 × LoxP-Tag mice had retained CTL responses against epitope IV of Tag. The tumors induced strong anti-Tag IgG responses. We report, for the first time, a mouse model based on stochastic, tissue-specific activation of a dormant oncogene in the colon allowing the analysis of antitumor immune response against primary colorectal cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Barker N, van Es JH, Kuipers J, Kujala P, van den Born M, Cozijnsen M et al. (2007). Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature 449: 1003–1007.
Barker N, Ridgway RA, van Es JH, van de Wetering M, Begthel H, van den Born M et al. (2009). Crypt stem cells as the cells-of-origin of intestinal cancer. Nature 457: 608–611.
Boivin GP, Washington K, Yang K, Ward JM, Pretlow TP, Russell R et al. (2003). Pathology of mouse models of intestinal cancer: consensus report and recommendations. Gastroenterology 124: 762–777.
Bos R, van Duikeren S, Morreau H, Franken K, Schumacher TN, Haanen JB et al. (2008). Balancing between antitumor efficacy and autoimmune pathology in T-cell-mediated targeting of carcinoembryonic antigen. Cancer Res 68: 8446–8455.
Boyle P, Ferlay J . (2005). Cancer incidence and mortality in Europe 2004. Ann Oncol 16: 481–488.
Brinster RL, Chen HY, Messing A, van Dyke T, Levine AJ, Palmiter RD . (1984). Transgenic mice harboring SV40 T-antigen genes develop characteristic brain tumors. Cell 37: 367–379.
Burke AB, Shekitka KM, Sobin LH . (1991). Small cell carcinomas of large intestine. Am J Clin Pathol 95: 315–321.
Calcagno SR, Li S, Colon M, Kreinest PA, Thompson EA, Fields AP et al. (2008). Oncogenic K-ras promotes early carcinogenesis in the mouse proximal colon. Int J Cancer 122: 2462–2470.
Cheng H, Leblond CP. (1974). Origin, differentiation and renewal of the four main epithelial cell types in the mouse small intestine. V. Unitarian Theory of the origin of the four epithelial cell types. Am J Anat 141: 537–561.
Corbett TH, Griswold Jr DP, Roberts BJ, Peckham JC, Schabel Jr FM . (1975). Tumor induction relationships in development of transplantable cancers of the colon in mice for chemotherapy assays, with a note on carcinogen structure. Cancer Res 35: 2434–2439.
Derbinski J, Schulte A, Kyewski B, Klein L . (2001). Promiscuous gene expression in medullary thymic epithelial cells mirrors the peripheral self. Nat Immunol 2: 1032–1039.
El Marjou F, Janssen KP, Chang BH, Li M, Hindle V, Chan L et al. (2004). Tissue-specific and inducible Cre-mediated recombination in the gut epithelium. Genesis 39: 186–193.
Enam S, Del Valle L, Lara C, Gan DD, Ortiz-Hodalgo C, Palazzo JP et al. (2002). Association of human polyomavirus JCV with colon cancer: evidence for interaction of viral T-antigen and β-catenin. Cancer Res 62: 7093–7101.
Faas SJ, Pan S, Pinkert CA, Brinster RL, Knowles BB . (1987). Simian virus 40 (SV40)-transgenic mice that develop tumors are specifically tolerant to SV40 T antigen. J Exp Med 165: 417–427.
Gum JR, Hicks JW, Gillespie AM, Rius JL, Treseler PA, Kogan SC et al. (2001). Mouse intestinal goblet cells expressing SV40 T antigen directed by the MUC2 mucin gene promoter undergo apoptosis upon migration to the villi. Cancer Res 61: 3472–3479.
Gum JR, Hicks JW, Crawley SC, Yang SC, Borowsky AD, Dahl CM et al. (2004). Mice expressing SV40 T antigen directed by the intestinal trefoil factor promoter develop tumors resembling human small cell carcinoma of the colon. Mol Cancer Res 2: 504–513.
Hauft SM, Kim SH, Schmidt GH, Pease S, Rees S, Harris S et al. (1992). Expression of SV-40 T antigen in the small intestinal epithelium of transgenic mice results in proliferative changes in the crypt and reentry of villus-associated enterocytes into the cell cycle but has no apparent effect on cellular differentiation programs and does not cause neoplastic transformation. J Cell Biol 117: 825–839.
Heyer J, Yang K, Lipkin M, Edelmann W, Kucherlapati R. (1999). Mouse models for colorectal cancer. Oncogene 18: 5325–5333.
Hori R, Murai Y, Tsuneyama K, Abdel-Aziz HO, Nomoto K, Takahashi H et al. (2005). Detection of JC virus DNA sequences in colorectal cancers in Japan. Virchows Arch 14: 1–8.
Janssen KP, el-Marjou F, Pinto D, Sastre X, Rouillard D, Fouquet C et al. (2002). Targeted expression of oncogenic K-ras in intestinal epithelium causes spontaneous tumorigenesis in mice. Gastroenterology 123: 492–504.
Jemal A, Siegel R, Ward E, Murray T, Xu J, Smigal C et al. (2006). Cancer Statistics, 2006. CA Cancer J Clin 56: 106–130.
Kinzler KW, Vogelstein B. (1996). Lessons from hereditary colorectal cancer. Cell 87: 159–170.
Kinzler KW, Vogelstein B . (1998). Landscaping the cancer terrain. Science 280: 1036–1037.
Laghi L, Randolph AE, Chauhan DP, Marra G, Major EO, Neel JV et al. (1999). JC virus DNA is present in the mucosa of the human colon and in colorectal cancers. Proc Natl Acad Sci USA 96: 7484–7489.
Lauwen MM, Zwaveling S, de Quartel L, Ferreira Mota SC, Grashorn JA, Melief CJ et al. (2008). Self-tolerance does not restrict the CD4+ T-helper response against the p53 tumor antigen. Cancer Res 68: 893–900.
Ludlow JW . (1993). Interactions between SV40 large-tumor antigen and the growth suppressor proteins pRB and p53. FASEB J 7: 866–871.
Madison BB, Dunbar L, Qiao XT, Braunstein K, Braunstein E, Gumucio DL . (2002). Cis elements of the villin gene control expression in restricted domains of the vertical (crypt) and horizontal (duodenum, cecum) axes of the intestine. J Biol Chem 277: 33275–33283.
Maunoury R, Robine S, Pringault E, Léonard N, Gaillard JA, Louvard D . (1992). Developmental regulation of villin gene expression in the epithelial cell lineages of mouse digestive and urogenital tracts. Development 115: 717–728.
Nagy A. (2000). Cre recombinase: the universal reagent for genomic tailoring. Genesis 26: 99–109.
Newcomb PA, Bush AC, Stoner GL, Lampe JW, Potter JD, Bigler J . (2004). No evidence of an association of JC virus and colon neoplasia. Cancer Epidemiol Biomarkers Prev 13: 662–666.
Otahal P, Schell TD, Hutchisnon SC, Knowles BB, Tevethia SS . (2006). Early immunization induces persistent tumor-infiltrating CD8+ T cells against an immunodominant epitope and promotes lifelong control of pancreatic tumor progression in SV40 tumor antigen transgenic mice. J Immunol 177: 3089–3099.
Pagès F, Galon J, Dieu-Nosjean MC, Tartour E, Sautès-Fridman C, Fridman WH . (2010). Immune infiltration in human tumors: a prognostic factor that should not be ignored. Oncogene 29: 1093–1102.
Palmiter RD, Chen HY, Messing A, Brinster RL . (1985). SV40 enhancer and large-T antigen are instrumental in development of choroid plexus tumours in transgenic mice. Nature 316: 457–460.
Pinto D, Robine S, Jaisser F, Marjou FE, Louvard D . (1999). Regulatory sequences of the mouse villin gene that efficiently drive transgenic expression in immature and differentiated epithelial cells of small and large intestines. J Biol Chem 274: 6476–6482.
Saam JR, Gordon JI . (1999). Inducible gene knockouts in the small intestinal and colonic epithelium. J Biol Chem 274: 38071–38082.
Sarsfield P, Anthony PP . (1990). Small cell undifferentiated (‘neuroendocrine’) carcinoma of the colon. Histopathology 16: 357–363.
Sauer B . (1998). Inducible gene targeting in mice using the Cre/lox system. Methods 14: 381–392.
Schell TD, Knowles BB, Tevethia SS . (2000). Sequential loss of cytotoxic T lymphocytes responses to simian virus 40 large T antigen epitopes in T antigen transgenic mice developing osteosarcomas. Cancer Res 60: 3002–3012.
Smith KM, Olson DC, Hirose R, Hanahan D . (1997). Pancreatic gene expression in rare cells of thymic medulla: evidence for functional contribution to T cell tolerance. Int Immunol 9: 1355–1365.
Soriano P . (1999). Generalized lacZ expression with the ROSA26 Cre reporter strain. Nat Genet 21: 70–71.
Syder AJ, Karam SM, Mills JC, Ippolito JE, Ansari HR, Farook V et al. (2004). A transgenic mouse model of metastatic carcinoma involving transdifferentiation of a gastric epithelial lineage progenitor to a neuroendocrine phenotype. Proc Natl Acad Sci USA 101: 4471–4476.
Weinreb DB . (2006). Re: Detection of JC virus DNA sequences in colorectal cancers in Japan. Virchows Arch 448: 239.
Willimsky G, Blankenstein T . (2005). Sporadic immunogenic tumours avoid destruction by inducing T-cell tolerance. Nature 437: 141–146.
Willimsky G, Czéh M, Loddenkemper C, Gellermann J, Schmidt K, Wust P et al. (2008). Immunogenicity of premalignant lesions is the primary cause of general cytotoxic T lymphocyte unresponsiveness. J Exp Med 205: 1687–1700.
Yen TH, Wright NA . (2006). The gastrointestinal tract stem cell niche. Stem Cell Rev 2: 203–212.
Acknowledgements
We thank S Spieckermann, M Rösch, C Westen and S Horn for excellent technical assistance and JC Schmollinger for reading the paper. This study was supported by the Research grants Sonderforschungsbereich SFB 633 and TR 36.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Czéh, M., Loddenkemper, C., Shalapour, S. et al. The immune response to sporadic colorectal cancer in a novel mouse model. Oncogene 29, 6591–6602 (2010). https://doi.org/10.1038/onc.2010.388
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2010.388
Keywords
This article is cited by
-
The potential oncogenic effect of tissue-specific expression of JC polyoma T antigen in digestive epithelial cells
Transgenic Research (2023)
-
Immunosuppressive plasma cells impede T-cell-dependent immunogenic chemotherapy
Nature (2015)
-
Vascular E-Selectin Expression Correlates with CD8 Lymphocyte Infiltration and Improved Outcome in Merkel Cell Carcinoma
Journal of Investigative Dermatology (2013)
-
Towards a vaccine to prevent cancer in Lynch syndrome patients
Familial Cancer (2013)