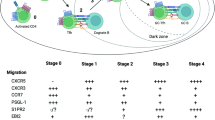
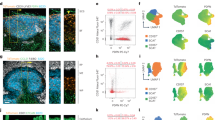
Abstract
Affinity maturation of antibodies during the course of an adaptive immune response requires germinal centre (GC) formation within B-cell follicles. Much of the current understanding of GC function has been derived from histology, but these static views have left unresolved many questions about cell movement in GCs. In this Progress article, we describe how several recent studies using time-resolved multiphoton microscopy to track GC B-cell movement within lymph nodes have shed light on the processes that influence GC B-cell dynamics.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Wolniak, K. L., Shinall, S. M. & Waldschmidt, T. J. The germinal center response. Crit. Rev. Immunol. 24, 39–65 (2004).
Hauser, A. E. et al. Definition of germinal-center B cell migration in vivo reveals predominant intrazonal circulation patterns. Immunity 26, 655–667 (2007).
Allen, C. D., Okada, T., Tang, H. L. & Cyster, J. G. Imaging of germinal center selection events during affinity maturation. Science 315, 528–531 (2007).
Schwickert, T. A. et al. In vivo imaging of germinal centres reveals a dynamic open structure. Nature 446, 83–87 (2007).
Cahalan, M. D., Parker, I., Wei, S. H. & Miller, M. J. Two-photon tissue imaging: seeing the immune system in a fresh light. Nature Rev. Immunol. 2, 872–880 (2002).
Germain, R. N., Miller, M. J., Dustin, M. L. & Nussenzweig, M. C. Dynamic imaging of the immune system: progress, pitfalls and promise. Nature Rev. Immunol. 6, 497–507 (2006).
Sumen, C., Mempel, T. R., Mazo, I. B. & von Andrian, U. H. Intravital microscopy: visualizing immunity in context. Immunity 21, 315–329 (2004).
MacLennan, I. C. Germinal centers. Annu. Rev. Immunol. 12, 117–139 (1994).
Liu, Y. J., Johnson, G. D., Gordon, J. & MacLennan, I. C. Germinal centres in T-cell-dependent antibody responses. Immunol. Today 13, 17–21 (1992).
Camacho, S. A., Kosco-Vilbois, M. H. & Berek, C. The dynamic structure of the germinal center. Immunol. Today 19, 511–514 (1998).
Nieuwenhuis, P. & Opstelten, D. Functional anatomy of germinal centers. Am. J. Anat. 170, 421–435 (1984).
Hardie, D. L., Johnson, G. D., Khan, M. & MacLennan, I. C. Quantitative analysis of molecules which distinguish functional compartments within germinal centers. Eur. J. Immunol. 23, 997–1004 (1993).
Rogerson, B., Hackett, J. Jr, Peters, A., Haasch, D. & Storb, U. Mutation pattern of immunoglobulin transgenes is compatible with a model of somatic hypermutation in which targeting of the mutator is linked to the direction of DNA replication. EMBO J. 10, 4331–4341 (1991).
Neuberger, M. S. et al. Somatic hypermutation at A•T pairs: polymerase error versus dUTP incorporation. Nature Rev. Immunol. 5, 171–178 (2005).
Liu, Y. J., Grouard, G., de Bouteiller, O. & Banchereau, J. Follicular dendritic cells and germinal centers. Int. Rev. Cytol. 166, 139–179 (1996).
Szakal, A. K., Kosco, M. H. & Tew, J. G. Microanatomy of lymphoid tissue during humoral immune responses: structure function relationships. Annu. Rev. Immunol. 7, 91–109 (1989).
Qin, D. et al. Fcγ receptor IIB on follicular dendritic cells regulates the B cell recall response. J. Immunol. 164, 6268–6275 (2000).
Carroll, M. C. The role of complement and complement receptors in induction and regulation of immunity. Annu. Rev. Immunol. 16, 545–568 (1998).
Liu, Y. J. et al. Follicular dendritic cells specifically express the long CR2/CD21 isoform. J. Exp. Med. 185, 165–170 (1997).
Cattoretti, G. et al. Stages of germinal center transit are defined by B cell transcription factor coexpression and relative abundance. J. Immunol. 177, 6930–6939 (2006).
Liu, Y. J., Zhang, J., Lane, P. J., Chan, E. Y. & MacLennan, I. C. Sites of specific B cell activation in primary and secondary responses to T cell-dependent and T cell-independent antigens. Eur. J. Immunol. 21, 2951–2962 (1991).
Clarke, S. H. et al. Inter- and intraclonal diversity in the antibody response to influenza hemagglutinin. J. Exp. Med. 161, 687–704 (1985).
Sablitzky, F., Wildner, G. & Rajewsky, K. Somatic mutation and clonal expansion of B cells in an antigen-driven immune response. EMBO J. 4, 345–350 (1985).
Weiss, U. & Rajewsky, K. The repertoire of somatic antibody mutants accumulating in the memory compartment after primary immunization is restricted through affinity maturation and mirrors that expressed in the secondary response. J. Exp. Med. 172, 1681–1689 (1990).
Shlomchik, M. J., Litwin, S. & Weigert, M. The influence of somatic mutation on clonal expansion. Prog. Immunol. Proc. 7th Int. Cong. Immunol. 7, 415–423 (1990).
Kleinstein, S. H. & Singh, J. P. Toward quantitative simulation of germinal center dynamics: biological and modeling insights from experimental validation. J. Theor. Biol. 211, 253–275 (2001).
Shlomchik, M. J., Watts, P., Weigert, M. G. & Litwin, S. Clone: a Monte-Carlo computer simulation of B cell clonal expansion, somatic mutation, and antigen-driven selection. Curr. Top. Microbiol. Immunol. 229, 173–197 (1998).
Liu, Y. J. et al. Mechanism of antigen-driven selection in germinal centres. Nature 342, 929–931 (1989).
Wu, J., Qin, D., Burton, G. F., Szakal, A. K. & Tew, J. G. Follicular dendritic cell-derived antigen and accessory activity in initiation of memory IgG responses in vitro. J. Immunol. 157, 3404–3411 (1996).
Liu, Y. J., de Bouteiller, O. & Fugier-Vivier, I. Mechanisms of selection and differentiation in germinal centers. Curr. Opin. Immunol. 9, 256–262 (1997).
Kosco, M. H., Pflugfelder, E. & Gray, D. Follicular dendritic cell-dependent adhesion and proliferation of B cells in vitro. J. Immunol. 148, 2331–2339 (1992).
Hannum, L. G., Haberman, A. M., Anderson, S. M. & Shlomchik, M. J. Germinal center initiation, variable gene region hypermutation, and mutant B cell selection without detectable immune complexes on follicular dendritic cells. J. Exp. Med. 192, 931–942 (2000).
Boes, M. et al. Enhanced B-1 cell development, but impaired IgG antibody responses in mice deficient in secreted IgM. J. Immunol. 160, 4776–4787 (1998).
Ehrenstein, M. R., O'Keefe, T. L., Davies, S. L. & Neuberger, M. S. Targeted gene disruption reveals a role for natural secretory IgM in the maturation of the primary immune response. Proc. Natl Acad. Sci. USA 95, 10089–10093 (1998).
Casamayor-Palleja, M., Khan, M. & MacLennan, I. C. A subset of CD4+ memory T cells contains preformed CD40 ligand that is rapidly but transiently expressed on their surface after activation through the T cell receptor complex. J. Exp. Med. 181, 1293–1301 (1995).
Vinuesa, C. G., Tangye, S. G., Moser, B. & Mackay, C. R. Follicular B helper T cells in antibody responses and autoimmunity. Nature Rev. Immunol. 5, 853–865 (2005).
Kepler, T. B. & Perelson, A. S. Cyclic re-entry of germinal center B cells and the efficiency of affinity maturation. Immunol. Today 14, 412–415 (1993).
Oprea, M. & Perelson, A. S. Somatic mutation leads to efficient affinity maturation when centrocytes recycle back to centroblasts. J. Immunol. 158, 5155–5162 (1997).
MacLennan, I. C., Johnson, G. D., Liu, Y. J. & Gordon, J. The heterogeneity of follicular reactions. Res. Immunol. 142, 253–257 (1991).
Haberman, A. M. & Shlomchik, M. J. Reassessing the function of immune-complex retention by follicular dendritic cells. Nature Rev. Immunol. 3, 757–764 (2003).
de Vinuesa, C. G. et al. Germinal centers without T cells. J. Exp. Med. 191, 485–494 (2000).
Meyer-Hermann, M. E. & Maini, P. K. Cutting edge: back to 'one-way' germinal centers. J. Immunol. 174, 2489–2493 (2005).
Shinall, S. M., Gonzalez-Fernandez, M., Noelle, R. J. & Waldschmidt, T. J. Identification of murine germinal center B cell subsets defined by the expression of surface isotypes and differentiation antigens. J. Immunol. 164, 5729–5738 (2000).
Acknowledgements
We are grateful to all our colleagues for discussion and advice, in particular S. Kleinstein, U. Hershberg, T. Junt, T. Mempel and U. von Andrian. We apologize to the many investigators who have made important contributions to this field that could not be cited owing to space limitations. A.M.H. is supported by the S.L.E. Lupus Foundation, A.E.H. by the Deutsche Forschungsgemeinschaft (DFG) Forschungsstipendium HA5354/1 and M.J.S. by the National Institutes of Health, USA, grant R01-AI43603.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Rights and permissions
About this article
Cite this article
Hauser, A., Shlomchik, M. & Haberman, A. In vivo imaging studies shed light on germinal-centre development. Nat Rev Immunol 7, 499–504 (2007). https://doi.org/10.1038/nri2120
Issue Date:
DOI: https://doi.org/10.1038/nri2120
This article is cited by
-
Dynamics of B cells in germinal centres
Nature Reviews Immunology (2015)
-
Cellular imaging in rheumatic diseases
Nature Reviews Rheumatology (2015)
-
How oligoclonal are germinal centers? A new method for estimating clonal diversity from immunohistological sections
BMC Bioinformatics (2013)
-
A Model for Migratory B Cell Oscillations from Receptor Down-Regulation Induced by External Chemokine Fields
Bulletin of Mathematical Biology (2013)
-
The proto-oncogene MYC is required for selection in the germinal center and cyclic reentry
Nature Immunology (2012)