Abstract
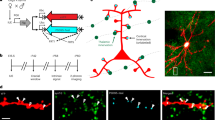
Can neuronal morphology predict functional synaptic circuits? In the rat barrel cortex, 'barrels' and 'septa' delineate an orderly matrix of cortical columns. Using quantitative laser scanning photostimulation we measured the strength of excitatory projections from layer 4 (L4) and L5A to L2/3 pyramidal cells in barrel- and septum-related columns. From morphological reconstructions of excitatory neurons we computed the geometric circuit predicted by axodendritic overlap. Within most individual projections, functional inputs were predicted by geometry and a single scale factor, the synaptic strength per potential synapse. This factor, however, varied between projections and, in one case, even within a projection, up to 20-fold. Relationships between geometric overlap and synaptic strength thus depend on the laminar and columnar locations of both the pre- and postsynaptic neurons, even for neurons of the same type. A large plasticity potential appears to be incorporated into these circuits, allowing for functional 'tuning' with fixed axonal and dendritic arbor geometry.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Change history
15 May 2005
Changed text
Notes
Note:In the version of this article initially published online, the legend for Figure 6 contained an error. The first line of the description of Figure 6a should read, “Average ratio of functional/geometric connectivity (< Qxy/ Gxy>) for projections to L2 septum and L3septumcells.” This error has been corrected for the HTML and print versions of the article.
References
Douglas, R.J. & Martin, K.A.C. Neuronal circuits of the neocortex. Annu. Rev. Neurosci. 27, 419–451 (2004).
Uttley, A.M. The probability of neural connexions. Proc. R. Soc. Lond. B 144, 229–240 (1955).
White, E.L. Cortical Circuits (Birkhäuser, Boston, 1989).
Braitenberg, V.B. & Schüz, A. Cortex: Statistics and Geometry of Neuronal Connectivity (Springer, Berlin, 1998).
Peters, A. Thalamic input to the cerebral cortex. Trends Neurosci. 2, 1183–1185 (1979).
Ramon y Cajal, S. Sur la structure de l'ecorce cerebrale de quelques mamiferes. Cellule 7, 125–176 (1891).
Gilbert, C.D. Microcircuitry of the visual cortex. Annu. Rev. Neurosci. 6, 217–247 (1983).
Douglas, R.J. & Martin, K.A.C. A functional microcircuit for cat visual cortex. J. Physiol. (Lond.) 440, 735–769 (1991).
Kalisman, N., Silberberg, G. & Markram, H. Deriving physical connectivity from neuronal morphology. Biol. Cybern. 88, 210–218 (2003).
Hellwig, B. A quantitative analysis of the local connectivity between pyramidal neurons in layers 2/3 of the rat visual cortex. Biol. Cybern. 82, 111–121 (2000).
Binzegger, T., Douglas, R.J. & Martin, K.A.C. A quantitative map of the circuit of cat primary visual cortex. J. Neurosci. 24, 8441–8453 (2004).
Petersen, C.C.H. & Sakmann, B. The excitatory neuronal network of rat layer 4 barrel cortex. J. Neurosci. 20, 7579–7586 (2000).
Koralek, K.A., Jensen, K.F. & Killackey, H.P. Evidence for two complementary patterns of thalamic input to the rat somatosensory cortex. Brain Res. 463, 346–351 (1988).
Chmielowska, J., Carvell, G.E. & Simons, D.J. Spatial organization of thalamocortical and corticothalamic projection systems in the rat SmI barrel cortex. J. Comp. Neurol. 285, 325–338 (1989).
Lu, S.M. & Lin, R.C.S. Thalamic afferents of the rat barrel cortex: a light- and electron-microscopic study using Phaseolus vulgaris leucoagglutinin as an anterograde tracer. Somatosens. Mot. Res. 10, 1–16 (1992).
Kim, U. & Ebner, F.F. Barrels and septa: separate circuits in rat barrels field cortex. J. Comp. Neurol. 408, 489–505 (1999).
Shepherd, G.M.G., Pologruto, T.A. & Svoboda, K. Circuit analysis of experience-dependent plasticity in the developing rat barrel cortex. Neuron 38, 277–289 (2003).
Brecht, M., Roth, A. & Sakmann, B. Dynamic receptive fields of reconstructed pyramidal cells in layers 3 and 2 of rat somatosensory barrel cortex. J. Physiol. 553, 243–265 (2003).
Callaway, E.M. & Katz, L.C. Photostimulation using caged glutamate reveals functional circuitry in living brain slices. Proc. Natl. Acad. Sci. USA 90, 7661–7665 (1993).
Dantzker, J.L. & Callaway, E.M. Laminar sources of synaptic input to cortical inhibitory interneurons and pyramidal neurons. Nat. Neurosci. 3, 701–707 (2000).
Bureau, I., Shepherd, G.M.G. & Svoboda, K. Precise development of functional and anatomical columns in the neocortex. Neuron 42, 789–801 (2004).
Shepherd, G.M.G. & Svoboda, K. Laminar and columnar organization of ascending excitatory projections to layer 2/3 pyramidal neurons in rat barrel cortex. J. Neurosci. (in the press).
Ren, J.Q., Aika, Y., Heizmann, C.W. & Kosaka, T. Quantitative analysis of neurons and glial cells in the rat somatosensory cortex, with special reference to GABAergic neurons and parvalbumin-containing neurons. Exp. Brain Res. 92, 1–14 (1992).
Keller, A. & Carlson, G.C. Neonatal whisker clipping alters intracortical, but not thalamocortical projections, in rat barrel cortex. J. Comp. Neurol. 412, 83–94 (1999).
Bender, K.J., Rangel, J. & Feldman, D.E. Development of columnar topography in the excitatory layer 4 to layer 2/3 projection in rat barrel cortex. J. Neurosci. 23, 8759–8770 (2003).
Lübke, J., Egger, V., Sakmann, B. & Feldmeyer, D. Columnar organization of dendrites and axons of single and synaptically coupled excitatory spiny neurons in layer 4 of the rat barrel cortex. J. Neurosci. 20, 5300–5311 (2000).
Petersen, C.C.H. & Sakmann, B. Functionally independent columns of rat somatosensory barrel cortex revealed with voltage-sensitive dye imaging. J. Neurosci. 21, 8435–8446 (2001).
Staiger, J.F. et al. Functional diversity of layer IV spiny neurons in rat somatosensory cortex: quantitative morphology of electrophysiologically characterized and biocytin labeled cells. Cereb. Cortex (2004).
Gottlieb, J.P. & Keller, A. Intrinsic circuitry and physiological properties of pyramidal neurons in rat barrel cortex. Exp. Brain Res. 115, 47–60 (1997).
Manns, I.D., Sakmann, B. & Brecht, M. Sub- and suprathreshold receptive field properties of pyramidal neurones in layers 5A and 5B of rat somatosensory barrel cortex. J. Physiol. (Lond.) 556, 601–622 (2004).
Zhang, Z.W. & Deschênes, M. Intracortical axonal projections of lamina VI cells of the primary somatosensory cortex in the rat: a single-cell labeling study. J. Neurosci. 17, 6365–6379 (1997).
Burkhalter, A. Intrinsic connections of rat primary visual cortex: laminar organization of axonal projections. J. Comp. Neurol. 279, 171–186 (1989).
Stepanyants, A., Hof, P.R. & Chklovskii, D.B. Geometry and structural plasticity of synaptic connectivity. Neuron 34, 275–288 (2002).
Peters, A. & Feldman, M.L. The projection of the lateral geniculate nucleus to area 17 of the rat cerebral cortex. I. General description. J. Neurocytol. 5, 63–84 (1976).
Song, S., Sjöström, P.J., Reigl, M., Nelson, S. & Chklovskii, D.B. Highly nonrandom features of synaptic connectivity in local cortical circuits. PLoS Biol. 3, 1–13 (2005).
Yoshimura, Y., Dantzker, J.L. & Callaway, E.M. Excitatory cortical neurons form fine-scale functional networks. Nature 433, 868–873 (2005).
Kalisman, N., Silberberg, G. & Markram, H. The neocortical microcircuit as a tabula rasa. Proc. Natl. Acad. Sci. USA 102, 880–885 (2005).
Gupta, A., Wang, Y. & Markram, H. Organizing principles for a diversity of GABAergic interneurons and synapses in the neocortex. Science 287, 273–278 (2000).
Stepanyants, A., Tamas, G. & Chklovskii, D.B. Class-specific features of neuronal wiring. Neuron 43, 251–259 (2004).
Yabuta, N.H., Sawatari, A. & Callaway, E.M. Two functional channels from primary visual cortex to dorsal visual cortical areas. Science 292, 297–300 (2001).
Schubert, D., Kötter, R., Zilles, K., Luhmann, H.J. & Staiger, J.F. Cell type-specific circuits of cortical layer IV spiny neurons. J. Neurosci. 23, 2961–2970 (2003).
Sawatari, A. & Callaway, E.M. Diversity and cell type specificity of local excitatory connections to neurons in layer 3B of monkey primary visual cortex. Neuron 25, 459–471 (2000).
Schubert, D. et al. Layer-specific intracolumnar and transcolumnar functional connectivity of layer V pyramidal cells in rat barrel cortex. J. Neurosci. 21, 3580–3592 (2001).
Linkenhoker, B.A. & Knudsen, E.I. Incremental training increases the plasticity of the auditory space map in adult barn owls. Nature 419, 293–296 (2002).
Kaas, J. & Ebner, F. Intrathalamic connections: a new way to modulate cortical plasticity? Nat. Neurosci. 1, 341–342 (1998).
Ichinohe, N., Fujiyama, F., Kaneko, T. & Rockland, K.S. Honeycomb-like mosaic at the border of layers 1 and 2 in the cerebral cortex. J. Neurosci. 23, 1372–1382 (2003).
Mountcastle, V.B. Modality and topographic properties of single neurons of cat's somatic sensory cortex. J. Neurophysiol. 20, 408–434 (1957).
Trachtenberg, J.T. et al. Long-term in vivo imaging of experience-dependent synaptic plasticity in adult cortex. Nature 420, 788–794 (2002).
Canepari, M., Nelson, L., Papageorgiou, G., Corrie, J.E. & Ogden, D. Photochemical and pharmacological evaluation of 7-nitroindolinyl-and 4-methoxy-7-nitroindolinyl-amino acids as novel, fast caged neurotransmitters. J. Neurosci. Methods 112, 29–42 (2001).
Grace, A.A. & Llinás, R. Morphological artifacts induced in intracellularly stained neurons by dehydration: circumvention using rapid dimethyl sulfoxide clearing. Neuroscience 16, 461–475 (1985).
Acknowledgements
We thank K. Zito and V. Scheuss for a critical reading of the manuscript, members of the Svoboda laboratory for useful discussions, J. Huang and C. Wu for access to their Neurolucida system and B.J. Burbach and C. Zhang for technical assistance. Funded by the Howard Hughes Medical Institute (G.S. and K.S.), US National Institutes of Health (D.C., A.S. and K.S.), Klingenstein Foundation (D.C.) and Human Frontier Science Program (I.B.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Geometric input calculation and optimal barrel cortex template. (a) Schematic depicting method used to calculated geometric input from a particular location. See Supplementary Methods for description. (b) Optimal template of the barrel cortex and the respective positions of all recorded and reconstructed neurons. In L2/3, positions of pyramidal cells for which LSPS maps where obtained are marked by green dots (32 cells). Positions of reconstructed cells used to produce potential connectivity maps are marked with red dots in L2/3 (13 pyramidal cells), and blue dots in L4 and L5A (49 spiny stellate and pyramidal cells). Bottom and left axes provide the scale in micrometers. Right axis indicates distance from pia. Numbers to left of right axis indicate widths of laminae. (GIF 8 kb)
Supplementary Fig. 2
Comparisons of functional and geometric input. (a) Inputs to L2barrel neurons: i, horizontal profile of functional input; ii, horizontal profile of geometric input; iii, plot of functional versus geometric horizontal profiles. (b) Inputs to L3barrel neurons. (c) Inputs to L2septum neurons. (d) Inputs to L3septumneurons. (e) Plot of the ratios of functional to geometric input. Projections from L4 are indicated by green lines (see inset); those from L5A indicated by blue lines. Home-column projections are indicated by black lines and bars; those from left and right side columns indicated by red lines and bars. Values were calculated using different home-column widths (from 100 to 400 m) and for the side columns separately or combined, as indicated in the boxed annotated example for the L4 L2barrel projection. (GIF 33 kb)
Rights and permissions
About this article
Cite this article
Shepherd, G., Stepanyants, A., Bureau, I. et al. Geometric and functional organization of cortical circuits. Nat Neurosci 8, 782–790 (2005). https://doi.org/10.1038/nn1447
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1447
This article is cited by
-
Single-neuron analysis of dendrites and axons reveals the network organization in mouse prefrontal cortex
Nature Neuroscience (2023)
-
Structure and function of a neocortical synapse
Nature (2021)
-
MAP2 is differentially phosphorylated in schizophrenia, altering its function
Molecular Psychiatry (2021)
-
Circuit asymmetries underlie functional lateralization in the mouse auditory cortex
Nature Communications (2019)
-
Investigating large-scale brain dynamics using field potential recordings: analysis and interpretation
Nature Neuroscience (2018)