Abstract
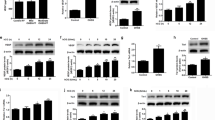
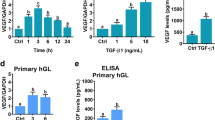
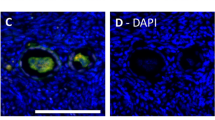
The development and endocrine function of the ovarian corpus luteum (CL) are dependent on the growth of new capillary vessels. Although several molecules have been implicated as mediators of CL angiogenesis, at present there is no direct evidence for the involvement of any. Here we report the unexpected finding that treatment with truncated soluble Flt-1 receptors, which inhibit vascular endothelial growth factor (VEGF) bioac-tivity, resulted in virtually complete suppression of CL angiogenesis in a rat model of hormonally induced ovulation. This effect was associated with inhibition of CL development and progesterone release. Failure of maturation of the en-dometrium was also observed. Areas of ischemic necrosis were demonstrated in the corpora lutea (CLs) of treated animals. However, no effect on the preexisting ovarian vasculature was observed. These findings demonstrate that, in spite of the redundancy of potential mediators, VEGF is essential for CL angiogenesis. Furthermore, they have implications for the control of fertility and the treatment of ovarian disorders characterized by hypervascularity and hyperplasia.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Bassett, D.E.L. The changes in the vascular pattern of the ovary of the albino rat during the estrous cycle. Am. J. Anat. 73, 251–278 (1943).
Gospodarowicz, D. & Takral, T.K. Production of a corpus luteum angiogenic factor responsible for proliferation of capillaries and neovascularization of the corpus luteum. Proc. Natl. Acad. Sci. USA 75, 847–851 (1978).
Ferrara, N. & Davis-Smyth, T. The biology of vascular endothelial growth factor. Endocr. Rev. 18, 4–25(1997).
Phillips, H.S., Hains, J., Leung, D.W. & Ferrara, N. Vascular endothelial growth factor is expressed in rat corpus luteum. Endocrinology 127, 965–967 (1990).
Kamat, B.R., Brown, L.F., Manseau, E.J., Senger, D.R. & Dvorak, H.F. Expression of vascular permeability factor/vascular endothelial growth factor by human granu-losa and theca lutein cells: Role in corpus luteum development. Am. J. Pathol. 146, 157–165(1995).
Shweiki, D., Itin, A., Neufeld C., Gitay-Goren, H. & Keshet, E. Patterns of expression of vascular endothelial growth factor (VEGF) and VEGF receptors in mice suggest a role in hormonally-mediated angiogenesis. J. Clin. Invest. 91, 2235–2243 (1993).
Gospodarowicz, D., Cheng, J., Lui, G.M., Baird, A. & Bohlen, P. Corpus luteum angiogenesis factor is related to fibroblast growth factor. Endocrinology 117, 2283–2290 (1985).
Maisonpierre, P.C. et al. Angiopoietin-2, a natural antagonist for Tie-2 that disrupts in vivo angiogenesis. Science 277, 55–60 (1997).
de Vries, C. et al. The fms-like tyrosine kinase, a receptor for vascular endothelial growth factor. Science 255, 989–991 (1992).
Aiello, L.P. et al. Suppression of retinal neovascularization in vivo by inhibition of vascular endothelial growth factor (VEGF) using soluble VEGF-receptor chimeric proteins. Proc. Natl. Acad. Sci. USA 92, 10457–10461 (1995).
Kendall, R.L., Wang, G. & Thomas, K.A. Identification of a natural soluble form of the vascular endothelial growth factor receptor, FLT-1, and its heterodimerization with KDR. Biochem. Biophys. Res. Commun. 226, 324–328 (1996).
Davis-Smyth, T., Chen, H., Park, J., Presta, L.G. & Ferrara, N. The second im-munoglobulin-like domain of the VEGF tyrosine kinase receptor Flt-1 determines ligand binding and may initiate a signal transduction cascade. EMBO J. 15, 4919–4927 (1996).
Gates, A.H. Maximizing yield and developmental uniformity of eggs, in Methods in Mammalian Embryology. (ed. Daniels, J.) 46–76 (Freeman, San Francisco, 1971).
Rothchild, I. The regulation of the mammalian corpus luteum. Rec. Prog. Horm. Res. 37, 183–298(1981).
Park, J.E., Chen, H.H., Winer, J., Houck, K.A. & Ferrara, N. Placenta growth factor: Potentiation of vascular endothelial growth factor bioactivity, in vitro and in vivo, and high affinity binding to Flt-1 but not to Flk-1/KDR. J. Biol. Chem. 269, 25646–25654(1994).
Cao, Y. et al. Heterodimers of placenta growth factor/vascular endothelial growth factor: Endothelial activity, tumor cell expression, and high affinity binding to Flk-1/KDR J. Biol. Chem. 271, 3154–3162 (1996).
Smith, L.E. et al. Essential role of growth hormone in ischemia-induced retinal neovascularization. Science 276, 1706–1709 (1997).
Goldziher, J.W. & Green, J.A. The polycystic ovary. I. Clinical and histologic features. J. Clin. Endocrinol. Metab. 22, 325–332 (1962).
Engel, T., Jewelewicz, R., Dyrenfurth, I., Speroff, L. & Vande Wiele, R.L. Ovarian hy-perstimulation syndrome: Report of one case with notes on pathogenesis and treatment. Am. J. Obstet. Cynecol. 112, 1052–1057 (1972).
McClure, N. et al. Vascular endothelial growth factor as a capillary permeability agent in ovarian hyperstimulation syndrome. Lancet 344, 235–269 (1994).
Murthy, Y.S., Arronet, G.H. & Pareck, M.C. Luteal phase inadequacy. Obstet. Gynecol. 36, 758–772 (1970).
Capon, D.J. et al. Designing CD4 immunoadhesins for AIDS therapy. Nature 337, 525–531 (1989).
Finnerty, H. et al. Molecular cloning of murine FLT and FLT4. Oncogene 8, 2293–2298 (1993).
Lucas, B.K. et al. High-level production of recombinant proteins using dicistronic DHFR into expression vector. Nucleic Acid Res. 24, 1774–1779 (1996).
Chisholm, V. High efficiency gene transfer into mammalian cells, in DNA Cloning 4: A Practical Approach, Mammalian systems,(eds. Glover, D.M. & Hames, B.D.) 1–39 (Oxford University Press, New York 1995).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ferrara, N., Chen, H., Davis-Smyth, T. et al. Vascular endothelial growth factor is essential for corpus luteum angiogenesis. Nat Med 4, 336–340 (1998). https://doi.org/10.1038/nm0398-336
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm0398-336
This article is cited by
-
Biology and therapeutic targeting of vascular endothelial growth factor A
Nature Reviews Molecular Cell Biology (2023)
-
Effect of low VEGF on lung development and function
Transgenic Research (2021)
-
Targeting the Angiopoietin/Tie Pathway: Prospects for Treatment of Retinal and Respiratory Disorders
Drugs (2021)
-
A potential function of RLIP76 in the ovarian corpus luteum
Journal of Ovarian Research (2019)
-
C/EBPβ regulates Vegf gene expression in granulosa cells undergoing luteinization during ovulation in female rats
Scientific Reports (2019)