Abstract
To investigate how the human γδ T cell pool is shaped during ontogeny and how it is regenerated after transplantation of hematopoietic stem cells (HSCs), we applied an RNA-based next-generation sequencing approach to monitor the dynamics of the repertoires of γδ T cell antigen receptors (TCRs) before and after transplantation in a prospective cohort study. We found that repertoires of rearranged genes encoding γδ TCRs (TRG and TRD) in the peripheral blood of healthy adults were stable over time. Although a large fraction of human TRG repertoires consisted of public sequences, the TRD repertoires were private. In patients undergoing HSC transplantation, γδ T cells were quickly reconstituted; however, they had profoundly altered TCR repertoires. Notably, the clonal proliferation of individual virus-reactive γδ TCR sequences in patients with reactivation of cytomegalovirus revealed strong evidence for adaptive anti-viral γδ T cell immune responses.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
Change history
14 February 2018
In the version of this Article originally published, in Acknowledgments section "Deutsche José Carreras Leukämie-Stiftung e.V. (DJCLS R12/29 to C.K. and I.P.)", text were missing. The text has been included in PDF and XML. These have been corrected after print.
References
Beldjord, K., Beldjord, C., Macintyre, E., Even, P. & Sigaux, F. Peripheral selection of Vδ1+ cells with restricted T cell receptor δ gene junctional repertoire in the peripheral blood of healthy donors. J. Exp. Med. 178, 121–127 (1993).
Holtmeier, W., Chowers, Y., Lumeng, A., Morzycka-Wroblewska, E. & Kagnoff, M.F. The δ T cell receptor repertoire in human colon and peripheral blood is oligoclonal irrespective of V region usage. J. Clin. Invest. 96, 1108–1117 (1995).
Chowers, Y., Holtmeier, W., Harwood, J., Morzycka-Wroblewska, E. & Kagnoff, M.F. The Vδ1 T cell receptor repertoire in human small intestine and colon. J. Exp. Med. 180, 183–190 (1994).
Bendelac, A., Bonneville, M. & Kearney, J.F. Autoreactivity by design: innate B and T lymphocytes. Nat. Rev. Immunol. 1, 177–186 (2001).
Vermijlen, D. & Prinz, I. Ontogeny of innate T lymphocytes - some innate lymphocytes are more innate than others. Front. Immunol. 5, 486 (2014).
Sheridan, B.S. et al. γδ T cells exhibit multifunctional and protective memory in intestinal tissues. Immunity 39, 184–195 (2013).
Murphy, A.G. et al. Staphylococcus aureus infection of mice expands a population of memory γδ T cells that are protective against subsequent infection. J. Immunol. 192, 3697–3708 (2014).
Ramirez-Valle, F., Gray, E.E. & Cyster, J.G. Inflammation induces dermal Vγ4+ γδ T17 memory-like cells that travel to distant skin and accelerate secondary IL-17-driven responses. Proc. Natl. Acad. Sci. USA 112, 8046–8051 (2015).
Hartwig, T., Pantelyushin, S., Croxford, A.L., Kulig, P. & Becher, B. Dermal IL-17-producing γδ T cells establish long-lived memory in the skin. Eur. J. Immunol. 45, 3022–3033 (2015).
Sherwood, A.M. et al. Deep sequencing of the human TCRγ and TCRβ repertoires suggests that TCRβ rearranges after αβ and γδ T cell commitment. Sci. Transl. Med. 3, 90ra61 (2011).
Schumacher, J.A., Duncavage, E.J., Mosbruger, T.L., Szankasi, P.M. & Kelley, T.W. A comparison of deep sequencing of TCRG rearrangements vs traditional capillary electrophoresis for assessment of clonality in T-cell lymphoproliferative disorders. Am. J. Clin. Pathol. 141, 348–359 (2014).
Harden, J.L., Hamm, D., Gulati, N., Lowes, M.A. & Krueger, J.G. Deep sequencing of the T-cell receptor repertoire demonstrates polyclonal T-cell infiltrates in psoriasis. F1000 Res. 4, 460 (2015).
Casorati, G., De Libero, G., Lanzavecchia, A. & Migone, N. Molecular analysis of human γ/δ+ clones from thymus and peripheral blood. J. Exp. Med. 170, 1521–1535 (1989).
McVay, L.D., Carding, S.R., Bottomly, K. & Hayday, A.C. Regulated expression and structure of T cell receptor γ/δ transcripts in human thymic ontogeny. EMBO J. 10, 83–91 (1991).
Davodeau, F. et al. Peripheral selection of antigen receptor junctional features in a major human γδ subset. Eur. J. Immunol. 23, 804–808 (1993).
Hirokawa, M., Horiuchi, T., Kawabata, Y., Kitabayashi, A. & Miura, A.B. Reconstitution of γδ T cell repertoire diversity after human allogeneic hematopoietic cell transplantation and the role of peripheral expansion of mature T cell population in the graft. Bone Marrow Transplant. 26, 177–185 (2000).
Fujishima, N. et al. Skewed T cell receptor repertoire of Vδ1+ γδ T lymphocytes after human allogeneic haematopoietic stem cell transplantation and the potential role for Epstein-Barr virus-infected B cells in clonal restriction. Clin. Exp. Immunol. 149, 70–79 (2007).
Haas, J.D. et al. Development of interleukin-17-producing γδ T cells is restricted to a functional embryonic wave. Immunity 37, 48–59 (2012).
Godder, K.T. et al. Long term disease-free survival in acute leukemia patients recovering with increased γδ T cells after partially mismatched related donor bone marrow transplantation. Bone Marrow Transplant. 39, 751–757 (2007).
Scheper, W. et al. γδ T cells elicited by CMV reactivation after allo-SCT cross-recognize CMV and leukemia. Leukemia 27, 1328–1338 (2013).
Scheper, W., Sebestyen, Z. & Kuball, J. Cancer immunotherapy using γδ T cells: dealing with diversity. Front. Immunol. 5, 601 (2014).
Airoldi, I. et al. γδ T-cell reconstitution after HLA-haploidentical hematopoietic transplantation depleted of TCR-αβ+/CD19+ lymphocytes. Blood 125, 2349–2358 (2015).
Scheper, W., Grunder, C., Straetemans, T., Sebestyen, Z. & Kuball, J. Hunting for clinical translation with innate-like immune cells and their receptors. Leukemia 28, 1181–1190 (2014).
Dechanet, J. et al. Implication of γδ T cells in the human immune response to cytomegalovirus. J. Clin. Invest. 103, 1437–1449 (1999).
Dechanet, J. et al. Major expansion of γδ T lymphocytes following cytomegalovirus infection in kidney allograft recipients. J. Infect. Dis. 179, 1–8 (1999).
Knight, A. et al. The role of Vδ2-negative γδ T cells during cytomegalovirus reactivation in recipients of allogeneic stem cell transplantation. Blood 116, 2164–2172 (2010).
Chien, Y.H. & Bonneville, M. γδ T cell receptors. Cell. Mol. Life Sci. 63, 2089–2094 (2006).
Kabelitz, D. Human γδ T cells: From a neglected lymphocyte population to cellular immunotherapy: A personal reflection of 30years of γδ T cell research. Clin. Immunol. 172, 90–97 (2016).
Dimova, T. et al. Effector Vγ9Vδ2 T cells dominate the human fetal γδ T-cell repertoire. Proc. Natl. Acad. Sci. USA 112, E556–E565 (2015).
Cairo, C. et al. Impact of age, gender, and race on circulating γδ T cells. Hum. Immunol. 71, 968–975 (2010).
van der Harst, D. et al. Selective outgrowth of CD45RO+ Vγ9+/Vδ2+ T-cell receptor γ/δ T cells early after bone marrow transplantation. Blood 78, 1875–1881 (1991).
Villers, D. et al. Alteration of the T cell repertoire after bone marrow transplantation. Bone Marrow Transplant. 13, 19–26 (1994).
Kashani, E. et al. A clonotypic Vγ4Jγ1/Vδ5Dδ2Jδ1 innate γδ T-cell population restricted to the CCR6+CD27− subset. Nat. Commun. 6, 6477 (2015).
Wang, H., Fang, Z. & Morita, C.T. Vγ2Vδ2 T cell receptor recognition of prenyl pyrophosphates is dependent on all CDRs. J. Immunol. 184, 6209–6222 (2010).
Pauza, C.D. & Cairo, C. Evolution and function of the TCR Vγ9 chain repertoire: It's good to be public. Cell. Immunol. 296, 22–30 (2015).
Parker, C.M. et al. Evidence for extrathymic changes in the T cell receptor γ/δ repertoire. J. Exp. Med. 171, 1597–1612 (1990).
Caccamo, N., Dieli, F., Wesch, D., Jomaa, H. & Eberl, M. Sex-specific phenotypical and functional differences in peripheral human Vγ9/Vδ2 T cells. J. Leukoc. Biol. 79, 663–666 (2006).
Evans, P.S. et al. In vitro stimulation with a non-peptidic alkylphosphate expands cells expressing Vγ2-Jγ1.2/Vδ2 T-cell receptors. Immunology 104, 19–27 (2001).
Hviid, L. et al. High frequency of circulating γ δ T cells with dominance of the vδ1 subset in a healthy population. Int. Immunol. 12, 797–805 (2000).
Hinz, T. et al. T cell receptor γδ repertoire in HIV-1-infected individuals. Eur. J. Immunol. 24, 3044–3049 (1994).
Chaudhry, S., Cairo, C., Venturi, V. & Pauza, C.D. The γδ T-cell receptor repertoire is reconstituted in HIV patients after prolonged antiretroviral therapy. AIDS 27, 1557–1562 (2013).
Lang, P. et al. Improved immune recovery after transplantation of TCRαβ/CD19-depleted allografts from haploidentical donors in pediatric patients. Bone Marrow Transplant. 50, S6–S10 (2015).
Wherry, E.J. & Kurachi, M. Molecular and cellular insights into T cell exhaustion. Nat. Rev. Immunol. 15, 486–499 (2015).
Halary, F. et al. Shared reactivity of Vδ2neg γδ T cells against cytomegalovirus-infected cells and tumor intestinal epithelial cells. J. Exp. Med. 201, 1567–1578 (2005).
Knight, A. et al. CMV-independent lysis of glioblastoma by ex vivo expanded/activated Vδ1+ γδ T cells. PLoS One 8, e68729 (2013).
Prinz, I. et al. Donor Vδ1+ γδ T cells expand after allogeneic hematopoietic stem cell transplantation and show reactivity against CMV-infected cells but not against progressing B-CLL. Exp. Hematol. Oncol. 2, 14 (2013).
Willcox, C.R. et al. Cytomegalovirus and tumor stress surveillance by binding of a human γδ T cell antigen receptor to endothelial protein C receptor. Nat. Immunol. 13, 872–879 (2012).
Vermijlen, D. et al. Human cytomegalovirus elicits fetal γδ T cell responses in utero. J. Exp. Med. 207, 807–821 (2010).
Sell, S. et al. Control of murine cytomegalovirus infection by γδ T cells. PLoS Pathog. 11, e1004481 (2015).
Khairallah, C. γδ T cells confer protection against murine cytomegalovirus (MCMV). PLoS Pathog. 11, e1004702 (2015).
Alamyar, E., Duroux, P., Lefranc, M.P. & Giudicelli, V. IMGT((R)) tools for the nucleotide analysis of immunoglobulin (IG) and T cell receptor (TR) V-(D)-J repertoires, polymorphisms, and IG mutations: IMGT/V-QUEST and IMGT/HighV-QUEST for NGS. Methods Mol. Biol. 882, 569–604 (2012).
Nazarov, V.I. et al. tcR: an R package for T cell receptor repertoire advanced data analysis. BMC Bioinformatics 16, 175 (2015).
Acknowledgements
We thank E. Hage and T. Schulz for assistance from the central project Z1, an NGS core facility of Collaborative Research Centre SFB900; C. Struckmann and M. Ballmaier for technical guidance and single-cell sorting; the Hannover Unified Biobank of Hannover Medical School; A. Krueger for reading and criticizing the manuscript; and J. Blume for help in cord-blood preparation. Supported by Deutsche Forschungsgemeinschaft, (SFB900/B8 to C.K. and I.P.), Deutsche José Carreras Leukämie-Stiftung e.V. (DJCLS R12/29 to C.K. and I.P.) and PR727/4-1 to I.P.) and the German Federal Ministry of Education and Research (01EO1302 to C.S.-F., C.K. and I.P.).
Author information
Authors and Affiliations
Contributions
Sa.R. wrote the manuscript; Sa.R., C.S.-F. and So.R. designed and performed experiments, discussed and analyzed data; I.S., A.R., I.R. and M.B. helped with performing experiments and data analysis; M.D. and L.O. organized, acquired and processed clinical samples; R.G., M.H. and F.T. helped supervise NGS; C.v.K. and A.G. helped supervise clinical sample acquisition; R.F. helped supervise research; C.K. supervised research and the clinical study, and discussed and analyzed data; and I.P. supervised research, discussed and analyzed data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 NGS strategy for human γδ TCR repertoires.
(a) NGS strategy to analyze human TRG and TRD repertoires. Amplicons were generated from sorted γδ T cells by mRNA/cDNA based multiplex PCR technology. Multiplex primer sets target Vγ or Vδ and constant gene segments to amplify CDR3 regions followed by Illumina MiSeq sequencing. Illumina sequencing adapters are added as overhangs (red). Obtained sequences were annotated by IMGT as described in the online methods section before downstream bioinformatics analysis. (b) Validation of reproducibility. PBMCs of the same person were isolated and independently processed for NGS. Dot plots represent clonotype frequencies of multiplex PCR replica. (c) Validation of multiplex PCR. PBMCs of the same person were isolated and independently processed for either multiplex (y-axis) or 5’RACE-based (x-axis) amplicon generation strategies.
Supplementary Figure 2 Human γδ TCR repertoires are highly diverse.
(a) Graphs demonstrate numbers and median of abundant clones with a frequency of > 1% within each adult healthy control and cord blood sample of TRG and TRD repertoires. (b) Average frequencies and median of abundant clones (>1%) was determined for each healthy control and cord blood sample. (c) Shannon indices were calculated for TRG and TRD repertoires of healthy adult controls, patients and cord blood. For direct comparison, samples were normalized to 20,000 random productive rearrangements. Horizontal lines display medians. Statistical analysis was performed by paired t test (for TRG and TRD repertoire comparisons) and one-way ANOVA (between unrelated samples). * p ≤ 0.1; ** p ≤ 0.01; *** p ≤ 0.001; **** p < 0.0001. (d) Vγ9+ and Vγ9− TRG as well as Vδ2+ and Vδ1+ TRD repertoires were separately analyzed to calculate Shannon diversity indices of all healthy controls and cord blood samples. Samples were normalized to 2000 random productive reads.
Supplementary Figure 3 Quick reconstitution of γδ T cells after alloHSCT.
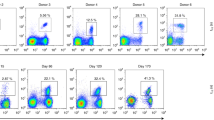
(a,b) Graphs show total numbers of (a) total gd T cells or (b) Vγ9+ cells per μl blood of healthy adult controls and patients before and up to 180 days after alloHSCT as determined by flow cytometry. Horizontal lines represent median values.
Supplementary Figure 4 Dynamics of the gd TCR repertoire in alloHSCT patients without and with reactivation of CMV.
(a,b) Stacked area graphs illustrate proportions of Top20 clones before and after alloHSCT in (a) four patients without and (b) four patients with CMV reactivation, while all other non-Top20 clones are summarized in light grey (related to Fig. 4a,b and 5a,b of the main manuscript). Expanded Top20 Vγ9+ and Vδ2+ sequences are highlighted in blue colors, Vγ9− and Vδ2− sequences are highlighted in orange colors. The day of transplantation (Tx) was set to zero to reflect T cell depletion, but no blood was drawn at Tx. Sequences appearing in the Top20 before, but not after Tx, are coded in gray shades. Arrows in (b) indicate the day of CMV-reactivation. Indicated pre-transplant repertoires could not be determined (nd) due to sample quality.
Supplementary Figure 5 Development of gd TCR repertoires after alloHSCT.
(a) NGS analysis of patients without CMV reactivation before and after alloHSCT. Box plots display median and range of Vγ2, Vγ3, Vγ4, Vγ5, Vγ8 and Vγ9 as well as Vγ1, Vδ2, Vδ3 and Vδ5 chains. (b) Shannon diversity calculation of all TRG and TRD repertoires from patients before and up to 180 days after alloHSCT. Samples were randomly sampled to 20,000 productive rearrangements. Vertical lines show medians. (c) TRG repertoires of all cord blood, healthy controls and patients before and up to 180 days after alloHSCT were separated into Vγ9+ clones to calculate frequencies of Vγ9JP+ clones. Horizontal lines represent median values.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 and Supplementary Tables 1 and 2 (PDF 1267 kb)
Rights and permissions
About this article
Cite this article
Ravens, S., Schultze-Florey, C., Raha, S. et al. Human γδ T cells are quickly reconstituted after stem-cell transplantation and show adaptive clonal expansion in response to viral infection. Nat Immunol 18, 393–401 (2017). https://doi.org/10.1038/ni.3686
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.3686
This article is cited by
-
Cytomegalovirus drives Vδ1+ γδ T cell expansion and clonality in common variable immunodeficiency
Nature Communications (2024)
-
Expansion of memory Vδ2 T cells following SARS-CoV-2 vaccination revealed by temporal single-cell transcriptomics
npj Vaccines (2024)
-
Mucosal T-cell responses to chronic viral infections: Implications for vaccine design
Cellular & Molecular Immunology (2024)
-
Unsynchronized butyrophilin molecules dictate cancer cell evasion of Vγ9Vδ2 T-cell killing
Cellular & Molecular Immunology (2024)
-
γδ T cells: origin and fate, subsets, diseases and immunotherapy
Signal Transduction and Targeted Therapy (2023)