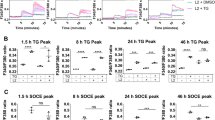
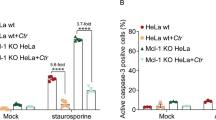
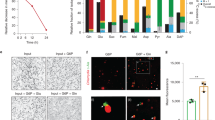
Abstract
The obligate intracellular bacterium Chlamydia trachomatis survives and replicates within a membrane-bound vacuole, termed the inclusion, which intercepts host exocytic pathways to obtain nutrients1,2,3. Like many other intracellular pathogens, C. trachomatis has a marked requirement for host cell lipids, such as sphingolipids and cholesterol, produced in the endoplasmic reticulum and the Golgi apparatus4,5,6. However, the mechanisms by which intracellular pathogens acquire host cell lipids are not well understood1,2,3. In particular, no host cell protein responsible for transporting Golgi-derived lipids to the chlamydial inclusions has yet been identified. Here we show that Chlamydia infection in human epithelial cells induces Golgi fragmentation to generate Golgi ministacks surrounding the bacterial inclusion. Ministack formation is triggered by the proteolytic cleavage of the Golgi matrix protein golgin-84. Inhibition of golgin-84 truncation prevents Golgi fragmentation, causing a block in lipid acquisition and maturation of C. trachomatis. Golgi fragmentation by means of RNA-interference-mediated knockdown of distinct Golgi matrix proteins before infection enhances bacterial maturation. Our data functionally connect bacteria-induced golgin-84 cleavage, Golgi ministack formation, lipid acquisition and intracellular pathogen growth. We show that C. trachomatis subverts the structure and function of an entire host cell organelle for its own advantage.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Carabeo, R. A., Mead, D. J. & Hackstadt, T. Golgi-dependent transport of cholesterol to the Chlamydia trachomatis inclusion. Proc. Natl Acad. Sci. USA 100, 6771–6776 (2003)
Hackstadt, T., Rockey, D. D., Heinzen, R. A. & Scidmore, M. A. Chlamydia trachomatis interrupts an exocytic pathway to acquire endogenously synthesized sphingomyelin in transit from the Golgi apparatus to the plasma membrane. EMBO J. 15, 964–977 (1996)
Scidmore, M. A., Fischer, E. R. & Hackstadt, T. Sphingolipids and glycoproteins are differentially trafficked to the Chlamydia trachomatis inclusion. J. Cell Biol. 134, 363–374 (1996)
Hatch, G. M. & McClarty, G. Phospholipid composition of purified Chlamydia trachomatis mimics that of the eucaryotic host cell. Infect. Immun. 66, 3727–3735 (1998)
van Ooij, C. et al. Host cell-derived sphingolipids are required for the intracellular growth of Chlamydia trachomatis . Cell. Microbiol. 2, 627–637 (2000)
Wylie, J. L., Hatch, G. M. & McClarty, G. Host cell phospholipids are trafficked to and then modified by Chlamydia trachomatis . J. Bacteriol. 179, 7233–7242 (1997)
De Matteis, M. A. & Luini, A. Exiting the Golgi complex. Nature Rev. Mol. Cell Biol. 9, 273–284 (2008)
Shorter, J. & Warren, G. Golgi architecture and inheritance. Annu. Rev. Cell Dev. Biol. 18, 379–420 (2002)
Short, B., Haas, A. & Barr, F. A. Golgins and GTPases, giving identity and structure to the Golgi apparatus. Biochim. Biophys. Acta 1744, 383–395 (2005)
Altan-Bonnet, N. et al. Golgi inheritance in mammalian cells is mediated through endoplasmic reticulum export activities. Mol. Biol. Cell 17, 990–1005 (2006)
Diao, A. et al. The coiled-coil membrane protein golgin-84 is a novel rab effector required for Golgi ribbon formation. J. Cell Biol. 160, 201–212 (2003)
Lowe, M. et al. Cdc2 kinase directly phosphorylates the cis-Golgi matrix protein GM130 and is required for Golgi fragmentation in mitosis. Cell 94, 783–793 (1998)
Wang, Y. et al. A direct role for GRASP65 as a mitotically regulated Golgi stacking factor. EMBO J. 22, 3279–3290 (2003)
Chiu, R., Novikov, L., Mukherjee, S. & Shields, D. A caspase cleavage fragment of p115 induces fragmentation of the Golgi apparatus and apoptosis. J. Cell Biol. 159, 637–648 (2002)
Lowe, M., Lane, J. D., Woodman, P. G. & Allan, V. J. Caspase-mediated cleavage of syntaxin 5 and giantin accompanies inhibition of secretory traffic during apoptosis. J. Cell Sci. 117, 1139–1150 (2004)
Mancini, M. et al. Caspase-2 is localized at the Golgi complex and cleaves golgin-160 during apoptosis. J. Cell Biol. 149, 603–612 (2000)
Rajalingam, K. et al. Epithelial cells infected with Chlamydophila pneumoniae (Chlamydia pneumoniae) are resistant to apoptosis. Infect. Immun. 69, 7880–7888 (2001)
Fan, T. et al. Inhibition of apoptosis in chlamydia-infected cells: blockade of mitochondrial cytochrome c release and caspase activation. J. Exp. Med. 187, 487–496 (1998)
Hood, J. L., Brooks, W. H. & Roszman, T. L. Differential compartmentalization of the calpain/calpastatin network with the endoplasmic reticulum and Golgi apparatus. J. Biol. Chem. 279, 43126–43135 (2004)
Goll, D. E. et al. The calpain system. Physiol. Rev. 83, 731–801 (2003)
Suzuki, K. & Sorimachi, H. A novel aspect of calpain activation. FEBS Lett. 433, 1–4 (1998)
Puthenveedu, M. A. et al. GM130 and GRASP65-dependent lateral cisternal fusion allows uniform Golgi-enzyme distribution. Nature Cell Biol. 8, 238–248 (2006)
Holthuis, J. C. & Levine, T. P. Lipid traffic: floppy drives and a superhighway. Nature Rev. Mol. Cell Biol. 6, 209–220 (2005)
Marie, M., Sannerud, R., Avsnes Dale, H. & Saraste, J. Membrane traffic in the secretory pathway: Take the 'A' train: on fast tracks to the cell surface. Cell. Mol. Life Sci. 65, 2859–2874 (2008)
Machuy, N. et al. A global approach combining proteome analysis and phenotypic screening with RNA interference yields novel apoptosis regulators. Mol. Cell. Proteomics 4, 44–55 (2005)
Heuer, D., Brinkmann, V., Meyer, T. F. & Szczepek, A. J. Expression and translocation of chlamydial protease during acute and persistent infection of the epithelial HEp-2 cells with Chlamydophila (Chlamydia) pneumoniae . Cell. Microbiol. 5, 315–322 (2003)
Acknowledgements
The authors would like to thank A. Greiser, C. Goosmann, B. Laube, M. Wicht and E. Ziska for technical support; M. A. De Matteis for the gift of the GFP–GM130 fusion plasmid; H. P. Hauri for the provision of the anti-GPP130 antibody and helpful discussions; and K. Astrahantseff, S. and J. Heuer, T. Wolff and L. Ogilvie for critically reading the manuscript and helpful suggestions. This work was financially supported by the Senate of Berlin and the BMBF through the RiNA Network.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-17 with Legends and Supplementary Table 1 which summarizes truncated golgin-84 expressing cell line, plasmids and primers. (PDF 2543 kb)
Supplementary Movie 1
Supplementary Movie 1 shows GM130-GFP expressing cells infected with C.trachomatis to reveal GA disruption during the infection period. The time lapse covers about 12 h. By following cells during the course of the movie, it is evident that the GA is dispersed during infection. (MOV 3373 kb)
Supplementary Movie 2
Supplementary Movie 2 shows ceramide trafficking in infected cells. BODIPY FL C5-ceramide transport into the inclusion. Time-lapse images of HeLa cells infected with C. trachomatis (MOI = 2) for 26 h. Labelled ceramide was added 1 to 2 minutes after starting image acquisition. The assigned numbers represent the time elapsed. (MOV 2259 kb)
Supplementary Movie 3
Supplementary Movie 3 shows ceramide trafficking in infected cells treated with Z-WEHD-FMK. BODIPY FL C5-ceramide transport into the inclusion is inhibited by treatment with Z-WEHD-FMK. Time-lapse images of HeLa cells infected with C. trachomatis (MOI = 2) treated with inhibitor 9 h p.i. Image acquisition was started at 27 h p.i. and labelled ceramide was added 1 to 2 minutes after starting the acquisition. The assigned numbers represent the time elapsed. (MOV 2566 kb)
Rights and permissions
About this article
Cite this article
Heuer, D., Lipinski, A., Machuy, N. et al. Chlamydia causes fragmentation of the Golgi compartment to ensure reproduction. Nature 457, 731–735 (2009). https://doi.org/10.1038/nature07578
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07578
This article is cited by
-
Intracellular lifestyle of Chlamydia trachomatis and host–pathogen interactions
Nature Reviews Microbiology (2023)
-
Surviving the host–pathogen interaction: Legionella uses its SidE arsenal to mediate phosphoribosyl-linked ubiquitination of Golgi proteins inducing organelle fragmentation
Cell Death & Differentiation (2021)
-
Outer membrane vesicles containing OmpA induce mitochondrial fragmentation to promote pathogenesis of Acinetobacter baumannii
Scientific Reports (2021)
-
Serine-ubiquitination regulates Golgi morphology and the secretory pathway upon Legionella infection
Cell Death & Differentiation (2021)
-
A predation assay using amoebae to screen for virulence factors unearthed the first W. chondrophila inclusion membrane protein
Scientific Reports (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.