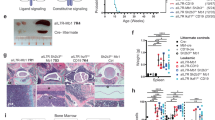
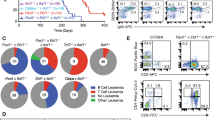
Abstract
Expression of c-Myb is required for normal hematopoiesis and for proliferation of myeloid leukemia blasts and a subset of T-cell leukemia, but its role in B-cell leukemogenesis is unknown. We tested the role of c-Myb in p190BCR/ABL-dependent B-cell leukemia in mice transplanted with p190BCR/ABL-transduced marrow cells with a c-Myb allele (Mybf/d) and in double transgenic p190BCR/ABL/Mybw/d mice. In both models, loss of a c-Myb allele caused a less aggressive B-cell leukemia. In p190BCR/ABL-expressing human B-cell leukemia lines, knockdown of c-Myb expression suppressed proliferation and colony formation. Compared with c-Mybw/f cells, expression of Bmi1, a regulator of stem cell proliferation and maintenance, was decreased in pre-B cells from Mybw/d p190BCR/ABL transgenic mice. Ectopic expression of a mutant c-Myb or Bmi1 enhanced the proliferation and colony formation of Mybw/d p190BCR/ABL B-cells; by contrast, Bmi1 downregulation inhibited colony formation of p190BCR/ABL-expressing murine B cells and human B-cell leukemia lines. Moreover, c-Myb interacted with a segment of the human Bmi1 promoter and enhanced its activity. In blasts from 19 Ph1 adult acute lymphoblastic leukemia patients, levels of c-Myb and Bmi1 showed a positive correlation. Together, these findings support the existence of a c-Myb–Bmi1 transcription-regulatory pathway required for p190BCR/ABL leukemogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Gleissner B, Gokbuget N, Bartram CR, Janssen B, Rieder H, Janssen JW et al. Leading prognostic relevance of the BCR-ABL translocation in adult acute B-lineage lymphoblastic leukemia: a prospective study of the German multicenter Trial Group and confirmed polymerase chain reaction analysis. Blood 2002; 99: 1536–1543.
Melo JV . The diversity of BCR-ABL fusion proteins and their relationship to leukemia phenotype. Blood 1996; 88: 2375–2384.
Clark SS, McLaughlin J, Timmons M, Pendergast AM, Ben-Neriah Y, Dow LW et al. Expression of a distinctive BCR-ABL oncogene in Ph1-positive acute lymphocytic leukemia (ALL). Science 1988; 239: 775–777.
Lugo TG, Pendergast AM, Muller AJ, Witte ON . Tyrosine kinase activity and transformation potency of bcr-abl oncogene products. Science 1990; 247: 1079–1082.
Goga A, McLaughlin J, Afar DE, Saffran DC, Witte ON . Alternative signals to RAS for hematopoietic transformation by the BCR-ABL oncogene. Cell 1995; 82: 981–988.
Shuai K, Halpern J, ten Hoeve J, Rao X, Sawyers CL . Constitutive activation of STAT5 by the BCR-ABL oncogene in chronic myelogenous leukemia. Oncogene 1996; 13: 247–254.
Skorski T, Bellacosa A, Nieborowska-Skorska M, Majewski M, Martinez R, Choi JK et al. Transformation of hematopoietic cells by BCR/ABL requires activation of a PI-3k/Akt-dependent pathway. EMBO J 1997; 16: 6151–6161.
Okuda K, Golub TR, Gilliland DG, Griffin JD . p210BCR/ABL, p190BCR/ABL, and TEL/ABL activate similar signal transduction pathways in hematopoietic cell lines. Oncogene 1996; 13: 1147–1152.
Ilaria RL, Van Etten RA . P210 and P190(BCR/ABL) induce the tyrosine phosphorylation and DNA binding activity of multiple specific STAT family members. J Biol Chem 1996; 271: 31704–31710.
Li S, Ilaria RL, Million RP, Daley GQ, Van Etten RA . The P190, P210, and P230 forms of the BCR/ABL oncogene induce a similar chronic myeloid leukemia-like syndrome in mice but have different lymphoid leukemogenic activity. J Exp Med 1999; 189: 1399–1412.
Hu Y, Liu Y, Pelletier S, Buchdunger E, Warmuth M, Fabbro D et al. Requirement of Src kinases Lyn, Hck and Fgr for BCR-ABL1-induced B-lymphoblastic leukemia but not chronic myeloid leukemia. Nat Genet 2004; 36: 453–461.
Xie S, Lin H, Sun T, Arlinghaus RB . Jak2 is involved in c-Myc induction by Bcr-Abl. Oncogene 2002; 21: 7137–7146.
Jamieson CH, Ailles LE, Dylla SJ, Muijtjens M, Jones C, Zehnder JL et al. Granulocyte–macrophage progenitors as candidate leukemic stem cells in blast-crisis CML. N Engl J Med 2004; 351: 657–667.
Zhao C, Blum J, Chen A, Kwon HY, Jung SH, Cook JM et al. Loss of beta-catenin impairs the renewal of normal and CML stem cells in vivo. Cancer Cell 2007; 12: 528–541.
Zhao C, Chen A, Jamieson CH, Fereshteh M, Abrahamsson A, Blum J et al. Hedgehog signalling is essential for maintenance of cancer stem cells in myeloid leukaemia. Nature 2009; 458: 776–779.
Perrotti D, Cesi V, Trotta R, Guerzoni C, Santilli G, Campbell K et al. BCR-ABL suppresses C/EBPalpha expression through inhibitory action of hnRNP E2. Nat Genet 2002; 30: 48–58.
Passegue E, Jochum W, Schorpp-Kistner M, Mohle-Steinlein U, Wagner EF . Chronic myeloid leukemia with increased granulocyte progenitors in mice lacking junB expression in the myeloid lineage. Cell 2001; 104: 21–32.
Yang MY, Liu TC, Chang JG, Lin PM, Lin SF . JunB gene expression is inactivated by methylation in chronic myeloid leukemia. Blood 2003; 101: 3205–3211.
Corradini F, Cesi V, Bartella V, Pani E, Bussolari R, Candini O, Calabretta B . Enhanced proliferative potential of hematopoietic cells expressing degradation-resistant Myb mutants. J Biol Chem 2005; 280: 30254–30262.
Calabretta B, Sims RB, Valtieri M, Caracciolo D, Szczylik C, Venturelli D et al. Normal and leukemic hematopoietic cells manifest differential sensitivity to inhibitory effects of Myb antisense oligodeoxynucleotides: an in vitro study relevant to bone marrow purging. Proc Natl Acad Sci USA 1991; 88: 2351–2355.
Ratajczak MZ, Hijiya N, Catani LK, DeRiel K, Luger SM, McGlave P et al. Acute- and chronic-phase chronic myelogenous leukemia colony-forming units are highly sensitive to the growth inhibitory effects of Myb antisense oligodeoxynucleotides. Blood 1992; 79: 1956–1961.
Lidonnici MR, Corradini F, Waldron T, Bender TP, Calabretta B . Requirement of Myb for p210(BCR/ABL)-dependent transformation of hematopoietic progenitors and leukemogenesis. Blood 2008; 111: 4771–4779.
Lahortiga I, De Keersmaecker K, Van Vlierberghe PC, Graux C, Cauwelier B, Lambert F et al. Duplication of the MYB oncogene in T cell acute lymphoblastic leukemia. Nat Genet 2007; 39: 593–595.
Clappier E, Cuccuini W, Kalota A, Crinquette A, Cayeuela JM, Dik WA et al. The MYB locus is involved in chromosomal translocation and genomic duplications in human T-cell acute leukemia (T-ALL), the translocation defining a new T-ALL subtype in very young children. Blood 2007; 110: 1251–1261.
Lessard J, Sauvageau G . Bmi1 determines the proliferative capacity of normal and leukaemic stem cells. Nature 2003; 423: 255–260.
Thomas MD, Kremer CS, Ravichandran KS, Rajewsky K, Bender TP . Myb is critical for B cell development and maintenance of follicular B cells. Immunity 2005; 23: 275–286.
Heisterkamp N, Jenster G, Ten Hoeve J, Zovich D, Pattengale PK, Groffen J . Acute leukaemia in bcr/abl transgenic mice. Nature 1990; 344: 251–253.
Xiao C, Calado DP, Galler G, Thai TH, Patterson HC, Wang J et al. MiR-150 controls B cell differentiation by targeting the transcription factor Myb. Cell 2007; 131: 146–159.
Alkema MJ, Jacobs H, van Lohuizen M, Berns A . Perturbation of B and T cell development and predisposition to lymphomagenesis in Emu Bmi1 transgenic mice require the Bmi1 RING finger. Oncogene 1997; 15: 899–910.
Jacobs JJ, Scheijen B, Voncken JW, Kieboom K, Berns A, van Lohuizen M . Bmi1 collaborates with c-Myc in tumorigenesis by inhibiting c-Myc-induced apoptosis via INK4a/ARF. Genes Dev 1999; 13: 2678–2690.
Guo WJ, Datta S, Band V, Dimri GP . Mel-18, a polycomb group protein, regulates cell proliferation and senescence via transcriptional repression of Bmi1 and c-Myc oncoproteins. Mol Biol Cell 2007; 18: 536–546.
Evans JL, Moore TL, Kuehl WM, Bender T, Ting JP . Functional analysis of Myb protein in T-lymphocytic cell lines shows that it transactivates the c-myc promoter. Mol Cell Biol 1990; 10: 5747–5752.
Estrov Z, Talpaz M, Zipf TF, Kantarjian HM, Ku S, Ouspenskaia MV et al. Role of granulocyte–macrophage colony-stimulating factor in Philadelphia (Ph1)-positive acute lymphoblastic leukemia: studies on two newly established Ph1-positive acute lymphoblastic leukemia cell lines (Z-119 and Z-181). J Cell Physiol 1996; 166: 618–630.
Mavilio F, Sposi NM, Petrini M, Bottero L, Matinucci M, De Rossi G et al. Expression of cellular oncogenes in primary cells from human acute leukemias. Proc Natl Acad Sci USA 1986; 83: 4394–4398.
Armstrong SA, Staunton JE, Silverman LB, Pieters R, den Boer ML, Minden MD et al. MLL translocations specify a distinct gene expression profile that distinguishes a unique leukemia. Nat Genet 2002; 30: 41–47.
Rizo A, Olthof S, Han L, Vellenga E, de Haan G, Schuringa JJ . Repression of BMI1 in normal and leukemic human CD34(+) cells impairs self-renewal and induces apoptosis. Blood 2009; 114: 1498–1505.
Haupt Y, Bath ML, Harris AW, Adams JM . Bmi1 transgene induces lymphomas and collaborates with Myc in tumorigenesis. Oncogene 1993; 8: 3161–3164.
Warner JK, Wang JC, Takenaka K, Doulatov S, McKenzie JL, Harrington L et al. Direct evidence for cooperating genetic events in the leukemic transformation of normal human hematopoietic cells. Leukemia 2005; 19: 1794–1805.
Datta S, Hoenerhoff MJ, Bommi P, Sainger R, Guo WJ, Dimri M et al. Bmi1 cooperates with H-Ras to transform human mammary epithelial cells via dysregulation of multiple growth-regulatory pathways. Cancer Res 2007; 67: 10286–10295.
Acknowledgements
We thank Dr Dimri (Department of Cancer Biology, Evanston Northwestern Healthcare Research Institute, Evanston, IL, USA) for the Bmi1-Luc plasmid and Dr Sankar Addya (Kimmel Cancer Center, Thomas Jefferson University, Philadelphia, PA, USA) for analysis of microarray data. This work was supported by National Cancer Institute Grants CA95111 and P0178890 (BC); and European LeukemiaNet, AIRC, AIL and Fondazione Del Monte di Bologna e Ravenna (GM and II); and NIAID Grant AI059294 (TPB).ARS was supported by a fellowship of the American-Italian Cancer Foundation (AICF) and is currently supported by a fellowship from Associazione Italiana Ricerca sul Cancro (AIRC). TW was supported by a National Institutes of Health pre-doctoral fellowship (T32-CA09683-14).
Author contributions
T Waldron performed most experiments and wrote the first draft of the manuscript. M De Dominici performed the experiments with human ALL lines. AR Soliera performed the luciferase and ChIP assays on the Bmi-1 promoter. I Iacobucci, A Leonetti and G Martinelli performed the real-time PCR studies on the ALL samples. Y Zhang and R Martinez performed microarray hybridization analyses. T Hyslop performed statistical analyses. TP Bender provided Mybf/d mice and help for the immunophenotype analyses. B Calabretta designed the experiments and wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
This work is dedicated to the memory of Dr Alan M Gewirtz.
Supplementary Information accompanies the paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Waldron, T., De Dominici, M., Soliera, A. et al. c-Myb and its target Bmi1 are required for p190BCR/ABL leukemogenesis in mouse and human cells. Leukemia 26, 644–653 (2012). https://doi.org/10.1038/leu.2011.264
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2011.264
Keywords
This article is cited by
-
c-Myb regulates tumorigenic potential of embryonal rhabdomyosarcoma cells
Scientific Reports (2019)
-
Targeting acute myeloid leukemia by drug-induced c-MYB degradation
Leukemia (2018)
-
Molecular processes involved in B cell acute lymphoblastic leukaemia
Cellular and Molecular Life Sciences (2018)
-
Intestinal-specific activatable Myb initiates colon tumorigenesis in mice
Oncogene (2016)
-
CDKN2A-independent role of BMI1 in promoting growth and survival of Ph+ acute lymphoblastic leukemia
Leukemia (2016)