Abstract
Background/Objectives:
To study the bioavailability of anthocyanins and the effects of a 20% blackcurrant juice drink on vascular reactivity, plasma antioxidant status and other cardiovascular disease risk markers.
Subjects/Methods:
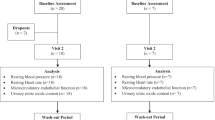
The study was a randomised, cross-over, double-blind, placebo-controlled acute meal study. Twenty healthy volunteers (11 females and 9 males) were recruited, and all subjects completed the study. Fasted volunteers consumed a 20% blackcurrant juice drink (250 ml) or a control drink following a low-flavonoid diet for the previous 72 h. Vascular reactivity was assessed at baseline and 120 min after juice consumption by laser Doppler imaging (LDI). Plasma and urine samples were collected periodically over an 8-h period for analysis, with a final urine sample collected at 24 h. The cross-over was performed after a 4-week washout.
Results:
There were no significant effects of the 20% blackcurrant juice drink on acute measures of vascular reactivity, biomarkers of endothelial function or lipid risk factors. Consumption of the test juice caused increases in plasma vitamin C (P=0.006), and urinary anthocyanins (P<0.001). Delphinidin-3-rutinoside and cyanidin-3-rutinoside were the main anthocyanins excreted in urine with delphinidin-3-glucoside also detected. The yield of anthocyanins in urine was 0.021±0.003% of the dietary intake of delphinidin glycosides and 0.009±0.002% of the dietary intake of cyanidin glycosides.
Conclusions:
The juice consumption did not have a significant effect on vascular reactivity. Anthocyanins were present at low concentrations in the urine, and microbial metabolites of flavonoids were detected in plasma after juice consumption.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Bub A, Watzl B, Abrahamse L, Delincee H, Adam S, Wever J et al. (2000). Moderate intervention with carotenoid-rich vegetable products reduces lipid peroxidation in men. J Nutr 130, 2200–2206.
Cao G, Russell RM, Lischner N, Prior RL (1998). Serum antioxidant capacity is increased by consumption of strawberries, spinach, red wine or vitamin C in elderly women. J Nutr 128, 2383–2390.
Cao G, Prior RL (1999). Measurement of oxygen radical absorbance capacity in biological samples. Methods Enzymol 299, 50–62.
Chambers JC, McGregor A, Jean-Marie J, Obeid OA, Kooner JS (1999). Demonstration of rapid onset vascular endothelial dysfunction after hyperhomocysteinemia: an effect reversible with vitamin C therapy. Circulation 99, 1156–1160.
Dalgard C, Nielsen F, Morrow JD, Enghusen-Poulsen H, Jonung T, Horder M et al. (2009). Supplementation with orange and blackcurrant juice, but not vitamin E, improves inflammatory markers in patients with peripheral arterial disease. Br J Nutr 101, 263–269.
Felgines C, Talavera S, Gonthier MP, Texier O, Scalbert A, Lamaison JL et al. (2003). Strawberry anthocyanins are recovered in urine as glucuro- and sulfoconjugates in humans. J Nutr 133, 1296–1301.
Felgines C, Talavera S, Texier O, Gil-Izquierdo A, Lamaison JL, Remesy C (2005). Blackberry anthocyanins are mainly recovered from urine as methylated and glucuronidated conjugates in humans. J Agric Food Chem 53, 7721–7727.
Fitzpatrick DF, Fleming RC, Bing B, Maggi DA, O′Malley RM (2000). Isolation and characterization of endothelium-dependent vasorelaxing compounds from grape seeds. J Agric Food Chem 48, 6384–6390.
Fleschhut J, Kratzer F, Rechkemmer G, Kulling SE (2006). Stability and biotransformation of various dietary anthocyanins in vitro. Eur J Nutr 45, 7–18.
Frei B, Stocker R, England L, Ames BN (1990). Ascorbate: the most effective antioxidant in human blood plasma. Adv Exp Med Biol 264, 155–163.
George TW, Niwat C, Waroonphan S, Gordon MH, Lovegrove JA (2009). Effects of chronic and acute consumption of fruit- and vegetable-puree-based drinks on vasodilation, risk factors for CVD and the response as a result of the eNOS G298T polymorphism. Proc Nutr Soc 68, 148–161.
Glantzounis GK, Tsimoyiannis EC, Kappas AM, Galaris DA (2005). Uric acid and oxidative stress. Curr Pharm Des 11, 4145–4151.
Gokce N, Keaney Jr JF, Frei B, Holbrook M, Olesiak M, Zachariah BJ et al. (1999). Long-term ascorbic acid administration reverses endothelial vasomotor dysfunction in patients with coronary artery disease. Circulation 99, 3234–3240.
Hertog MG, Kromhout D, Aravanis C, Blackburn H, Buzina R, Fidanza F et al. (1995). Flavonoid intake and long-term risk of coronary heart disease and cancer in the seven countries study. Arch Intern Med 155, 381–386.
Hollands W, Brett GM, Radreau P, Saha S, Teucher B, Bennett RN et al. (2008). Processing blackcurrants dramatically reduces the content and does not enhance the urinary yield of anthocyanins in human subjects. Food Chem 108, 869–878.
Hornig B, Arakawa N, Kohler C, Drexler H (1998). Vitamin C improves endothelial function of conduit arteries in patients with chronic heart failure. Circulation 97, 363–368.
Hou DX (2003). Potential mechanisms of cancer chemoprevention by anthocyanins. Curr Mol Med 3, 149–159.
Kang SY, Seeram NP, Nair MG, Bourquin LD (2003). Tart cherry anthocyanins inhibit tumor development in Apc(Min) mice and reduce proliferation of human colon cancer cells. Cancer Lett 194, 13–19.
Kay CD, Holub BJ (2002). The effect of wild blueberry (Vaccinium angustifolium) consumption on postprandial serum antioxidant status in human subjects. Br J Nutr 88, 389–397.
Kay CD, Mazza G, Holub BJ, Wang J (2004). Anthocyanin metabolites in human urine and serum. Br J Nutr 91, 933–942.
Kay CD, Mazza GJ, Holub BJ (2005). Anthocyanins exist in the circulation primarily as metabolites in adult men. J Nutr 135, 2582–2588.
Kay CD (2006). Aspects of anthocyanin absorption, metabolism and pharmacokinetics in humans. Nutr Res Rev 19, 137–146.
Keli SO, Hertog MG, Feskens EJ, Kromhout D (1996). Dietary flavonoids, antioxidant vitamins, and incidence of stroke: the Zutphen study. Arch Intern Med 156, 637–642.
Keppler K, Humpf HU (2005). Metabolism of anthocyanins and their phenolic degradation products by the intestinal microflora. Bioorg Med Chem 13, 5195–5205.
Kim DH, Jung EA, Sohng IS, Han JA, Kim TH, Han MJ (1998). Intestinal bacterial metabolism of flavonoids and its relation to some biological activities. Arch Pharm Res 21, 17–23.
Knekt P, Jarvinen R, Reunanen A, Maatela J (1996). Flavonoid intake and coronary mortality in Finland: a cohort study. BMJ 312, 478–481.
Kugiyama K, Motoyama T, Hirashima O, Ohgushi M, Soejima H, Misumi K et al. (1998). Vitamin C attenuates abnormal vasomotor reactivity in spasm coronary arteries in patients with coronary spastic angina. J Am Coll Cardiol 32, 103–109.
Levine GN, Frei B, Koulouris SN, Gerhard MD, Keaney Jr JF, Vita JA (1996). Ascorbic acid reverses endothelial vasomotor dysfunction in patients with coronary artery disease. Circulation 93, 1107–1113.
Liau LS, Lee BL, New AL, Ong CN (1993). Determination of plasma ascorbic acid by high-performance liquid chromatography with ultraviolet and electrochemical detection. J Chromatogr 612, 63–70.
Lotito SB, Frei B (2006). Consumption of flavonoid-rich foods and increased plasma antioxidant capacity in humans: cause, consequence, or epiphenomenon? Free Radic Biol Med 41, 1727–1746.
Nakamura Y, Matsumoto H, Todoki K (2002). Endothelium-dependent vasorelaxation induced by black currant concentrate in rat thoracic aorta. Jpn J Pharmacol 89, 29–35.
Pedersen CB, Kyle J, Jenkinson AM, Gardner PT, McPhail DB, Duthie GG (2000). Effects of blueberry and cranberry juice consumption on the plasma antioxidant capacity of healthy female volunteers. Eur J Clin Nutr 54, 405–408.
Prior RL, Cao GH (2000). Flavonoids: diet and health relationships. Nutr Clin Care 3, 279–288.
Prior RL, Hoang H, Gu LW, Wu XL, Bacchiocca M, Howard L et al. (2003). Assays for hydrophilic and lipophilic antioxidant capacity (oxygen radical absorbance capacity (ORAC(FL))) of plasma and other biological and food samples. J Agric Food Chem 51, 3273–3279.
Rechner AR, Kroner C (2005). Anthocyanins and colonic metabolites of dietary polyphenols inhibit platelet function. Thromb Res 116, 327–334.
Ross R (1999). Atherosclerosis--an inflammatory disease. N Engl J Med 340, 115–126.
Samman S, Sivarajah G, Man JC, Ahmad ZI, Petocz P, Caterson ID (2003). A mixed fruit and vegetable concentrate increases plasma antioxidant vitamins and folate and lowers plasma homocysteine in men. J Nutr 133, 2188–2193.
Slimestad R, Solheim H (2002). Anthocyanins from black currants (Ribes nigrum L.). J Agric Food Chem 50, 3228–3231.
Stein JH, Keevil JG, Wiebe DA, Aeschlimann S, Folts JD (1999). Purple grape juice improves endothelial function and reduces the susceptibility of LDL cholesterol to oxidation in patients with coronary artery disease. Circulation 100, 1050–1055.
Toromanovic J, Kovac-Besovic E, Sapcanin A, Tahirovic I, Rimpapa Z, Kroyer G et al. (2008). Urinary hippuric acid after ingestion of edible fruits. Bosn J Basic Med Sci 8, 38–43.
Tsuda T, Shiga K, Ohshima K, Kawakishi S, Osawa T (1996). Inhibition of lipid peroxidation and the active oxygen radical scavenging effect of anthocyanin pigments isolated from Phaseolus vulgaris L. Biochem Pharmacol 52, 1033–1039.
Vauzour D, Houseman EJ, George TW, Corona G, Garnotel R, Jackson KG et al. (2010). Moderate Champagne consumption promotes an acute improvement in acute endothelial-independent vascular function in healthy human volunteers. B J Nutr 103, 1168–1178.
Volkovova K, Barancokova M, Kazimirova A, Collins A, Raslova K, Smolkova B et al. (2005). Antioxidant supplementation reduces inter-individual variation in markers of oxidative damage. Free Radic Res 39, 659–666.
Wu X, Cao G, Prior RL (2002). Absorption and metabolism of anthocyanins in elderly women after consumption of elderberry or blueberry. J Nutr 132, 1865–1871.
Xu JW, Ikeda K, Yamori Y (2004). Upregulation of endothelial nitric oxide synthase by cyanidin-3-glucoside, a typical anthocyanin pigment. Hypertension 44, 217–222.
Zhang K, Zuo Y (2004). GC-MS determination of flavonoids and phenolic and benzoic acids in human plasma after consumption of cranberry juice. J Agric Food Chem 52, 222–227.
Acknowledgements
We acknowledge funding from GlaxoSmithKline plc.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Jin, Y., Alimbetov, D., George, T. et al. A randomised trial to investigate the effects of acute consumption of a blackcurrant juice drink on markers of vascular reactivity and bioavailability of anthocyanins in human subjects. Eur J Clin Nutr 65, 849–856 (2011). https://doi.org/10.1038/ejcn.2011.55
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejcn.2011.55
Keywords
This article is cited by
-
Food intake biomarkers for berries and grapes
Genes & Nutrition (2020)
-
Timed consumption of a New Zealand blackcurrant juice support positive affective responses during a self-motivated moderate walking exercise in healthy sedentary adults
Journal of the International Society of Sports Nutrition (2019)
-
Phenolic compounds in fruits and beverages consumed as part of the mediterranean diet: their role in prevention of chronic diseases
Phytochemistry Reviews (2016)
-
Characterisation of the main drivers of intra- and inter- breed variability in the plasma metabolome of dogs
Metabolomics (2016)
-
Cyanidin and malvidin in aqueous extracts of black carrots fermented with Aspergillus oryzae prevent the impairment of energy, lipid and glucose metabolism in estrogen-deficient rats by AMPK activation
Genes & Nutrition (2015)