Abstract
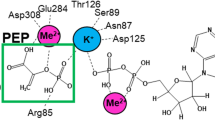
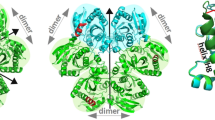
CRYSTAL structures of the high- and low-activity states of the allosteric enzyme phosphofructokinase implicate three arginines in substrate binding, catalysis and cooperativity1–5. Arginines 162 and 243 reach into the active site from an adjacent subunit and interact with the cooperative substrate fructose 6-phosphate. Mutation of these arginines to serine results in mutant enzymes with reduced substrate binding and lowered cooperativity, but with little change in their catalytic ability (kcat). Arg72 bridges the two substrates fructose 6-phosphate and ATP, and interacts with the 1-phosphate of the product fructose 1,6-bisphosphate. Mutation of this residue to serine reduces the catalytic activity, cooperativity and binding of fructose 6-phosphate and fructose 1,6-bisphosphate. In the reverse reaction, the kinetics of wild-type and the Ser 72 mutant with respect to fructose 1,6-bisphosphate are hyperbolic, whereas those of the Ser 162 and Ser 243 mutants are sigmoidal. These results show that each of the three arginines contributes to cooperativity and to the transmission of allosteric signals between the four subunits of the enzyme.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Evans, P. R. & Hudson, P. J. Nature 279, 500–504 (1979).
Evans, P. R., Farrants, G. W. & Hudson, P. J. Phil. Trans. R. Soc. B293, 53–62 (1981).
Evans, P. R., Farrants, G. W. & Lawrence, M. C. J. molec. Biol. 191, 713–720 (1986).
Shirakihara, Y. & Evans, P. J. molec. Biol. 204, 973–994 (1988).
Schirmer, T. & Evans, P. R. Nature 343, 140–145 (1990).
Blangy, D., Buc, H. & Monod, J. J. molec. Biol. 31, 13–35 (1968).
Monod, J., Wyman, J. & Changeux, J. P. J. molec. Biol. 12, 88–118 (1965).
Hellinga, H. W. & Evans, P. R. Nature 327, 437–439 (1987).
Zoller, M. J. & Smith, M. Meth. Enzym. 100, 468–500 (1983).
Lau, F. K.-T., Fersht, A. R., Hellinga, H. W. & Evans, P. R. Biochemistry 26, 4143–4148 (1987).
Daldal, F. J. molec. Biol. 168, 285–305 (1983).
Kotlarz, D. & Buc, H. Meth. Enzym. 90, 60–70 (1982).
French, B. A. & Chang, S. H. Gene 54, 65–71 (1987).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Berger, S., Evans, P. Active-site mutants altering the cooperativity of E. coliphosphofructokinase. Nature 343, 575–576 (1990). https://doi.org/10.1038/343575a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/343575a0
This article is cited by
-
When one and one are not two
Nature Structural & Molecular Biology (1995)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.