Abstract
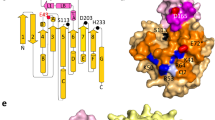
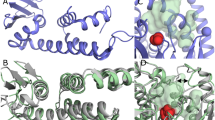
Here we report the first structure of a mammalian 15-lipoxygenase. The protein is composed of two domains; a catalytic domain and a previously unrecognized β-barrel domain. The N-terminal β-barrel domain has topological and sequence identity to a domain in the mammalian lipases, suggesting that these domains may have similar functions in vivo. Within the C-terminal domain, the lipoxygenase substrate binding site is a hydrophobic pocket defined by a bound inhibitor. Arachidonic acid can be docked into this deep hydrophobic pocket with the methyl end extending down into the bottom of the pocket and the acid end tethered by a conserved basic residue on the surface of the enzyme. This structure provides a unifying hypothesis for the positional specificity of mammalian lipoxygenases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Samuelsson, B., Dahlén, S.-E., Lingren, J.Å., Rouzer, C.A. & Serhan, C.N. Leukotrienes and Lipoxins: Structures, Biosynthesis, and Biological Effects. Science 237, 1171–1176 (1987).
Yokomizo, T., Izumi, T., Chang, K., Takuwa, Y. & Shimizu, T. A G-protein coupled receptor for leukotriene B4 that mediates chemotaxis. Nature 387, 620–624 (1997).
Devchand, P.R. et al. The PPARa-Leukotriene B4 pathway to inflammation control. Nature 384, 39–43 (1996).
Forman, B.M., Chen, J. & Evans, R.M. Hypolipidemic drugs, polyunsaturated fatty acids, and eicosanoids are ligands for peroxisome proliferator-activated receptors alpha and delta. Proc. Nat. Acad. Sci. USA 94, 4312–4317 (1997).
Israel, E., Cohn, J., Dubé, L. & Drazen, J.M. Effect of treatment with Zileuton, a 5-lipoxygenase inhibitor, in patients with asthma. J. Am. Med. Assn. 275, 931–936 (1996).
Shannon, V.R., Stenson, W.F. & Holtzman, M.J. Induction of epithelial arachidonate 12-lipoxygenase at active sites of inflammatory bowel disease. Am. J. Physiol. 264, G104–111 (1993).
Steinberg, D., Parthasarathy, S., Carew, T.E., Khoo, J.C. & Witztum, M.D. Beyond cholesterol: Modifications of low-density lipoprotein that increase its atherogenicity. N. Engl. J. Med. 320, 915–924 (1989).
Yla-Herttuala, S. et al. Gene expression in macrophage-rich human atherosclerotic lesions. 15-lipoxygenase and acetyl low density lipoprotein receptor messenger RNA colocalize with oxidation specific lipid-protein adducts. J. Clin. Invest. 87, 1146–1152 (1991).
Kühn, J., Belkner, J., Zaiss, S., Fahrenklemper, T. & Wohlfeil, S. Involvement of 15-lipoxygenase in early stages of atherosclerosis. J. Exp. Med. 179, 1903–1911 (1994).
Ford-Hutchinson, A.W. Arachidonate 15-lipoxygenase; characteristics and potential biological significance. Eicosanoids 4, 65–74 (1991).
The CCP4 Suite:Programs for protein crystallography. Acta Crystallogr. D50, 760–763 (1994).
Minor, W. et al. Crystal structure of soybean lipoxygenase-1 at 1.4Å resolution. Biochemistry 35, 10687–10701 (1996).
Boyington, J.C., Gaffney, B.J. & Amzel, L.M. The three-dimensional structure of an arachidonic acid 15-lipoxygenase. Science 260, 1482–1486 (1993).
Funk, C.D. The molecular biology of mammalian lipoxygenases and the quest for eicosanoid functions using lipoxygenase-deficient mice. Biochem. Biophys. Acta 1304, 65–84 (1996).
Prigge, S.T., Boyington, J.C., Gaffney, B.J. & Amzel, L.M. Structure conservation in lipoyxgenases: structural analysis of soybean lipoxygenase-1 and modeling of human lipoxygenases. Proteins 24, 275–291 (1996).
Sigal, E. et al. Molecular cloning and primary structure of human 15-lipoxygenase. Biochem. Biophys. Res. Comm. 157, 457–464 (1988).
Winkler, F.W., D′Arcy, A. & Hunziker, W. Structure of human pancreatic lipase. Nature 343, 771–774 (1990).
Williams, S.E. et al. The carboxyl-terminal domain of lipoprotein lipase binds to the low density lipoprotein receptor related protein/a2-macroglobulin receptor (LRP) and mediates binding of normal very low density lipoproteins to LRP. J. Biol. Chem. 269, 8653–8658 (1994).
Van Tilbeurgh, H., Sarda, L., Verger, R. & Cambillau, C. Structure of the pancreatic lipase-procolipase complex. Nature 359, 159–163 (1992).
Carriere, F. et al. Pancreatic lipase structure-funtion relationships by domain exchange. Biochemistry 36, 239–248 (1997).
Miller, D.K. et al. Identification and isolation of a membrane protein necessary for leukotriene production. Nature 343, 278–281 (1990).
Dixon, R.A.F. et al. Requirement of a 5-lipoxygenase activating protein for leukotriene synthesis. Nature 343, 282–284 (1990).
Zhang, Y., Gan, Q.-F., Pavel, E.G., Sigal, E. & Solomon, E.I. EPR definition of the non-heme ferric active sites of mammalian 15-lipoxygenase: major spectral difference relative to human 5-lipoxygenase and plant lipoxygenase and their ligand field origin. J. Am. Chem. Soc. 117, 7422–7427 (1995).
Gorins, G., Kuhnert, L., Johnson, C.R. & Marnett, L.J. (Carboxyalkyl)benzyl propargyl ethers as selective inhibitors of leukocyte-type 12-lipoxygenases. J. Med. Chem. 39, 4871–4878 (1996).
Corey, E.J. & Nagata, R. Evidence in favor of an organoiron-mediated pathway for lipoxygenation of fatty acids by soybean lipoxygenase. J. Am. Chem. Soc. 109, 8107–8108 (1987).
de Groot, J.J. et al. Demonstration by EPR spectroscopy of the functional role of iron in soybean lipoxygenase-1. Biochim. Biophys. Acta 377, 71–79 (1975).
Glickman, M.H. & Klinman, J.P. Lipoxygenase reaction mechanism: demonstration that hydrogen abstraction from substrate predeces dioxygen binding during catalytic turnover. Biochemistry 35, 12881–12892 (1996).
Gan, Q.F., Browner, M.F., Sloane, D.L. & Sigal, E. Defining the arachidonic acid binding site of human 15-lipoxygenase. Molecular modeling and mutagenesis. J. Biol. Chem. 271, 25412–25418 (1996).
Sloane, D.L., Leung, R., Craik, C.S. & Sigal, E. A primary determinant for lipoxygenase positional specificity. Nature 354, 149–152 (1991).
Chen, X.S. & Funk, C.D. Structure-function properties of human platelet 12-lipoxygenase: chimeric enzyme and in vitro mutagenesis studies. Faseb J. 7, 694–701 (1993).
Borngräber, S., Kuban, R.-J., Anton, M. & Kühn, H. Phenylalanine 353 is a primary determinant for the positional specificity of Mammalian 15-lipoxygenases. J. Mol. Biol. 264, 1145–1153 (1996).
Brash, A.R., Boeglin, W.E. & Chang, M.S. Discovery of a second 15S-lipoxygenase in humans. Proc. Natl. Acad. Sci. USA 94, 6148–6152 (1997).
Schewe, T., Rapoport, S.M. & Kuhn, H. Enzymology and Physiology of reticulocyte lipoxygenase: comparison with other lipoxygenases. in Advances in enzymology Vol. 58 (ed. Meister, A.) 191–272 (John Wiley and Sons, New York, 1986).
Picot, D., Loll, P.J. & Garavito, R.M. The X-ray crystal structure of the membrane protein prostaglandin H2 synthase-1. Nature 367, 243–249 (1994).
Luong, C. et al. Flexibility of the NSAID binding site in the structure of human cyclooxygenase-2. Nature Struct. Biol. 3, 927–933 (1996).
Li, H. & Poulos, T.L. The structure of the cytochrome p450BM-3 haem domain complexed with the fatty acid substrate palmitoleic acid. Nature Struct. Biol. 4, 140–146 (1997).
Sloane, D.L. et al. Purification and crystallization of 15-lipoxygenase from rabbit reticulocytes. Biochem. Biophys. Res. Comm. 173, 507–513 (1990).
Otwinowski, Z. Data collection and processing. in Proceedings of the CCP4 Study Weekend: Data Collection and Processing (eds Sawyer, L., Isaacs, N. & Baily, S.) 56–62 (Science and Engineering Research Council, Daresbury Laboratory, Daresbury, England, 1993).
Furey, W. & Swamintha, S. PHASES. in Macromolecular crystallography Vol. 276 (eds Carter, C. & Sweet, R.) (Academic Press, Orlando, 1996).
Muller, K. MOLOC: A molecular modeling program. Bull. Soc. Chim. 97, 655–667 (1988).
Brünger, A.T. X-PLOR: version 3.1 (Yale Press, New Haven, 1987).
Holm, L. & Sander, C. Dali ver. 2.0. J. Mol. Biol. 233, 123–138 (1993).
Nicholls, A., Sharp, K. & Honig, B. Protein folding and association: insights from the interfacical and thermodynamic properties of hydrocarbons. Proteins 11, 281–296 (1991).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gillmor, S., Villaseñor, A., Fletterick, R. et al. The structure of mammalian 15-lipoxygenase reveals similarity to the lipases and the determinants of substrate specificity. Nat Struct Mol Biol 4, 1003–1009 (1997). https://doi.org/10.1038/nsb1297-1003
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsb1297-1003
This article is cited by
-
Production of C20 9S- and C22 11S-hydroxy fatty acids by cells expressing Shewanella hanedai arachidonate 9S-lipoxygenase
Applied Microbiology and Biotechnology (2023)
-
A Bifunctional Coordination Polymer for Al3+ Ion Detection and its Treatment Effect on Nonalcoholic Fatty Liver Disease by Promoting Lipoxygenases and Reducing Intrahepatic Triglyceride Content
Journal of Fluorescence (2021)
-
Structural and mechanistic insights into 5-lipoxygenase inhibition by natural products
Nature Chemical Biology (2020)
-
Discovery two potent and new inhibitors of 15-lipoxygenase: (E)-3-((3,4-dihydroxybenzylidene) amino)-7-hydroxy-2H-chromen-2-one and (E)-O-(4-(((7-hydroxy-2-oxo-2H-chromen-3-yl) imino)methine) phenyl)dimethylcarbamothioate
Medicinal Chemistry Research (2017)
-
Characterization of the depolymerizing activity of commercial lipases and detection of lipase-like activities in animal organ extracts using poly(3-hydroxybutyrate-co-4-hydroxybutyrate) thin film
AMB Express (2016)