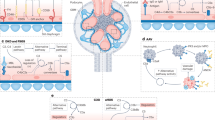
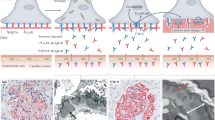
Abstract
Several distinct pathological patterns of glomerular inflammation are associated with abnormal regulation of the complement system, specifically, with dysregulation of the alternative pathway of the complement system. However, these conditions share the pathological finding of complement C3 (C3) deposited within the glomerulus in the absence of substantial immunoglobulin. This finding has alerted us and others to the possible presence of genetic and acquired complement dysregulation in individual patients. This article summarizes our current understanding of the relationship between dysregulation of the complement system and glomerular inflammation. Here, we suggest that glomerular pathologies that are characterized by the isolated deposition of C3 could usefully be classified by the term C3 glomerulopathy. In our view, this classification would alert the pathologist and nephrologist to the importance of screening for acquired and genetic abnormalities in complement regulation. In the future, it could help to identify individuals who might benefit from therapeutic inhibition of the complement system.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Servais, A. et al. Primary glomerulonephritis with isolated C3 deposits: a new entity which shares common genetic risk factors with haemolytic uraemic syndrome. J. Med. Genet. 44, 193–199 (2007).
Alchi, B. & Jayne, D. Membranoproliferative glomerulonephritis. Pediatr. Nephrol. doi:10.1007/s00467-009-1322–1327.
Habib, R., Gubler, M. C., Loirat, C., Maiz, H. B. & Levy, M. Dense deposit disease: a variant of membranoproliferative glomerulonephritis. Kidney Int. 7, 204–215 (1975).
Walker, P. D., Ferrario, F., Joh, K. & Bonsib, S. M. Dense deposit disease is not a membranoproliferative glomerulonephritis. Mod. Pathol. 20, 605–616 (2007).
Levy, M., Gubler, M. C., Sich, M., Beziau, A. & Habib, R. Immunopathology of membranoproliferative glomerulonephritis with subendothelial deposits (type I MPGN). Clin. Immunol. Immunopa thol. 10, 477–492 (1978).
Lachmann, P. J. The amplification loop of the complement pathways. Adv. Immunol. 104, 115–149 (2009).
Licht, C. & Frémeaux-Bacchi, V. Hereditary and acquired complement dysregulation in membranoproliferative glomerulonephritis. Thromb. Haemost. 101, 271–278 (2009).
Montes, T., Tortajada, A., Morgan, B. P., Rodriguez de Cordoba, S. & Harris, C. L. Functional basis of protection against age-related macular degeneration conferred by a common polymorphism in complement factor B. Proc. Natl Acad. Sci. USA 106, 4366–4371 (2009).
Tortajada, A. et al. The disease-protective complement factor H allotypic variant Ile62 shows increased binding affinity for C3b and enhanced cofactor activity. Hum. Mol. Genet. 18, 3452–3461 (2009).
Pickering, M. C. & Cook, H. T. Translational mini-review series on complement factor H: renal diseases associated with complement factor H: novel insights from humans and animals. Clin. Exp. Immuno l. 151, 210–230 (2008).
Jokiranta, T. S. et al. Where next with atypical hemolytic uremic syndrome? Mol. Immunol. 44, 3889–3900 (2007).
Kavanagh, D., Richards, A. & Atkinson, J. Complement regulatory genes and hemolytic uremic syndromes. Annu. Rev. Med. 59, 293–309 (2008).
Loirat, C., Noris, M. & Frémeaux-Bacchi, V. Complement and the atypical hemolytic uremic syndrome in children. Pediatr. Nephrol. 23, 1957–1972 (2008).
Smith, R. J. et al. New approaches to the treatment of dense deposit disease. J. Am. Soc. Nephrol. 18, 2447–2456 (2007).
West, C. D., Witte, D. P. & McAdams, A. J. Composition of nephritic factor-generated glomerular deposits in membranoproliferative glomerulonephritis type 2. Am. J. Kidney Dis. 37, 1120–1130 (2001).
Sethi, S. et al. Glomeruli of dense deposit disease contain components of the alternative and terminal complement pathway. Kidney Int. 75, 952–960 (2009).
Vallota, E. H., Forristal, J., Spitzer, R. E., Davis, N. C. & West, C. D. Continuing C3 breakdown after bilateral nephrectomy in patients with membrano-proliferative glomerulonephritis. J. Clin. Invest. 50, 552–558 (1971).
Nasr, S. H. et al. Dense deposit disease: clinicopathologic study of 32 pediatric and adult patients. Clin. J. Am. Soc. Nephrol. 4, 22–32 (2009).
Appel, G. B. et al. Membranoproliferative glomerulonephritis type II (dense deposit disease): an update. J. Am. Soc. Nephrol. 16, 1392–1403 (2005).
Bennett, W. M. et al. Mesangiocapillary glomerulonephritis type II (dense-deposit disease): clinical features of progressive disease. Am. J. Kidney Dis. 13, 469–476 (1989).
Cameron, J. S. et al. Idiopathic mesangiocapillary glomerulonephritis. Comparison of types I and II in children and adults and long-term prognosis. Am. J. Med. 74, 175–192 (1983).
Neary, J. J. et al. Linkage of a gene causing familial membranoproliferative glomerulonephritis type III to chromosome 1. J. Am. Soc. Nephrol. 13, 2052–2057 (2002).
Neary, J., Dorman, A., Campbell, E., Keogan, M. & Conlon, P. Familial membranoproliferative glomerulonephritis type III. Am. J. Kidney Dis. 40, E1 (2002).
Goicoechea de Jorge, E. et al. A mutant complement factor H-related 5 protein is associated with familial C3 glomerulonephritis Mol. Immunol. 46, 2822 (2009).
Roumenina, l. et al. Acquired and hereditary complement dysregulation in patients with C3 glomerulopathy. Mol. Immunol. 46, 2864 (2009).
Trouw, L. A. et al. Anti-C1q autoantibodies deposit in glomeruli but are only pathogenic in combination with glomerular C1q-containing immune complexes. J. Clin. Invest. 114, 679–688 (2004).
Pickering, M. C., Botto, M., Taylor, P. R., Lachmann, P. J. & Walport, M. J. Systemic lupus erythematosus, complement deficiency, and apoptosis. Adv. Immunol. 76, 227–324 (2000).
Rose, K. L. et al. Factor I is required for the development of membranoproliferative glomerulonephritis in factor H-deficient mice. J. C lin. Invest. 118, 608–618 (2008).
Wagner, E. & Frank, M. M. Therapeutic potential of complement modulation. Nat. Rev. Drug Discov. 9, 43–56 (2010).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Fakhouri, F., Frémeaux-Bacchi, V., Noël, LH. et al. C3 glomerulopathy: a new classification. Nat Rev Nephrol 6, 494–499 (2010). https://doi.org/10.1038/nrneph.2010.85
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneph.2010.85
This article is cited by
-
Renal pathology in adult and paediatric population of Japan: review of the Japan renal biopsy registry database from 2007 to 2017
Journal of Nephrology (2023)
-
Successful treatment of infectious endocarditis-associated glomerulonephritis during active hepatitis C infection: a case report
BMC Nephrology (2022)
-
Gross hematuria, edema, and hypocomplementemia in a 9-year-old boy: Answers
Pediatric Nephrology (2022)
-
Biomarkers in pediatric glomerulonephritis and nephrotic syndrome
Pediatric Nephrology (2021)
-
C3 glomerulopathy and atypical hemolytic uremic syndrome: an updated review of the literature on alternative complement pathway disorders
International Urology and Nephrology (2021)