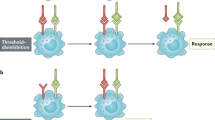
Key Points
-
Interleukin-7 (IL-7) is required, and limiting, for T-cell development and survival.
-
IL-7 production by stromal cells in lymphoid tissues is constitutive.
-
The amount of available IL-7 is proposed to be significantly reduced by T-cell consumption.
-
IL-7 receptor expression varies dramatically during the life of a T cell. In the thymus, it is expressed at the double-negative stage, not expressed at the double-positive stage, and then re-expressed at the single-positive stage. In the periphery, it is expressed by naive T cells, but not by activated T cells, and is re-expressed by memory T cells. Naive T cells also turn off receptor expression transiently after IL-7 stimulation.
-
This dramatic downregulation of IL-7 receptor expression is proposed to have two benefits: first, the T cell at these discrete stages has alternative survival stimuli (such as antigen); second, the T cell 'altruistically' stops consuming IL-7, leaving more for its neighbouring cells.
Abstract
Interleukin-7 (IL-7) is produced by stromal cells in lymphoid tissues and is required for the development of T cells and for their persistence in the periphery. Unlike many other cytokines that act on lymphocytes, IL-7 production by stromal cells is not substantially affected by extrinsic stimuli. So, the amount of available IL-7 protein is thought to be regulated by the rate that it is scavenged by T cells. As we review here, there is mounting evidence indicating that the amount of IL-7 receptor expressed on a cell not only determines how vigorously the cell responds to IL-7, but it can also determine how efficiently the cell consumes IL-7 and, therefore, affect the supply of this limiting resource in the niche.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Grabstein, K. H. et al. Inhibition of murine B and T lymphopoiesis in vivo by an anti-interleukin 7 monoclonal antibody. J. Exp. Med. 178, 257–264 (1993).
Sudo, T. et al. Expression and function of the interleukin 7 receptor in murine lymphocytes. Proc. Natl Acad. Sci. USA 90, 9125–9129 (1993).
Bhatia, S. K., Tygrett, L. T., Grabstein, K. H. & Waldschmidt, T. J. The effect of in vivo IL-7 deprivation on T cell maturation. J. Exp. Med. 181, 1399–1409 (1995).
von Freeden-Jeffry, U. et al. Lymphopenia in interleukin (IL)-7 gene-deleted mice identifies IL-7 as a nonredundant cytokine. J. Exp. Med. 181, 1519–1526 (1995). Knockout of the Il7 gene recapitulates the marked lymphopaenia seen in Il7r -knockout mice.
Peschon, J. J. et al. Early lymphocyte expansion is severely impaired in interleukin 7 receptor-deficient mice. J. Exp. Med. 180, 1955–1960 (1994). Knockout of Il7r results in severe thymic deficiency and lymphopaenia in mice.
Maki, K. et al. Interleukin 7 receptor-deficient mice lack γδ T cells. Proc. Natl Acad. Sci. USA 93, 7172–7177 (1996).
He, W., Zhang, Y., Deng, Y. & Kabelitz, D. Induction of TCR-γδ expression on triple-negative (CD3−4−8−) human thymocytes. Comparative analysis of the effects of IL-4 and IL-7. J. Immunol. 154, 3726–3731 (1995).
Plum, J., De, S. M., Leclercq, G., Verhasselt, B. & Vandekerckhove, B. Interleukin-7 is a critical growth factor in early human T-cell development. Blood 88, 4239–4245 (1996).
Yeoman, H., Clark, D. R. & DeLuca, D. Development of CD4 and CD8 single positive T cells in human thymus organ culture: IL-7 promotes human T cell production by supporting immature T cells. Dev. Comp. Immunol. 20, 241–263 (1996).
Schluns, K. S., Kieper, W. C., Jameson, S. C. & Lefrancois, L. Interleukin-7 mediates the homeostasis of naive and memory CD8 T cells in vivo. Nature Immunol. 1, 426–432 (2000). Shows that IL-7 is required for the survival of mature peripheral T cells and notes that antigen stimulation downregulates IL-7Rα expression.
Kondrack, R. M. et al. Interleukin 7 regulates the survival and generation of memory CD4 cells. J. Exp. Med. 198, 1797–1806 (2003).
Cunningham-Rundles, C. & Ponda, P. P. Molecular defects in T- and B-cell primary immunodeficiency diseases. Nature Rev. Immunol. 5, 880–892 (2005). Reviews the requirements for the IL-7 pathway in humans.
He, Y. W. & Malek, T. R. Interleukin-7 receptor α is essential for the development of γδ+ T cells, but not natural killer cells. J. Exp. Med. 184, 289–293 (1996).
Boesteanu, A. et al. Distinct roles for signals relayed through the common cytokine receptor γ chain and interleukin 7 receptor α chain in natural T cell development. J. Exp. Med. 186, 331–336 (1997).
Khaled, A. R. & Durum, S. K. Lymphocide: cytokines and the control of lymphoid homeostasis. Nature Rev. Immunol. 2, 817–830 (2002).
Durum, S. K. et al. Interleukin 7 receptor control of T cell receptor γ gene rearrangement: role of receptor-associated chains and locus accessibility. J. Exp. Med. 188, 2233–2241 (1998).
Corcoran, A. E., Riddell, A., Krooshoop, D. & Venkitaraman, A. R. Impaired immunoglobulin gene rearrangement in mice lacking the IL-7 receptor. Nature 391, 904–907 (1998).
Brugnera, E. et al. Coreceptor reversal in the thymus: signaled CD4+8+ thymocytes initially terminate CD8 transcription even when differentiating into CD8+ T cells. Immunity 13, 59–71 (2000).
Khaled, A. R. et al. Cytokine-driven cell cycling is mediated through Cdc25A. J. Cell Biol. 169, 755–763 (2005).
Li, W. Q. et al. IL-7 promotes T cell proliferation through destabilization of p27Kip1. J. Exp. Med. 203, 573–582 (2006).
Ziegler, S. F. & Liu, Y. J. Thymic stromal lymphopoietin in normal and pathogenic T cell development and function. Nature Immunol. 7, 709–714 (2006).
Al-Shami, A. et al. A role for thymic stromal lymphopoietin in CD4+ T cell development. J. Exp. Med. 200, 159–168 (2004).
Fry, T. J. & Mackall, C. L. The many faces of IL-7: from lymphopoiesis to peripheral T cell maintenance. J. Immunol. 174, 6571–6576 (2005).
Park, J. H. et al. Suppression of IL7Rα transcription by IL-7 and other prosurvival cytokines: a novel mechanism for maximizing IL-7-dependent T cell survival. Immunity 21, 289–302 (2004). Shows that IL-7 downregulates IL-7Rα expression by mature T cells in vitro and in vivo , and proposes the altruistic hypothesis.
Munitic, I. et al. Dynamic regulation of IL-7 receptor expression is required for normal thymopoiesis. Blood 104, 4165–4172 (2004). Forced expression of IL-7Rα in thymocytes is shown to inhibit thymopoiesis, and the altruistic hypothesis is proposed.
Kondo, M., Weissman, I. L. & Akashi, K. Identification of clonogenic common lymphoid progenitors in mouse bone marrow. Cell 91, 661–672 (1997).
Miller, J. P. et al. The earliest step in B lineage differentiation from common lymphoid progenitors is critically dependent upon interleukin 7. J. Exp. Med. 196, 705–711 (2002).
Bhandoola, A. & Sambandam, A. From stem cell to T cell: one route or many? Nature Rev. Immunol. 6, 117–126 (2006).
Porter, B. O., Scibelli, P. & Malek, T. R. Control of T cell development in vivo by subdomains within the IL-7 receptor α-chain cytoplasmic tail. J. Immunol. 166, 262–269 (2001).
Yu, Q., Erman, B., Park, J. H., Feigenbaum, L. & Singer, A. IL-7 receptor signals inhibit expression of transcription factors TCF-1, LEF-1, and RORγt: impact on thymocyte development. J. Exp. Med. 200, 797–803 (2004). Forced expression of IL-7Rα in thymocytes is shown to inhibit thymopoiesis through interference with developmental signals.
Yu, Q. et al. Cytokine signal transduction is suppressed in preselection double-positive thymocytes and restored by positive selection. J. Exp. Med. 203, 165–175 (2006).
Purohit, S. J. et al. Determination of lymphoid cell fate is dependent on the expression status of the IL-7 receptor. EMBO J. 22, 5511–5521 (2003).
Jiang, Q. et al. Retroviral transduction of IL-7Rα into IL-7Rα−/− bone marrow progenitors: correction of lymphoid deficiency and induction of neutrophilia. Gene Ther. 12, 1761–1768 (2005).
Van De Wiele, C. J. et al. Thymocytes between the β-selection and positive selection checkpoints are nonresponsive to IL-7 as assessed by STAT-5 phosphorylation. J. Immunol. 172, 4235–4244 (2004).
Maraskovsky, E. et al. Bcl-2 can rescue T lymphocyte development in interleukin-7 receptor-deficient mice but not in mutant rag-1−/− mice. Cell 89, 1011–1019 (1997).
Kondo, M., Akashi, K., Domen, J., Sugamura, K. & Weissman, I. L. Bcl-2 rescues T lymphopoiesis, but not B or NK cell development, in common γ chain-deficient mice. Immunity 7, 155–162 (1997).
Khaled, A. R. et al. Bax deficiency partially corrects interleukin-7 receptor α deficiency. Immunity 17, 561–573 (2002).
Candeias, S., Muegge, K. & Durum, S. K. IL-7 receptor and VDJ recombination: trophic versus mechanistic actions. Immunity 6, 501–508 (1997).
Jiang, Q. et al. Distinct regions of the interleukin-7 receptor regulate different Bcl2 family members. Mol. Cell. Biol. 24, 6501–6513 (2004).
Prockop, S. E. & Petrie, H. T. Regulation of thymus size by competition for stromal niches among early T cell progenitors. J. Immunol. 173, 1604–1611 (2004).
Yasuda, Y., Kaneko, A., Nishijima, I., Miyatake, S. & Arai, K. Interleukin-7 inhibits pre-T-cell differentiation induced by the pre-T-cell receptor signal and the effect is mimicked by hGM-CSF in hGM-CSF receptor transgenic mice. Immunology 106, 212–221 (2002).
Hassan, J. & Reen, D. J. Human recent thymic emigrants–identification, expansion, and survival characteristics. J. Immunol. 167, 1970–1976 (2001).
Webb, L. M., Foxwell, B. M. & Feldmann, M. Putative role for interleukin-7 in the maintenance of the recirculating naive CD4+ T-cell pool. Immunology 98, 400–405 (1999).
Soares, M. V. et al. IL-7-dependent extrathymic expansion of CD45RA+ T cells enables preservation of a naive repertoire. J. Immunol. 161, 5909–5917 (1998).
Dardalhon, V. et al. IL-7 differentially regulates cell cycle progression and HIV-1-based vector infection in neonatal and adult CD4+ T cells. Proc. Natl Acad. Sci. USA 98, 9277–9282 (2001).
Franchimont, D. et al. Positive effects of glucocorticoids on T cell function by up-regulation of IL-7 receptor α. J. Immunol. 168, 2212–2218 (2002). Shows that TCR activation downregulates IL-7Rα expression.
Kaech, S. M. et al. Selective expression of the interleukin 7 receptor identifies effector CD8 T cells that give rise to long-lived memory cells. Nature Immunol. 4, 1191–1198 (2003). Shows that memory T cells derive from a subset of activated T cells that do not downregulate IL-7Rα expression.
Huster, K. M. et al. Selective expression of IL-7 receptor on memory T cells identifies early CD40L-dependent generation of distinct CD8+ memory T cell subsets. Proc. Natl Acad. Sci. USA 101, 5610–5615 (2004).
Madakamutil, L. T. et al. CD8αα-mediated survival and differentiation of CD8 memory T cell precursors. Science 304, 590–593 (2004).
Chandele, A. & Kaech, S. M. Cutting edge: memory CD8 T cell maturation occurs independently of CD8αα. J. Immunol. 175, 5619–5623 (2005).
Klonowski, K. D., Williams, K. J., Marzo, A. L. & Lefrancois, L. Cutting edge: IL-7-independent regulation of IL-7 receptor α expression and memory CD8 T cell development. J. Immunol. 177, 4247–4251 (2006).
Sallusto, F., Lenig, D., Forster, R., Lipp, M. & Lanzavecchia, A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 401, 708–712 (1999).
Bachmann, M. F., Wolint, P., Schwarz, K., Jager, P. & Oxenius, A. Functional properties and lineage relationship of CD8+ T cell subsets identified by expression of IL-7 receptor α and CD62L. J. Immunol. 175, 4686–4696 (2005).
Tan, J. T. et al. Interleukin (IL)-15 and IL-7 jointly regulate homeostatic proliferation of memory phenotype CD8+ cells but are not required for memory phenotype CD4+ cells. J. Exp. Med. 195, 1523–1532 (2002).
Buentke, E. et al. Do CD8 effector cells need IL-7R expression to become resting memory cells? Blood 108, 1949–1956 (2006).
Lenz, D. C. et al. IL-7 regulates basal homeostatic proliferation of antiviral CD4+ T cell memory. Proc. Natl Acad. Sci. USA 101, 9357–9362 (2004).
Trautmann, L. et al. Upregulation of PD-1 expression on HIV-specific CD8+ T cells leads to reversible immune dysfunction. Nature Med. 12, 1198–1202 (2006).
Day, C. L. et al. PD-1 expression on HIV-specific T cells is associated with T-cell exhaustion and disease progression. Nature 443, 350–354 (2006).
Carini, C., McLane, M. F., Mayer, K. H. & Essex, M. Dysregulation of interleukin-7 receptor may generate loss of cytotoxic T cell response in human immunodeficiency virus type 1 infection. Eur. J. Immunol. 24, 2927–2934 (1994).
MacPherson, P. A., Fex, C., Sanchez-Dardon, J., Hawley-Foss, N. & Angel, J. B. Interleukin-7 receptor expression on CD8+ T cells is reduced in HIV infection and partially restored with effective antiretroviral therapy. J. Acquir. Immune Defic. Syndr. 28, 454–457 (2001).
Mussini, C. et al. Features of 'CD4-exploders', HIV-positive patients with an optimal immune reconstitution after potent antiretroviral therapy. AIDS 16, 1609–1616 (2002).
Lang, K. S. et al. Inverse correlation between IL-7 receptor expression and CD8 T cell exhaustion during persistent antigen stimulation. Eur. J. Immunol. 35, 738–745 (2005).
van Leeuwen, E. M. et al. IL-7 receptor α chain expression distinguishes functional subsets of virus-specific human CD8+ T cells. Blood 106, 2091–2098 (2005).
de Bree, G. J. et al. Respiratory syncytial virus-specific CD8+ memory T cell responses in elderly persons. J. Infect. Dis. 191, 1710–1718 (2005).
Paiardini, M. et al. Loss of CD127 expression defines an expansion of effector CD8+ T cells in HIV-infected individuals. J. Immunol. 174, 2900–2909 (2005).
Koesters, S. A. et al. IL-7Rα expression on CD4+ T lymphocytes decreases with HIV disease progression and inversely correlates with immune activation. Eur. J. Immunol. 36, 336–344 (2006).
Colle, J. H. et al. CD127 expression and regulation are altered in the memory CD8 T cells of HIV-infected patients — reversal by highly active anti-retroviral therapy (HAART). Clin. Exp. Immunol. 143, 398–403 (2006).
Boettler, T. et al. Expression of the interleukin-7 receptor α chain (CD127) on virus-specific CD8+ T cells identifies functionally and phenotypically defined memory T cells during acute resolving hepatitis B virus infection. J. Virol. 80, 3532–3540 (2006).
Lacombe, M. H., Hardy, M. P., Rooney, J. & Labrecque, N. IL-7 receptor expression levels do not identify CD8+ memory T lymphocyte precursors following peptide immunization. J. Immunol. 175, 4400–4407 (2005).
Kieper, W. C. et al. Overexpression of interleukin (IL)-7 leads to IL-15-independent generation of memory phenotype CD8+ T cells. J. Exp. Med. 195, 1533–1539 (2002).
Seddon, B., Tomlinson, P. & Zamoyska, R. Interleukin 7 and T cell receptor signals regulate homeostasis of CD4 memory cells. Nature Immunol. 4, 680–686 (2003).
Li, J., Huston, G. & Swain, S. L. IL-7 promotes the transition of CD4 effectors to persistent memory cells. J. Exp. Med. 198, 1807–1815 (2003).
Lee, H. C., Shibata, H., Ogawa, S., Maki, K. & Ikuta, K. Transcriptional regulation of the mouse IL-7 receptor α promoter by glucocorticoid receptor. J. Immunol. 174, 7800–7806 (2005).
DeKoter, R. P., Lee, H. J. & Singh, H. PU.1 regulates expression of the interleukin-7 receptor in lymphoid progenitors. Immunity 16, 297–309 (2002).
Medina, K. L. et al. Assembling a gene regulatory network for specification of the B cell fate. Dev. Cell 7, 607–617 (2004).
Singh, H., Medina, K. L. & Pongubala, J. M. Contingent gene regulatory networks and B cell fate specification. Proc. Natl Acad. Sci. USA 102, 4949–4953 (2005).
Anderson, M. K., Hernandez-Hoyos, G., Diamond, R. A. & Rothenberg, E. V. Precise developmental regulation of Ets family transcription factors during specification and commitment to the T cell lineage. Development 126, 3131–3148 (1999).
Xue, H. H. et al. GA binding protein regulates interleukin 7 receptor α-chain gene expression in T cells. Nature Immunol. 5, 1036–1044 (2004).
Ristevski, S. et al. The ETS transcription factor GABPα is essential for early embryogenesis. Mol. Cell. Biol. 24, 5844–5849 (2004).
O'Leary, D. A., Koleski, D., Kola, I., Hertzog, P. J. & Ristevski, S. Identification and expression analysis of alternative transcripts of the mouse GA-binding protein (Gabp) subunits α and β. Gene 344, 79–92 (2005).
Galon, J. et al. Gene profiling reveals unknown enhancing and suppressive actions of glucocorticoids on immune cells. FASEB J. 16, 61–71 (2002).
Mittelstadt, P. R. & Ashwell, J. D. Disruption of glucocorticoid receptor exon 2 yields a ligand-responsive C-terminal fragment that regulates gene expression. Mol. Endocrinol. 17, 1534–1542 (2003).
Pleiman, C. M. et al. Organization of the murine and human interleukin-7 receptor genes: two mRNAs generated by differential splicing and presence of a type I-interferon-inducible promoter. Mol. Cell. Biol. 11, 3052–3059 (1991).
Tian, B., Nowak, D. E., Jamaluddin, M., Wang, S. & Brasier, A. R. Identification of direct genomic targets downstream of the nuclear factor-κB transcription factor mediating tumor necrosis factor signaling. J. Biol. Chem. 280, 17435–17448 (2005).
Doan, L. L. et al. Growth factor independence-1B expression leads to defects in T cell activation, IL-7 receptor α expression, and T cell lineage commitment. J. Immunol. 170, 2356–2366 (2003).
Xue, H. H. et al. IL-2 negatively regulates IL-7 receptor α chain expression in activated T lymphocytes. Proc. Natl Acad. Sci. USA 99, 13759–13764 (2002). Shows that IL-7Rα expression is downregulated by IL-2, a relative of IL-7.
Zhang, L. & Rogers, T. J. k-opioid regulation of thymocyte IL-7 receptor and C-C chemokine receptor 2 expression. J. Immunol. 164, 5088–5093 (2000).
Kim, H. R., Hong, M. S., Dan, J. M. & Kang, I. Altered IL-7Rα expression with aging and the potential implications of IL-7 therapy on CD8+ T-cell immune responses. Blood 107, 2855–2862 (2006).
Powell, D. J. Jr, Dudley, M. E., Robbins, P. F. & Rosenberg, S. A. Transition of late-stage effector T cells to CD27+ CD28+ tumor-reactive effector memory T cells in humans after adoptive cell transfer therapy. Blood 105, 241–250 (2005).
Yamazaki, M. et al. Mucosal T cells expressing high levels of IL-7 receptor are potential targets for treatment of chronic colitis. J. Immunol. 171, 1556–1563 (2003).
Laouar, Y., Crispe, I. N. & Flavell, R. A. Overexpression of IL-7Rα provides a competitive advantage during early T-cell development. Blood 103, 1985–1994 (2004).
Rich, B. E., Campos-Torres, J., Tepper, R. I., Moreadith, R. W. & Leder, P. Cutaneous lymphoproliferation and lymphomas in interleukin 7 transgenic mice. J. Exp. Med. 177, 305–316 (1993).
Fisher, A. G., Burdet, C., Bunce, C., Merkenschlager, M. & Ceredig, R. Lymphoproliferative disorders in IL-7 transgenic mice: expansion of immature B cells which retain macrophage potential. Int. Immunol. 7, 415–423 (1995).
Aranami, T., Iclozan, C., Iwabuchi, K. & Onoe, K. IL-7-dependent homeostatic proliferation in the presence of a large number of T cells in gld mice. Microbiol. Immunol. 48, 477–484 (2004).
Borge, O. J., Adolfsson, J. & Jacobsen, A. M. Lymphoid-restricted development from multipotent candidate murine stem cells: distinct and complimentary functions of the c-kit and flt3-ligands. Blood 94, 3781–3790 (1999).
Martin, C. H. et al. Efficient thymic immigration of B220+ lymphoid-restricted bone marrow cells with T precursor potential. Nature Immunol. 4, 866–873 (2003).
Namen, A. E. et al. Stimulation of B-cell progenitors by cloned murine interleukin-7. Nature 333, 571–573 (1988).
Hardy, R. R., Carmack, C. E., Shinton, S. A., Kemp, J. D. & Hayakawa, K. Resolution and characterization of pro-B and pre-pro-B cell stages in normal mouse bone marrow. J. Exp. Med. 173, 1213–1225 (1991).
Carvalho, T. L., Mota-Santos, T., Cumano, A., Demengeot, J. & Vieira, P. Arrested B lymphopoiesis and persistence of activated B cells in adult interleukin 7−/− mice. J. Exp. Med. 194, 1141–1150 (2001).
Hikida, M. et al. Expression of recombination activating genes in germinal center B cells: involvement of interleukin 7 (IL-7) and the IL-7 receptor. J. Exp. Med. 188, 365–372 (1998).
Gartner, F., Alt, F. W., Monroe, R. J. & Seidl, K. J. Antigen-independent appearance of recombination activating gene (RAG)-positive bone marrow B cells in the spleens of immunized mice. J. Exp. Med. 192, 1745–1754 (2000).
Buckley, R. H. Molecular defects in human severe combined immunodeficiency and approaches to immune reconstitution. Annu. Rev. Immunol. 22, 625–655 (2004).
Prieyl, J. A. & LeBien, T. W. Interleukin 7 independent development of human B cells. Proc. Natl Acad. Sci. USA 93, 10348–10353 (1996).
Wolf, M. L., Buckley, J. A., Goldfarb, A., Law, C. L. & LeBien, T. W. Development of a bone marrow culture for maintenance and growth of normal human B cell precursors. J. Immunol. 147, 3324–3330 (1991).
Saeland, S. et al. Interleukin-7 induces the proliferation of normal human B-cell precursors. Blood 78, 2229–2238 (1991).
Moreau, I. et al. Myofibroblastic stromal cells isolated from human bone marrow induce the proliferation of both early myeloid and B-lymphoid cells. Blood 82, 2396–2405 (1993).
Moreau, I., Duvert, V., Banchereau, J. & Saeland, S. Culture of human fetal B-cell precursors on bone marrow stroma maintains highly proliferative CD20dim cells. Blood 81, 1170–1178 (1993).
Johnson, S. E., Shah, N., Panoskaltsis-Mortari, A. & LeBien, T. W. Murine and human IL-7 activate STAT5 and induce proliferation of normal human pro-B cells. J. Immunol. 175, 7325–7331 (2005).
Dittel, B. N. & LeBien, T. W. The growth response to IL-7 during normal human B cell ontogeny is restricted to B-lineage cells expressing CD34. J. Immunol. 154, 58–67 (1995).
Akashi, K., Kondo, M., von Freeden-Jeffry, U., Murray, R. & Weissman, I. L. Bcl-2 rescues T lymphopoiesis in interleukin-7 receptor-deficient mice. Cell 89, 1033–1041 (1997).
Corcoran, A. E. et al. The interleukin-7 receptor α chain transmits distinct signals for proliferation and differentiation during B lymphopoiesis. EMBO J. 15, 1924–1932 (1996).
Dias, S., Silva, H. Jr, Cumano, A. & Vieira, P. Interleukin-7 is necessary to maintain the B cell potential in common lymphoid progenitors. J. Exp. Med. 201, 971–979 (2005).
Kikuchi, K., Lai, A. Y., Hsu, C. L. & Kondo, M. IL-7 receptor signaling is necessary for stage transition in adult B cell development through up-regulation of EBF. J. Exp. Med. 201, 1197–1203 (2005).
Wei, C., Zeff, R. & Goldschneider, I. Murine pro-B cells require IL-7 and its receptor complex to up-regulate IL-7Rα, terminal deoxynucleotidyltransferase, and cμ expression. J. Immunol. 164, 1961–1970 (2000).
Rolink, A., Grawunder, U., Haasner, D., Strasser, A. & Melchers, F. Immature surface Ig+ B cells can continue to rearrange κ and λ L chain gene loci. J. Exp. Med. 178, 1263–1270 (1993).
Milne, C. D. & Paige, C. J. IL-7: a key regulator of B lymphopoiesis. Semin. Immunol. 18, 20–30 (2006).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- γδ T cells
-
T cells that express heterodimers consisting of the γ-chain and δ-chain of the T-cell receptor. They are mainly present in the intestinal epithelium as intraepithelial lymphocytes (IELs). Although the exact function of γδ T cells (or IELs) is still unknown, it has been suggested that mucosal γδ T cells are involved in the innate immune responses of the mucosal immune system.
- Natural killer T cells
-
(NKT cells). T cells that express both natural killer (NK)-cell receptors and an αβ-T-cell receptor (αβ-TCR). In mice, these cells were first identified by their expression of the alloantigen NK1.1 (also known as NKR-P1C). Some NKT cells recognize CD1d-associated lipid antigens and express a restricted repertoire of TCRs. NKT cells are characterized by cytolytic activity and the rapid production of cytokines, including interferon-γ and interleukin-4.
- BCL-2-family members
-
(B-cell-lymphoma-2-family members). Mitochondrial proteins that increase or decrease the susceptibility of a cell to apoptosis. When levels of either BCL-2 or BCL-XL are increased, a cell is more resistant to cell death.
- V(D)J recombination
-
Somatic rearrangement of variable (V), diversity (D) and joining (J) regions of the genes that encode antigen receptors, leading to repertoire diversity of both T-cell and B-cell receptors.
- Immature single-positive cells
-
(ISP cells). Cells at a stage of thymocyte differentiation that occurs between the double-negative (CD4−CD8−) stage and the double-positive (CD4+CD8+) stage. They are characterized by rapid proliferation and, phenotypically, by the presence of either CD8 (in mice) or CD4 (in humans), with low levels of CD3 expression (in contrast to the more mature single-positive cells, which are CD3hi).
- gld mice
-
These mice have a naturally occurring mutation in CD95 ligand (also known as FAS ligand) that causes a generalized lymphoproliferative disease.
Rights and permissions
About this article
Cite this article
Mazzucchelli, R., Durum, S. Interleukin-7 receptor expression: intelligent design. Nat Rev Immunol 7, 144–154 (2007). https://doi.org/10.1038/nri2023
Issue Date:
DOI: https://doi.org/10.1038/nri2023
This article is cited by
-
Mechanisms and functions of SUMOylation in health and disease: a review focusing on immune cells
Journal of Biomedical Science (2024)
-
New insights into the stemness of adoptively transferred T cells by γc family cytokines
Cell Communication and Signaling (2023)
-
Intravenously administered interleukin-7 to reverse lymphopenia in patients with septic shock: a double-blind, randomized, placebo-controlled trial
Annals of Intensive Care (2023)
-
Identification of hub genes and immune infiltration in ulcerative colitis using bioinformatics
Scientific Reports (2023)
-
High glucose-induced IL-7/IL-7R upregulation of dermal fibroblasts inhibits angiogenesis in a paracrine way in delayed diabetic wound healing
Journal of Cell Communication and Signaling (2023)