Abstract
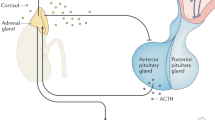
The weight of current evidence supports the presence of the following factors related to hypothalamic–pituitary–adrenal (HPA) axis dysfunction in patients with chronic fatigue syndrome (CFS): mild hypocortisolism; attenuated diurnal variation of cortisol; enhanced negative feedback to the HPA axis; and blunted HPA axis responsiveness. Furthermore, HPA axis changes seem clinically relevant, as they are associated with worse symptoms and/or disability and with poorer outcomes to standard treatments for CFS. Regarding etiology, women with CFS are more likely to have reduced cortisol levels. Studies published in the past 8 years provide further support for a multifactorial model in which several factors interact to moderate HPA axis changes. In particular, low activity levels, depression and early-life stress appear to reduce cortisol levels, whereas the use of psychotropic medication can increase cortisol. Addressing these factors—for example, with cognitive behavioral therapy—can increase cortisol levels and is probably the first-line approach for correcting HPA axis dysfunction at present, as steroid replacement is not recommended. Given what is now a fairly consistent pattern of findings for the type of HPA axis changes found in CFS, we recommend that future work focuses on improving our understanding of the cause and relevance of these observed changes.
Key Points
-
The bulk of evidence points to modest reductions in cortisol levels in some cohorts of patients with chronic fatigue syndrome (CFS), and these changes are more apparent in women than in men
-
Underlying the reduction in cortisol levels is a hypothalamic–pituitary–adrenal (HPA) axis with attenuated diurnal variation, enhanced negative feedback and blunted response to challenges
-
Low cortisol levels have clinical relevance as they might contribute to symptoms—along with other factors—and are associated with a worsened outcome of currently recommended treatments for CFS
-
A multidimensional etiological model remains most probable, with low cortisol levels occurring at a later stage of the illness, moderated by factors such as activity levels, depression, early-life stress and psychotropic medication
-
Cortisol levels can be increased by treatment with cognitive behavioral therapy, potentially because of reversal of some moderating factors
-
Further improvements in research designs remain necessary to fully understand HPA axis dysfunction in CFS
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Change history
26 June 2012
In the version of this article initially published online in Table 3 the findings in relation to the study by Van den Eede et al. should have read "Patients without early-life stress had lower mean cortisol post DEX and lower AUC total post DEX plus CRH than both controls and patients with early-life stress". The error has been corrected for the HTML and PDF versions of the article.
References
Fukuda, K. et al. The chronic fatigue syndrome: a comprehensive approach to its definition and study. International Chronic Fatigue Syndrome Study Group. Ann. Intern. Med. 121, 953–959 (1994).
Cho, H. J., Skowera, A., Cleare, A. & Wessely, S. Chronic fatigue syndrome: an update focusing on phenomenology and pathophysiology. Curr. Opin. Psychiatry 19, 67–73 (2006).
Dinos, S. et al. A systematic review of chronic fatigue, its syndromes and ethnicity: prevalence, severity, co-morbidity and coping. Int. J. Epidemiol. 38, 1554–1570 (2009).
Cleare, A. J. & Wessely, S. Chronic fatigue syndrome. eLS doi:10.1038/npg.els.0002207.
Demitrack, M. A. et al. Evidence for impaired activation of the hypothalamic–pituitary–adrenal axis in patients with chronic fatigue syndrome. J. Clin. Endocrinol. Metab. 73, 1224–1234 (1991).
Cleare, A. J. The neuroendocrinology of chronic fatigue syndrome. Endocr. Rev. 24, 236–252 (2003).
Cleare, A. J. The HPA axis and the genesis of chronic fatigue syndrome. Trends Endocrinol. Metab. 15, 55–59 (2004).
Cleare, A. J. in Handbook of Chronic Fatigue Syndrome 1st edn Ch. 16 (eds Jason, L. A., Fennell, P. A. & Taylor, R. R.) 331–362 (John Wiley & Sons, Hoboken, 2003).
Roberts, A. D., Wessely, S., Chalder, T., Papadopoulos, A. & Cleare, A. J. Salivary cortisol response to awakening in chronic fatigue syndrome. Br. J. Psychiatry 184, 136–141 (2004).
Papadopoulos, A. et al. Glucocorticoid receptor mediated negative feedback in chronic fatigue syndrome using the low dose (0.5 mg) dexamethasone suppression test. J. Affect. Disord. 112, 289–294 (2009).
Cleare, A. J., O'Keane, V. & Miell, J. P. Levels of DHEA and DHEAS and responses to CRH stimulation and hydrocortisone treatment in chronic fatigue syndrome. Psychoneuroendocrinology 29, 724–732 (2004).
Cevik, R., Gur, A., Acar, S., Nas, K. & Sarac, A. J. Hypothalamic–pituitary–gonadal axis hormones and cortisol in both menstrual phases of women with chronic fatigue syndrome and effect of depressive mood on these hormones. BMC Musculoskelet. Disord. 5, 47 (2004).
Inder, W. J., Prickett, T. C. & Mulder, R. T. Normal opioid tone and hypothalamic-pituitary-adrenal axis function in chronic fatigue syndrome despite marked functional impairment. Clin. Endocrinol. (Oxf.) 62, 343–348 (2005).
Turan, T. et al. The effects of galantamine hydrobromide treatment on dehydroepiandrosterone sulfate and cortisol levels in patients with chronic fatigue syndrome. Psychiatry Investig. 6, 204–210 (2009).
Izgi, H. B. et al. Investigation of the hypothalamopituitary-adrenal axis by low-dose (1 μg) adrenocorticotrophic hormone test and metyrapone test in patients with chronic fatigue syndrome. Endocrinologist 15, 89–92 (2005).
Gur, A., Cevik, R., Nas, K., Colpan, L. & Sarac, S. Cortisol and hypothalamic-pituitary-gonadal axis hormones in follicular-phase women with fibromyalgia and chronic fatigue syndrome and effect of depressive symptoms on these hormones. Arthritis Res. Ther. 6, R232–R238 (2004).
Maes, M., Mihaylova, I. & De Ruyter, M. Decreased dehydroepiandrosterone sulfate but normal insulin-like growth factor in chronic fatigue syndrome (CFS): relevance for the inflammatory response in CFS. Neuro. Endocrinol. Lett. 26, 487–492 (2005).
Meeus, M., Nijs, J., Van de Wauwer, N., Toeback, L. & Truijen, S. Diffuse noxious inhibitory control is delayed in chronic fatigue syndrome: an experimental study. Pain 139, 439–448 (2008).
Crofford, L. J. et al. Basal circadian and pulsatile ACTH and cortisol secretion in patients with fibromyalgia and/or chronic fatigue syndrome. Brain Behav. Immun. 18, 314–325 (2004).
Di Giorgio, A., Hudson, M., Jerjes, W. & Cleare, A. J. 24-hour pituitary and adrenal hormone profiles in chronic fatigue syndrome. Psychosom. Med. 67, 433–440 (2005).
Jerjes, W. K., Cleare, A. J., Wessely, S., Wood, P. J. & Taylor, N. F. Diurnal patterns of salivary cortisol and cortisone output in chronic fatigue syndrome. J. Affect. Disord. 87, 299–304 (2005).
Nater, U. M. et al. Alterations in diurnal salivary cortisol rhythm in a population-based sample of cases with chronic fatigue syndrome. Psychosom. Med. 70, 298–305 (2008).
Jerjes, W. K., Taylor, N. F., Peters, T. J., Wessely, S. & Cleare, A. J. Urinary cortisol and cortisol metabolite excretion in chronic fatigue syndrome. Psychosom. Med. 68, 578–582 (2006).
Jerjes, W. K. et al. Diurnal excretion of urinary cortisol, cortisone, and cortisol metabolites in chronic fatigue syndrome. J. Psychosom. Res. 60, 145–153 (2006).
Jerjes, W. K., Taylor, N. F., Wood, P. J. & Cleare, A. J. Enhanced feedback sensitivity to prednisolone in chronic fatigue syndrome. Psychoneuroendocrinology 32, 192–198 (2007).
Tak, L. M. et al. Meta-analysis and meta-regression of hypothalamic-pituitary-adrenal axis acitivity in functional somatic disorders. Biol. Psychol. 87, 183–194 (2011).
Roberts, A. D. et al. Does hypocortisolism predict a poor response to cognitive behavioural therapy in chronic fatigue syndrome? Psychol. Med. 40, 515–522 (2010).
Roberts, A. D., Papadopoulos, A. S., Wessely, S., Chalder, T. & Cleare, A. J. Salivary cortisol output before and after cognitive behavioural therapy for chronic fatigue syndrome. J. Affect. Disord. 115, 280–286 (2009).
Torres-Harding, S. et al. The associations between basal salivary cortisol and illness symptomatology in chronic fatigue syndrome. J. Appl. Biobehav. Res. 13, 157–180 (2008).
Kirschbaum, C., Pirke, K. M. & Hellhammer, D. H. The 'Trier Social Stress Test'—a tool for investigating psychobiological stress responses in a laboratory setting. Neuropsychobiology 28, 76–81 (1993).
Melmed, S. & Kleinberg, D. in Williams Textbook of Endocrinology 10th edn Ch. 8 (eds Larsen, P. R., Kronenberg, H. M., Melmed, S. & Polonsky, K. S.) 177–260 (Saunders, 2003).
Pruessner, J. C. et al. Free cortisol levels after awakening: a reliable biological marker for the assessment of adrenocortical activity. Life Sci. 61, 2539–2549 (1997).
Schmidt-Reinwald, A. et al. The cortisol response to awakening in relation to different challenge tests and a 12-hour cortisol rhythm. Life Sci. 64, 1653–1660 (1999).
Gaab, J. et al. Associations between neuroendocrine responses to the insulin tolerance test and patient characteristics in chronic fatigue syndrome. J. Psychosom. Res. 56, 419–424 (2004).
Gaab, J. et al. Stress-induced changes in LPS-induced pro-inflammatory cytokine production in chronic fatigue syndrome. Psychoneuroendocrinology 30, 188–198 (2005).
Nater, U. M. et al. Attenuated morning salivary cortisol concentrations in a population-based study of persons with chronic fatigue syndrome and well controls. J. Clin. Endocrinol. Metab. 93, 703–709 (2008).
Heim, C. et al. Childhood trauma and risk for chronic fatigue syndrome: association with neuroendocrine dysfunction. Arch. Gen. Psychiatry 66, 72–80 (2009).
Gaab, J. et al. Low-dose dexamethasone suppression test in chronic fatigue syndrome and health. Psychosom. Med. 64, 311–318 (2002).
Pruessner, J. C., Hellhammer, D. H. & Kirschbaum, C. Burnout, perceived stress, and cortisol responses to awakening. Psychosom. Med. 61, 197–204 (1999).
Yehuda, R., Boisoneau, D., Lowy, M. T. & Giller, E. L. Jr . Dose-response changes in plasma cortisol and lymphocyte glucocorticoid receptors following dexamethasone administration in combat veterans with and without posttraumatic stress disorder. Arch. Gen. Psychiatry 52, 583–593 (1995).
Goenjian, A. K. et al. Basal cortisol, dexamethasone suppression of cortisol, and MHPG in adolescents after the 1998 earthquake in Armenia. Am. J. Psychiatry 153, 929–934 (1996).
Stein, M. B., Yehuda, R., Koverola, C. & Hanna, C. Enhanced dexamethasone suppression of plasma cortisol in adult women traumatized by childhood sexual abuse. Biol. Psychiatry 42, 680–686 (2011).
Heim, C., Ehlert, U., Hanker, J. P. & Hellhammer, D. H. Abuse-related posttraumatic stress disorder and alterations of the hypothalamic–pituitary–adrenal axis in women with chronic pelvic pain. Psychosom. Med. 60, 309–318 (1998).
Gaab, J. et al. Enhanced glucocorticoid sensitivity in patients with chronic fatigue syndrome. Acta Neuropsychiatr. 15, 184–191 (2003).
Visser, J. et al. CD4 T lymphocytes from patients with chronic fatigue syndrome have decreased interferon-g production and increased sensitivity to dexamethasone. J. Infect. Dis. 177, 451–454 (1998).
Visser, J. et al. Increased sensitivity to glucocorticoids in peripheral blood mononuclear cells of chronic fatigue syndrome patients, without evidence for altered density or affinity of glucocorticoid receptors. J. Investig. Med. 49, 195–204 (2001).
Van Den Eede, F. et al. Combined dexamethasone/corticotropin-releasing factor test in chronic fatigue syndrome. Psychol. Med. 38, 963–973 (2008).
Juruena, M. F., Pariante, C. M., Papadopoulos, A. & Cleare, A. J. The development and application of the prednisolone suppression test in psychiatry: a novel tool for assessing glucocorticoid and mineralocorticoid receptor function. Mind Brain 1, 115–122 (2010).
Cleare, A. J., O'Keane, V. & Miell, J. Plasma leptin in chronic fatigue syndrome and a placebo-controlled study of the effects of low-dose hydrocortisone on leptin secretion Clin. Endocrinol. (Oxf.) 55, 113–119 (2001).
Baulieu, E. E. Dehydroepiandrosterone (DHEA): a fountain of youth? J. Clin. Endocrinol. Metab. 81, 3147–3151 (1996).
Kalimi, M., Shafagoj, Y., Loria, R., Padgett, D. & Regelson, W. Anti-glucocorticoid effects of dehydroepiandrosterone (DHEA). Mol. Cell Biochem. 131, 99–104 (1994).
Kroboth, P. D., Salek, F. S., Pittenger, A. L., Fabian, T. J. & Frye, R. F. DHEA and DHEA-S: a review. J. Clin. Pharmacol. 39, 327–348 (1999).
Maninger, N., Wolkowitz, O. M., Reus, V. I., Epel, E. S. & Mellon, S. H. Neurobiological and neuropsychiatric effects of dehydroepiandrosterone (DHEA) and DHEA sulfate (DHEAS). Front. Neuroendocrinol. 30, 65–91 (2009).
Ngai, L.-Y. & Herbert, J. in DHEA and the Brain 1st edn Ch. 3 (ed. Morfin, R.) 44–71 (Taylor and Francis, London and New York, 2002).
Burtis, C. A., Ashwood, E. R. & Bruns, D. E. Tietz textbook of clinical chemistry and molecular diagnostics (Saunders, Pennsylvania, USA, 2005).
Rosenfeld, R. S., Rosenberg, B. J., Fukushima, D. K. & Hellman, L. 24-Hour secretory pattern of dehydroisoandrosterone and dehydroisoandrosterone sulfate. J. Clin. Endocrinol. Metab. 40, 850–855 (1975).
Nieschlag, E. et al. The secretion of dehydroepiandrosterone and dehydroepiandrosterone sulphate in man. J. Endocrinol. 57, 123–134 (1973).
Salek, F. S., Bigos, K. L. & Kroboth, P. D. The influence of hormones and pharmaceutical agents on DHEA and DHEA-S. concentrations: a review of clinical studies. J. Clin. Pharmacol. 42, 247–266 (2002).
Kroboth, P. D. et al. Influence of DHEA administration on 24-hour cortisol concentrations. J. Clin. Psychopharmacol. 23, 96–99 (2003).
Wolf, O. T. et al. A single administration of dehydroepiandrosterone does not enhance memory performance in young healthy adults, but immediately reduces cortisol levels. Biol. Psychiatry 42, 845–848 (1997).
Kalimi, M., Shafagoj, Y., Loria, R., Padgett, D. & Regelson, W. Anti-glucocorticoid effects of dehydroepiandrosterone (DHEA). Mol. Cell Biochem. 131, 99–104 (1994).
Hechter, O., Grossman, A. & Chatterton, R. T. Jr . Relationship of dehydroepiandrosterone and cortisol in disease. Med. Hypotheses 49, 85–91 (1997).
Goodyer, I. M., Herbert, J. & Altham, P. M. Adrenal steroid secretion and major depression in 8- to 16-year-olds, III. Influence of cortisol/DHEA ratio at presentation on subsequent rates of disappointing life events and persistent major depression. Psychol. Med. 28, 265–273 (1998).
Reid, S. F., Chalder, T., Cleare, A., Hotopf, M. & Wessely, S. Chronic fatigue syndrome. Clin. Evid. (Online) pii, 1101 (2011).
Jason, L. et al. Baseline cortisol levels predict treatment outcomes in chronic fatigue syndrome non-pharmacologic clinical trial. J. Chronic Fatigue Syndr. 14, 39–59 (2007).
Tak, L. M. et al. As good as it gets? A meta-analysis and systematic review of methodological quality of heart rate variability studies in functional somatic disorders. Biol. Psychol. 82, 101–110 (2009).
von Elm, E. et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Plos Med. 4, e296 (2007).
Altman, D. G. & Lyman, G. H. Methodological challenges in the evaluation of prognostic factors in breast cancer. Breast Cancer Res. Treat. 52, 289–303 (1998).
Candy, B. et al. Predictors of fatigue following the onset of infectious mononucleosis. Psychol. Med. 33, 847–855 (2003).
Rubin, G. J., Hotopf, M., Papadopoulos, A. & Cleare, A. Salivary cortisol as a predictor of postoperative fatigue. Psychosom. Med. 67, 441–447 (2005).
Fries, E., Hesse, J., Hellhammer, J. & Hellhammer, D. H. A new view on hypocortisolism. Psychoneuroendocrinology 30, 1010–1016 (2005).
Fries, E in Stress. The brain–body connection. (eds Hellhammer, D. H. & Hellhammer, J.) 60–77 (Karger, Basel, 2008). [Series Eds Riecher-Rössler, A. & Steiner, M. Key issues in mental health Vol. 174].
Miller, G. E., Chen, E. & Zhou, E. S. If it goes up, must it come down? Chronic stress and the hypothalamic–pituitary-–adrenocortical axis in humans. Psychol. Bull. 133, 25–45 (2007).
Van Houdenhove, B., Van Den Eede, F. & Luyten, P. Does hypothalamic-pituitary-adrenal axis hypofunction in chronic fatigue syndrome reflect a 'crash' in the stress system? Med. Hypotheses 72, 701–705 (2009).
Gold, P. W., Licinio, J., Wong, M. L. & Chrousos, G. P. Corticotropin releasing hormone in the pathophysiology of melancholic and atypical depression and in the mechanism of action of antidepressant drugs. Ann. N. Y. Acad. Sci. 771, 716–729 (1995).
Carruthers, B. M. Definitions and aetiology of myalgic encephalomyelitis: how the Canadian consensus clinical definition of myalgic encephalomyelitis works. J. Clin. Pathol. 60, 117–119 (2007).
Cryer, P. E., Davis, S. N. & Shamoon, H. Hypoglycemia in diabetes. Diabetes Care 26, 1902–1912 (2003).
Kirschbaum, C., Tietze, A., Skoluda, N. & Dettenborn, L. Hair as a retrospective calendar of cortisol production-Increased cortisol incorporation into hair in the third trimester of pregnancy. Psychoneuroendocrinology 34, 32–37 (2009).
Thomson, S. et al. Hair analysis provides a historical record of cortisol levels in Cushing's syndrome. Exp. Clin. Endocrinol. Diabetes 118, 133–138 (2010).
Dettenborn, L., Tietze, A., Bruckner, F. & Kirschbaum, C. Higher cortisol content in hair among long-term unemployed individuals compared to controls. Psychoneuroendocrinology 35, 1404–1409 (2010).
Warnock, F. et al. Measuring cortisol and DHEA in fingernails: a pilot study. Neuropsychiatr. Dis. Treat. 6, 1–7 (2010).
Steudte, S. et al. Decreased hair cortisol concentrations in generalised anxiety disorder. Psychiatry Res. 186, 310–314 (2011).
Gow, R., Thomson, S., Rieder, M., Van, U. S. & Koren, G. An assessment of cortisol analysis in hair and its clinical applications. Forensic Sci. Int. 196, 32–37 (2010).
Koper, J. W., Manenschijn, L., Lamberts, S. W. & van Rossum, E. F. Evaluation of a method to measure long term cortisol levels. Steroids 76, 1032–1036 (2011).
Gold, P. W. & Chrousos, G. P. Organization of the stress system and its dysregulation in melancholic and atypical depression: high vs low CRH/NE states. Mol. Psychiatry 7, 254–275 (2002).
Juruena, M. F. & Cleare, A. J. Overlap between atypical depression, seasonal affective disorder and chronic fatigue syndrome [Portuguese]. Rev. Bras. Psiquiatr. 29 (Suppl. 1), S19–S26 (2007).
Acknowledgements
The authors' work was supported by the NIHR Biomedical Research Centre at South London and Maudsley NHS Trust & Institute of Psychiatry (King's College London).
Author information
Authors and Affiliations
Contributions
Both authors contributed equally to all aspects of this manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Papadopoulos, A., Cleare, A. Hypothalamic–pituitary–adrenal axis dysfunction in chronic fatigue syndrome. Nat Rev Endocrinol 8, 22–32 (2012). https://doi.org/10.1038/nrendo.2011.153
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2011.153
This article is cited by
-
Hair cortisol and self-perceived stress in adolescents with multi-system functional somatic disorders
BMC Psychiatry (2024)
-
Polyphenols as possible alternative agents in chronic fatigue: a review
Phytochemistry Reviews (2023)
-
Treatments of chronic fatigue syndrome and its debilitating comorbidities: a 12-year population-based study
Journal of Translational Medicine (2022)
-
Are there subgroups of chronic fatigue syndrome? An exploratory cluster analysis of biological markers
Journal of Translational Medicine (2021)
-
How peptic ulcer disease could potentially lead to the lifelong, debilitating effects of chronic fatigue syndrome: an insight
Scientific Reports (2021)