Abstract
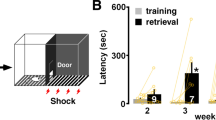
Hebb's rule (1949) states that learning and memory are based on modifications of synaptic strength among neurons that are simultaneously active. This implies that enhanced synaptic coincidence detection would lead to better learning and memory. If the NMDA (N-methyl-D-aspartate) receptor, a synaptic coincidence detector1,2,3,4, acts as a graded switch for memory formation, enhanced signal detection by NMDA receptors should enhance learning and memory. Here we show that overexpression of NMDA receptor 2B (NR2B) in the forebrains of transgenic mice leads to enhanced activation of NMDA receptors, facilitating synaptic potentiation in response to stimulation at 10–100 Hz. These mice exhibit superior ability in learning and memory in various behavioural tasks, showing that NR2B is critical in gating the age-dependent threshold for plasticity and memory formation. NMDA-receptor-dependent modifications of synaptic efficacy, therefore, represent a unifying mechanism for associative learning and memory. Our results suggest that genetic enhancement of mental and cognitive attributes such as intelligence and memory in mammals is feasible.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Stevens,C. F. & Sullivan,J. Synaptic plasticity. Curr. Biol. 8, R151–153 (1998).
Bliss,T. V. & Collingridge,G. L. A synaptic model of memory: long-term potentiation in the hippocampus. Nature 361, 31–39 (1993).
Bear,M. F. & Malenka,R. C. Synaptic plasticity: LTP and LTD. Curr. Opin. Neurobiol. 4, 389–399 (1994).
Bourne,H. R. & Nicoll,R. Molecular machines integrate coincident synaptic signals. Cell (suppl.) 72, 65–75 (1993).
Nakanishi,S. Molecular diversity of glutamate receptors and implications for brain function. Science 258, 597–603 (1992).
Hollmann,M. & Heinemann,S. Cloned glutamate receptors. Annu. Rev. Neurosci. 17, 31–108 (1994).
Monyer,H. et al. Heteromeric NMDA receptors: molecular and functional distinction of subtypes. Science 256, 1217–1221 (1992).
Monyer,H., Burnashev,N., Laurie,D. J., Sakmann,B. & Seeburg,P. H. Developmental and regional expression in the rat brain and functional properties of four NMDA receptors. Neuron 12, 529–540 (1994).
Sheng,M., Cummings,J., Roldan,L. A., Yan,Y. N. & Yan,L. Y. Changing subunit composition of heteromeric NMDA receptors during development of rat cortex. Nature 368, 144–147 (1994).
Okabe,S. et al. Hippocampal synaptic plasticity in mice overexpressing an embryonic subunit of the NMDA receptor. J. Neurosci. 18, 4177–4188 (1998).
Carmignoto,G. & Vicini,S. Activity-dependent decrease in NMDA receptor responses during development of the visual cortex. Science 258, 1007–1011 (1992).
Hestrin,S. Activation and desensitization of glutamate-activated channels mediating fast excitatory synaptic currents in the visual cortex. Neuron 9, 991–999 (1992).
Kuhl,P. K. Learning and representation in speech and language. Curr. Opin. Neurobiol. 4, 812–822 (1994).
Konishi,M. Birdsong for neurobiologists. Neuron 3, 541–549 (1989).
Mayford,M., Wang,J., Kandel,E. R. & O'Dell,T. J. CaMKII regulates the frequency-response function of hippocampal synapses for the production of both LTD and LTP. Cell 81, 891–904 (1995).
Tsien,J. Z. et al. Subregion- and cell type-restricted gene knockout in mouse brain. Cell 87, 1317–1326 (1996).
Liu,G., Choi,S. & Tsien,R. W. Variability of neurotransmitter concentration and nonsaturation of postsynaptic AMPA receptors of synapses in hippocampal cultures and slices. Neuron 22, 395–409 (1999).
Dudek,S. M. & Bear,B. F. Bidirectional long-term modification of synaptic effectiveness in the adult and immature hippocampus. J. Neurosci. 13, 2910–2918 (1993).
Harris,K. M. & Teyler,T. J. Developmental onset of long-term potentiation in area CA1 of the rat hippocampus. J. Physiol. (Lond.) 346, 27–48 (1984).
Stubli,U. & Chun,D. J. Factors regulating the reversibility of long-term potentiation. J. Neurosci. 16, 853–860 (1996).
Tsien,J. Z., Huerta,P. T. & Tonegawa,S. The essential role of hippocampal CA1 NMDA receptor-dependent synaptic plasticity in spatial memory. Cell 87, 1327–1338 (1996).
Migaud,M. et al. Enhanced long-term potentiation and impaired learning in mice with mutant postsynaptic density-95 protein. Nature 396, 433–439 (1998).
Reed,J. M. & Squire,L. R. Impaired recognition memory in patients with lesions limited to the hippocampal formation. Behav. Neurosci. 111, 667–675 (1997).
Myhrer,T. Exploratory behavior and reaction to novelty in rats with hippocampal perforant path systems disrupted. Behav. Neurosci. 102, 356–362 (1988).
Mumby,D. G. et al. Ischemia-induced object-recognition defects in rats are attenuated by hippocampal ablation before or soon after ischemia. Behav. Neurosci. 110, 266–281 (1996).
Phillips,R. G. & LeDoux,J. E. Differential contribution of amygdala and hippocampus to cued and contextual fear conditioning. Behav. Neurosci. 106, 274–285 (1992).
Kim,J. J., Fanselow,M. S., DeCola,J. P. & Landeira-Fernandez,J. Selective impairment of long-term but not short-term conditional fear by the N-methyl-D-aspartate antagonist APV. Behav. Neurosci. 106, 591–596 (1992).
Davis,M., Hitchcock,J. & Rosen,J. B. in The Psychology of Learning and Memory (ed. Bower, G. H.) (Academic, New York, 1987).
Falls,W. A., Miserendino,M. J. D. & Davis,M. Extinction of fear-potentiated startle: blockade by infusion of an NMDA antagonist into the amygdala. J. Neurosci. 12, 854–863 (1992).
Morris,R. G., Garrud,P., Rawlins,J. N. & O'Keefe,J. Place navigation impaired in rats with hippocampal lesions. Nature 24, 681–683 (1982).
Acknowledgements
We thank G. Bracket and L. Antonucci and their staff for the maintenance of Princeton Mouse Facility, M. Mayford and E. Kandel for providing the CaMKII promoter, S. Tonegwa for support of J.R.T.'s early cloning work (1995) on plasmid p279NB from which pJT-NR2B is derived and D. Prout for secretarial assistance and proofreading. This work was supported by Princeton University, Beckman Foundation and NIH (J.Z.T.).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Rights and permissions
About this article
Cite this article
Tang, YP., Shimizu, E., Dube, G. et al. Genetic enhancement of learning and memory in mice. Nature 401, 63–69 (1999). https://doi.org/10.1038/43432
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/43432
This article is cited by
-
Selective enhancement of fear extinction by inhibiting neuronal adenylyl cyclase 1 (AC1) in aged mice
Molecular Brain (2024)
-
The role of the GABAergic cells of the median raphe region in reinforcement-based learning
Scientific Reports (2024)
-
Therapeutic potential of N-methyl-D-aspartate receptor modulators in psychiatry
Neuropsychopharmacology (2024)
-
Myosin Va-dependent Transport of NMDA Receptors in Hippocampal Neurons
Neuroscience Bulletin (2024)
-
Intranasal delivery of siRNA targeting NR2B attenuates cancer-associated neuropathic pain
Journal of Pharmaceutical Investigation (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.