Abstract
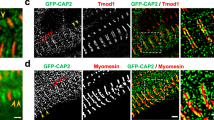
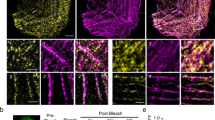
Regulation of actin dynamics at filament ends determines the organization and turnover of actin cytoskeletal structures. In striated muscle, it is believed that tight capping of the fast-growing (barbed) ends by CapZ and of the slow-growing (pointed) ends by tropomodulin (Tmod) stabilizes the uniform lengths of actin (thin) filaments in myofibrils. Here we demonstrate for the first time that both CapZ and Tmod are dynamic on the basis of the rapid incorporation of microinjected rhodamine-labelled actin (rho-actin) at both barbed and pointed ends and from the photobleaching of green fluorescent protein (GFP)-labelled Tmod. Unexpectedly, the inhibition of actin dynamics at pointed ends by GFP–Tmod overexpression results in shorter thin filaments, whereas the inhibition of actin dynamics at barbed ends by cytochalasin D has no effect on length. These data demonstrate that the actin filaments in myofibrils are relatively dynamic despite the presence of capping proteins, and that regulated actin assembly at pointed ends determines the length of thin filaments.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Littlefield, R. & Fowler, V. M. Defining actin filament length in striated muscle: rulers and caps or dynamic stability? Annu. Rev. Cell Dev. Biol. 14, 487–525 (1998).
Cooper, J. A. The role of actin polymerization in cell motility. Annu. Rev. Physiol. 53, 585–605 (1991).
Schafer, D. A. & Cooper, J. A. Control of actin assembly at filament ends. Annu. Rev. Cell Dev. Biol. 11, 497–518 (1995).
Carlier, M. F. & Pantaloni, D. Control of actin dynamics in cell motility. J Mol. Biol. 269, 459–467 (1997).
Wang, Y. L. Exchange of actin subunits at the leading edge of living fibroblasts: possible role of treadmilling. J. Cell Biol. 101, 597–602 (1985).
Svitkina, T. M. & Borisy, G. G. Arp2/3 complex and actin depolymerizing factor/cofilin in dendritic organization and treadmilling of actin filament array in lamellipodia. J. Cell Biol. 145, 1009–1026 (1999).
Wanger, M., Keiser, T., Neuhaus, J. M. & Wegner, A. The actin treadmill. Can. J. Biochem. Cell Biol. 63, 414–421 (1985).
Pollard, T. D. Rate constants for the reactions of ATP- and ADP-actin with the ends of actin filaments. J. Cell Biol. 103, 2747–2754 (1986).
Theriot, J. A. & Mitchison, T. J. Actin microfilament dynamics in locomoting cells. Nature 352, 126–131 (1991).
Symons, M. H. & Mitchison, T. J. Control of actin polymerization in live and permeabilized fibroblasts. J. Cell Biol. 114, 503–513 (1991).
Amato, P. A. & Taylor, D. L. Probing the mechanism of incorporation of fluorescently labeled actin into stress fibers. J. Cell Biol. 102, 1074–1084 (1986).
Turnacioglu, K. K., Sanger, J. W. & Sanger, J. M. Sites of monomeric actin incorporation in living PtK2 and REF-52 cells. Cell Motil. Cytoskel. 40, 59–70 (1998).
Schafer, D. A. et al. Visualization and molecular analysis of actin assembly in living cells. J. Cell Biol. 143, 1919–1930 (1998).
Forscher, P. & Smith, S. J. Actions of cytochalasins on the organization of actin filaments and microtubules in a neuronal growth cone. J. Cell Biol. 107, 1505–1516 (1988).
Casella, J. F., Craig, S. W., Maack, D. J. & Brown, A. E. Cap Z(36/32), a barbed end actin-capping protein, is a component of the Z-line of skeletal muscle. J. Cell Biol. 105, 371–379 (1987).
Fowler, V. M., Sussmann, M. A., Miller, P. G., Flucher, B. E. & Daniels, M. P. Tropomodulin is associated with the free (pointed) ends of the thin filaments in rat skeletal muscle. J. Cell Biol. 120, 411–420 (1993).
Weber, A., Pennise, C. R., Babcock, G. G. & Fowler, V. M. Tropomodulin caps the pointed ends of actin filaments. J. Cell Biol. 127, 1627–1635 (1994).
Schafer, D. A., Hug, C. & Cooper, J. A. Inhibition of CapZ during myofibrillogenesis alters assembly of actin filaments. J. Cell Biol. 128, 61–70 (1995).
Hart, M. C. & Cooper, J. A. Vertebrate isoforms of actin capping protein beta have distinct functions In vivo. J. Cell Biol. 147, 1287–1298 (1999).
Sussman, M. A. et al. Altered expression of tropomodulin in cardiomyocytes disrupts the sarcomeric structure of myofibrils. Circ. Res. 82, 94–105 (1998).
Gregorio, C. C., Weber, A., Bondad, M., Pennise, C. R. & Fowler, V. M. Requirement of pointed-end capping by tropomodulin to maintain actin filament length in embryonic chick cardiac myocytes. Nature 377, 83–86 (1995).
Sussman, M. A. et al. Myofibril degeneration caused by tropomodulin overexpression leads to dilated cardiomyopathy in juvenile mice. J Clin. Invest. 101, 51–61 (1998).
Schafer, D. A., Jennings, P. B. & Cooper, J. A. Dynamics of capping protein and actin assembly in vitro: uncapping barbed ends by polyphosphoinositides. J. Cell Biol. 135, 169–179 (1996).
Weber, A., Pennise, C. R. & Fowler, V. M. Tropomodulin increases the critical concentration of barbed end-capped actin filaments by converting ADP●Pi-actin to ADP-actin at all pointed filament ends. J. Biol. Chem. 274, 34637–34645 (1999).
Schafer, D. A., Waddle, J. A. & Cooper, J. A. Localization of CapZ during myofibrillogenesis in cultured chicken muscle. Cell Motil. Cytoskel. 25, 317–335 (1993).
Ishiwata, S. & Funatsu, T. Does actin bind to the ends of thin filaments in skeletal muscle? J. Cell Biol. 100, 282–291 (1985).
Sanger, J. W., Mittal, B. & Sanger, J. M. Analysis of myofibrillar structure and assembly using fluorescently labeled contractile proteins. J. Cell Biol. 98, 825–833 (1984).
Imanaka-Yoshida, K., Sanger, J. M. & Sanger, J. W. Contractile protein dynamics of myofibrils in paired adult rat cardiomyocytes. Cell Motil. Cytoskel. 26, 301–312 (1993).
Nwe, T. M., Maruyama, K. & Shimada, Y. Relation of nebulin and connectin (titin) to dynamics of actin in nascent myofibrils of cultured skeletal muscle cells. Exp. Cell Res. 252, 33–40 (1999).
McKenna, N., Meigs, J. B. & Wang, Y. L. Identical distribution of fluorescently labeled brain and muscle actins in living cardiac fibroblasts and myocytes. J. Cell Biol. 100, 292–296 (1985).
Dome, J. S., Mittal, B., Pochapin, M. B., Sanger, J. M. & Sanger, J. W. Incorporation of fluorescently labeled actin and tropomyosin into muscle cells. Cell Differ. 23, 37–52 (1988).
Yamaguchi, M., Izumimoto, M., Robson, R. M. & Stromer, M. H. Fine structure of wide and narrow vertebrate muscle Z-lines. A proposed model and computer simulation of Z-line architecture. J. Mol. Biol. 184, 621–643 (1985).
Sampath, P. & Pollard, T. D. Effects of cytochalasin, phalloidin, and pH on the elongation of actin filaments. Biochemistry 30, 1973–1980 (1991).
Tilney, L. G. & DeRosier, D. J. Actin filaments, stereocilia, and hair cells of the bird cochlea. IV. How the actin filaments become organized in developing stereocilia and in the cuticular plate. Dev. Biol. 116, 119–129. (1986).
Michele, D. E., Albayya, F. P. & Metzger, J. M. Thin filament protein dynamics in fully differentiated adult cardiac myocytes: toward a model of sarcomere maintenance. J. Cell Biol. 145, 1483–1495 (1999).
Hug, C. et al. Capping protein levels influence actin assembly and cell motility in Dictyostelium. Cell 81, 591–600 (1995).
Mallavarapu, A. & Mitchison, T. Regulated actin cytoskeleton assembly at filopodium tips controls their extension and retraction. J. Cell Biol. 146, 1097–1106 (1999).
Theriot, J. A. & Mitchison, T. J. Comparison of actin and cell surface dynamics in motile fibroblasts. J. Cell Biol. 119, 367–377 (1992).
Theriot, J. A., Mitchison, T. J., Tilney, L. G. & Portnoy, D. A. The rate of actin-based motility of intracellular Listeria monocytogenes equals the rate of actin polymerization. Nature 357, 257–260 (1992).
Suzuki, H., Komiyama, M., Konno, A. & Shimada, Y. Exchangeability of actin in cardiac myocytes and fibroblasts as determined by fluorescence photobleaching recovery. Tissue Cell 30, 274–280 (1998).
Almenar-Queralt, A., Gregorio, C. C. & Fowler, V. M. Tropomodulin assembles early in myofibrillogenesis in chick skeletal muscle: evidence that thin filaments rearrange to form striated myofibrils. J. Cell Sci. 112, 1111–1123 (1999).
Gregorio, C. C. & Fowler, V. M. Tropomodulin function and thin filament assembly in cardiac myocytes. Trends Cardiovasc. Med. 6, 136–141 (1996).
Draeger, A., Amos, W. B., Ikebe, M. & Small, J. V. The cytoskeletal and contractile apparatus of smooth muscle: contraction bands and segmentation of the contractile elements. J. Cell Biol. 111, 2463–2473 (1990).
Cramer, L. P., Siebert, M. & Mitchison, T. J. Identification of novel graded polarity actin filament bundles in locomoting heart fibroblasts: implications for the generation of motile force. J. Cell Biol. 136, 1287–1305 (1997).
Conley, C. A., Fritz-Six, K. A., Almernar-Queralt, A. & Fowler, V. M. Leiomodins: larger members of the tropomodulin (tmod) gene family. Genomics 73, 127–139 (2001).
Cox, P. R. & Zoghbi, H. Y. Sequencing, expression analysis, and mapping of three unique human tropomodulin genes and their mouse orthologs. Genomics 63, 97–107 (2000).
Tait, J. F. & Frieden, C. Polymerization-induced changes in the fluorescence of actin labeled with iodoacetamidotetramethylrhodamine. Arch. Biochem. Biophys. 216, 133–141 (1982).
Gregorio, C. C. & Fowler, V. M. Mechanisms of thin filament assembly in embryonic chick cardiac myocytes: tropomodulin requires tropomyosin for assembly. J. Cell Biol. 129, 683–695 (1995).
Babcock, G. G. & Fowler, V. M. Isoform-specific interaction of tropomodulin with skeletal muscle and erythrocyte tropomyosins. J. Biol. Chem. 269, 27510–27518 (1994).
Almenar-Queralt, A., Lee, A., Conley, C. A., Ribas de Pouplana, L. & Fowler, V. M. Identification of a novel tropomodulin isoform, skeletal tropomodulin, that caps actin filament pointed ends in fast skeletal muscle. J. Biol. Chem. 274, 28466–28475 (1999).
Acknowledgements
We thank K. Hahn for use of his microscope and for advice, K. Sullivan for his instruction in live-cell imaging, K. Monier for advice on photobleaching, C. Waterman-Storer for editorial comments, and C. Subauste and J. Montebella for help in preparing the rhodamine-actin. We also thank past and present members of the Fowler laboratory for suggestions and help. R.L. was the recipient of a research fellowship from the American Heart Association, Western States Affiliate. This work was supported by grants to V.M.F. from the N.I.H. (GM 34225) and from the Human Frontiers in Science Program.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary figures
Figure S1 Overview of distributed deconvolution method. (PDF 96 kb)
Figure S2 Rho-actin and phallacidin distribution functions (a, c) and profiles (b, d).
Figure S3 Comparison of 3-peak and 1-peak rho-actin profiles reveals that incorporation sites are uniformly organized within myofibrils.
Figure S4 Rho-actin distribution parameters determined by distributed deconvolution are uniform within cells but exhibit variation between cells. for all 20 images.
Supplementary movie
Movie 1 A living, beating cardiac myocyte imaged 1 h after microinjection with rho-actin. There are 20 frames, each ~1 s apart. Myofibrils do not seem to contract smoothly when imaged at this frame rate, and only occasional snapshots of myofibril stretching and contraction are visible. The myofibril shown in Fig. 1b is in the bottom half of this movie. (MOV 659 kb)
Supplementary movie
Movie 2 A different region from the same cell as in movie 1. There are several myofibrils that show instances of myofibril stretching and contraction. (MOV 1513 kb)
Rights and permissions
About this article
Cite this article
Littlefield, R., Almenar-Queralt, A. & Fowler, V. Actin dynamics at pointed ends regulates thin filament length in striated muscle. Nat Cell Biol 3, 544–551 (2001). https://doi.org/10.1038/35078517
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/35078517
This article is cited by
-
Recessive TMOD1 mutation causes childhood cardiomyopathy
Communications Biology (2024)
-
CAP2 is a regulator of actin pointed end dynamics and myofibrillogenesis in cardiac muscle
Communications Biology (2021)
-
Quantitative high-precision imaging of myosin-dependent filamentous actin dynamics
Journal of Muscle Research and Cell Motility (2020)
-
integRATE: a desirability-based data integration framework for the prioritization of candidate genes across heterogeneous omics and its application to preterm birth
BMC Medical Genomics (2018)
-
Multifunctional roles of tropomodulin-3 in regulating actin dynamics
Biophysical Reviews (2018)