Abstract
B7-1 is thought to play a pathogenic role in minimal-change disease (MCD). Recently, however, doubts have arisen regarding the role of B7-1 expression in MCD. Therefore, we aimed to identify the presence and clinical significance of B7-1 expression in MCD patients. The study participants included 28 adult MCD patients for whom kidney specimens were available. The intensity of B7-1 expression was assessed by two independent specialists. We analysed the association between the intensity of B7-1 expression and clinicopathological variables. No B7-1 expression in the glomeruli was observed in any of the 28 patients. Unexpectedly, however, 75.0% of the patients exhibited tubular B7-1 expression, with 35.7% demonstrating weak positive expressions and 39.3% demonstrating strong positive expressions. The level of proteinuria significantly increased as the intensity of tubular B7-1 expression increased. We also found trends of increasing blood urea nitrogen and serum creatinine levels with increased intensity of tubular B7-1 expression. However, we could not observe definite differences in long- and short-term clinical outcomes depending on the intensity of tubular B7-1 expression. In conclusion, B7-1 was expressed in renal tubular cells but not in glomeruli in adult MCD patients. The intensity of tubular B7-1 expression paralleled proteinuria levels, but not clinical outcomes.
Similar content being viewed by others
Introduction
Minimal-change disease (MCD) accounts for approximately 30% of cases of adult primary nephrotic syndrome (NS)1,2,3. Although MCD is well known for its good response to immunosuppressive drugs4,5, clinicians also need to keep in mind the high relapse rate4,5, which causes considerable treatment-related complications6. To date, the exact pathogenesis of MCD remains unknown, but T-cell dysfunction is suggested as a central player7,8,9.
B7-1, also known as CD80, is a surface glycoprotein expressed in antigen-presenting cells. As a co-stimulatory signal for T-cell regulation, B7-1 activates T cells by binding to CD 28 or suppresses T cells by binding to CTLA-410,11. Since Reiser et al. reported an unanticipated role of B7-1 in podocytes as an inducible modifier of glomerular permeability in proteinuric diseases12, several studies have been performed to examine the clinical significance of B7-1 expression in patients with MCD13,14,15,16,17.
However, doubts have arisen regarding the possible role of B7-1 in MCD. First, glomerular B7-1 expression might not be specific for MCD, as it is also found in other types of glomerulonephritis12,18,19. Second, glomerular B7-1 expression might not be a key player in MCD, as B7-1 expression is not always positive18 and is even completely negative in some studies20,21. Finally, B7-1 expression might be induced not only in the glomeruli but also in the tubules22,23,24. To elucidate these uncertainties, we performed the present study in adult patients with biopsy-proven MCD.
Results
Of the 28 patients, the median age was 49.0 years (range, 25.0–67.0 years) and 57.1% were men. During the follow-up period of 4.4 years (2.8–8.3 years), steroid was prescribed to every patient with a cumulative dose of 11.0 g (5.6–21.1 g) for 14.8 months (5.7–26.1 months). Calcineurin inhibitors and cyclophosphamide were prescribed to 46.4% and 10.7% of patients, respectively. Complete remission (CR) as the maximum clinical response was achieved in 96.4% of the patients, but 50.0% experienced at least one relapse. Two patients (7.1%) died of a stroke and an unidentified reason, respectively. Although none of the patients had a progression to ESRD, one patient developed doubling of creatinine level.
On light microscopic examination, 46.4% of the patients showed glomerular lesions as follows: 17.9%, global sclerosis of ≥10%; 7.1%, glomerular ischemia; 10.7%, increased glomerular cellularity; and 17.9%, increased glomerular size. In addition, 71.4% of the patients showed tubulointerstitial lesions as follows: 64.3%, tubular atrophy; 57.1%, interstitial inflammation; and 60.7%, interstitial fibrosis. On immunofluorescence (IF) examination, the positivity of IgG, IgA, IgM, C3, and C1q were 3.6%, 3.6%, 14.3%, 0%, and 7.1%, respectively.
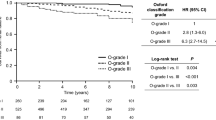
None of the patients showed B7-1-positive staining results in the glomeruli. However, 75.0% of the patients showed B7-1-positive staining results in renal tubular cells as follows: 35.7% demonstrated weak positive expression and 39.3% demonstrated strong positive expression (Fig. 1). We compared baseline characteristics according to the intensity of tubular B7-1 expression. The level of proteinuria significantly increased with the progression of the intensity of tubular B7-1 expression (Fig. 2A). We also found trends of increasing blood urea nitrogen and serum creatinine levels, with the progression of tubular B7-1 staining intensity (Fig. 2B,C). In parallel with the increased disease severity, more patients were prescribed calcineurin inhibitors with the progression of the intensity of tubular B7-1 expression (Table 1). However, we could not observe statistically significant differences in other clinical, pathological, and therapeutic parameters. In the comparison of clinical outcomes, the intensity of tubular B7-1 expression was not associated with non-CR, relapse, and death, nor with doubling of creatinine level (Fig. 3).
Discussion
MCD is thought to be a benign disease with excellent long-term prognosis4. However, many patients experience relapse5, and prolonged immunosuppression could lead to considerable treatment-related complications6. Recently, B7-1 expression has been suggested to play a key role in proteinuric renal disease by increasing glomerular permeability12, and the activity of B7-1 could be suppressed by CTLA-4 fusion proteins such as abatacept18. Therefore, clinicians expect that patients with MCD resistant to the classic treatment might be treated effectively with CTLA-4 fusion protein in cases of positive B7-1 expression. Although several studies have reported the clinical significance of B7-1 expression in MCD13,14,15,16,17, human studies with adult patients with MCD are lacking. In the present study, we found that B7-1 was not expressed in the glomeruli, but was expressed in renal tubules to varying degrees. This expression was paralleled by proteinuria levels.
Previous studies that described B7-1 expressions in MCD are summarised in Table 2. Among the study groups, one has consistently reported that the urinary B7-1 levels seemed to be higher in patients with MCD in relapse than in those with MCD in remission or other glomerular diseases13,14,25,26. To investigate the source of urinary B7-1, they evaluated kidney tissues. They found that kidney tissues from patients with MCD who were in relapse exhibited glomerular B7-1 expression, while tissues from those with MCD in remission or other glomerular diseases did not14. However, the findings of these studies cannot be generalised because they only included children for analysis and the number of kidney specimens evaluated was not sufficient (only 8 patients with MCD). Yu et al. investigated B7-1 expression in adult patients with various kidney diseases18. In the analysis, 5 patients with MCD were included, 60% of whom exhibited glomerular B7-1 expression. On the other hand, Larsen et al. reported different results after evaluating B7-1 expression in various proteinuric kidney diseases20. Among the 19 adult patients with MCD included, none exhibited glomerular B7-1 expression. This finding was confirmed by the present study and the study by Novelli et al.27. The main cause of the discrepant results on glomerular B7-1 expression level can be assumed to be the difficult work process involving anti-B7-1 antibodies, particularly in using the IF method to maintain reproducibility and accuracy20,28. As shown in Table 2, studies with positive glomerular B7-1 expression14,18 used the IF method only, whereas those with negative glomerular B7-1 expression20,27 used the immunoperoxidase (IP) method, which is known as not influenced by non-specific binding of secondary antibodies20, in addition to the IF method to confirm the results. Therefore, in future studies regarding B7-1 expression in MCD, the IF and IP methods are highly recommended for detection of kidney B7-1 expression.
In the present study, we observed B7-1 expression in renal tubular cells in adult patients with MCD. The results were mostly concordant with those of the previous study by Novelli et al.27. In their analysis with 15 patients with MCD (age range, 1.7–67.4 years), B7-1 expression was observed in injured epithelial tubular cells. However, they did not analyse the further clinical significance of tubular B7-1 expression. Compared with the above-mentioned research, our study indicates that the level of proteinuria increased with the progression of the intensity of the tubular B7-1 expression. Blood urea nitrogen and serum creatinine levels also showed increasing trends with the progression of the intensity of tubular B7-1 expression, although the statistical significance was marginal. Considering that B7-1 expression correlates with loss of kidney function, an underlying tubular injury may account for the tubular B7-1 expression. In this study, more patients with positive tubular B7-1 expression tended to have tubular atrophy than those with negative tubular B7-1 expression, although the difference was not statistically significant. With these results, we hypothesised that B7-1 may not have a pathogenic role in the development of MCD, but may have an important role in the progression of MCD. Although the clinical outcomes in this study were not affected by the intensity of the tubular B7-1 expression, we assume that the effect of tubular B7-1 expression on MCD progression can be masked by the more-intense treatment administered, with increasing intensity of tubular B7-1 expression. Theoretically, tubular B7-1 expression can potentiate kidney damages by allowing tubules to act as non-professional antigen-presenting cells23,24,29,30. Therefore, efforts to determine the role of tubular B7-1 in MCD progression are needed in future experimental or large clinical studies.
The present study has several limitations. First, no stored urine specimens were available for the measurement of B7-1 levels, as this was a retrospective study. Therefore, we cannot provide data regarding urinary B7-1 levels, although it is a good mechanistic connection between tubular B7-1 expression and proteinuria, and a fine non-invasive surrogate biomarker13,14,25,26,31. Second, selection bias existed because we only included patients for whom biopsy specimens were available. However, this weakness is inevitable in retrospective studies13,14,18,20,22,23. Moreover, the sample size was relatively larger than those of previous studies that examined the significance of tissue B7-1 expression in MCD patients14,18,20,27. Third, the intensity of B7-1 expression was assessed semi-quantitatively and subjectively. However, we think that the blinded evaluation by the two independent specialists enhanced the reliability of the assessment of B7-1 expression. Fourth, we used a single B7-1 antibody to detect B7-1 expression. In previous studies20,27, the IP method was adopted to confirm results from the IF method. The antibody used in this study was for the IP method and was identical to that used in previous studies20,27. Therefore, we think that the effect of this limitation on the study outcome is acceptable. Finally, as this was a single-centre study, the generalisability of its results is limited.
In conclusion, B7-1 was expressed in renal tubular cells but not in the glomeruli of the adult patients with MCD. The intensity of the tubular B7-1 expression paralleled proteinuria levels but not clinical outcomes. Further studies are needed to elucidate the exact significance of tubular B7-1 expression in adult patients with MCD.
Methods
Patients
From 2003 to 2013, 83 patients were diagnosed as having a biopsy-proven MCD at Seoul National University Bundang Hospital (SNUBH), a tertiary care hospital. Among these patients, 70 met the study inclusion criteria as adults with disease classified as MCD with nephrotic-range proteinuria. Four patients aged <15 years, 8 patients with urine protein-to-creatinine ratio (UPCR) of <3.0 g/g, and 1 patient with focal segmental glomerulosclerosis on subsequent biopsy were excluded. After further exclusion of 42 patients for whom kidney specimens were unavailable, we ultimately included 28 patients in the study. We concluded that all the 28 patients had primary MCD because of the lack of evidence of secondary causes such as lupus, IgA nephropathy, active cancer, or current use of non-steroidal anti-inflammatory drugs. This study was approved by the institutional review board (IRB) of SNUBH (IRB No. B-1510/320-114). All the following methods were performed in accordance with the guidelines and regulations of the IRB of SNUBH. The need for informed consent was waived because the study did not infringe on the patients’ privacy or health status.
Definitions and measurements
Demographic, physiological, laboratory, and therapeutic data were obtained from the electronic medical records database. After different patient datasets were merged, data verification was performed manually. The first date of nephrotic range proteinuria was the start of our study, and the end of the study was a later date between the date of creatinine doubling and onset of end-stage renal disease (ESRD), or the time of death. Definitions of MCD courses are summarised in Supplemental Table 1. Body mass index was calculated as weight (kg) per square of height (m2). Serum creatinine level was measured by using the rate-blanked compensated kinetic alkaline picrate Jaffe method with an automatic analyser (Toshiba-200FR, Tokyo, Japan). The between-day coefficients of variation for serum creatinine level were 1.5–2.6% and 1.1–2.4% at low (168.0–176.8 μmol/L) and high concentrations (583.4–627.6 μmol/L), respectively, throughout the study period. Microscopic haematuria was defined as ≥5 red blood cells/high-power field on urine sediment microscopy. Acute kidney injury was defined as an increase in serum creatinine level of ≥0.3 mg/dL within 48 hours or ≥1.5 times higher than that at baseline32.
Kidney pathology
All the specimens were embedded in paraffin and stained with periodic acid-Schiff, Masson trichrome, methenamine silver, and haematoxylin-eosin. Glomerular lesions were defined as global sclerosis of ≥10.0%33, increased glomerular size or cellularity, or the presence of glomerular ischemia. Tubular atrophy, and interstitial inflammation and fibrosis were defined as non-normal reports34. Vascular lesions were defined as the presence of arteriolar hyalinosis and arteriosclerosis. Detailed information regarding the IF study methods are described elsewhere4. In brief, the classic direct technique with antibodies against 5 antigens (immunoglobulin [Ig] G, IgM, IgA, C3, and C1q) was used. Positive IF staining was defined as a score of >2 in the sum of linear-, granular-, peripheral-, and mesangial-term in the glomerulus.
B7-1 immunohistochemical staining
We performed immunohistochemical staining for B7-1 by using a BenchMark XT automated immunostaining system (Ventana Medical Systems, Inc., Tucson, AZ, USA). Briefly, the paraffin blocks were cut into 4-μm-thick sections, deparaffinised in xylene, and then hydrated by using alcohol (3×). After microwave antigen retrieval, the samples were incubated with a monoclonal mouse anti-human B7-1 antibody (diluted 1:20; catalogue No. MAB140; R&D Systems, Minneapolis, MN, USA) and subsequently treated with the UltraView Universal DAB kit (Ventana Medical Systems, Inc.). Harris haematoxylin was used as a counterstain. Two independent specialists blinded to the patients’ clinical data evaluated the stained slides. If the interpretation of the staining results was not consistent, the two specialists discussed conflicting findings. The intensity of the signal was rated 0 (negative), 1 (weak), or 2 (strong). Staining results for the glomerulus and tubular epithelium were recorded separately. Normal tonsillar tissue was used as a positive control for B7-1.
Statistical analysis
Values were expressed as median (interquartile range) for continuous variables and % (n/total) for categorical variables. The difference was analysed by using the Kruskal-Wallis test or Mann-Whitney U test for continuous variables, and the Chi-square or Fisher exact test for categorical variables. A P value of <0.05 was considered statistically significant. All the analyses were performed by using SPSS Statistics version 22 (IBM Corp, Armonk, NY, USA).
Additional Information
How to cite this article: Lee, S. W. et al. Tubular B7-1 expression parallels proteinuria levels, but not clinical outcomes in adult minimal change disease patients. Sci. Rep. 7, 41859; doi: 10.1038/srep41859 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Lee, H. et al. Mortality and renal outcome of primary glomerulonephritis in Korea: observation in 1,943 biopsied cases. Am J Nephrol 37, 74–83, doi: 10.1159/000345960 (2013).
Sharpstone, P., Ogg, C. S. & Cameron, J. S. Nephrotic syndrome due to primary renal disease in adults: I. Survey of incidence in South-east England. Br Med J 2, 533–535 (1969).
Malafronte, P. et al. Paulista Registry of glomerulonephritis: 5-year data report. Nephrol Dial Transplant 21, 3098–3105, doi: 10.1093/ndt/gfl237 (2006).
Lee, S. W. et al. Glomerular immune deposits are predictive of poor long-term outcome in patients with adult biopsy-proven minimal change disease: a cohort study in Korea. PloS One 11, e0147387, doi: 10.1371/journal.pone.0147387 (2016).
Lee, S. W. et al. Glomerular hypertrophy is a risk factor for relapse in minimal change disease patients. Nephron 132, 43–50, doi: 10.1159/000442680 (2016).
Kyrieleis, H. A. et al. Long-term outcome of biopsy-proven, frequently relapsing minimal-change nephrotic syndrome in children. Clin J Am Soc Nephrol 4, 1593–1600, doi: 10.2215/CJN.05691108 (2009).
Mathieson, P. W. Immune dysregulation in minimal change nephropathy. Nephrol Dial Transplant 18, Suppl 6, vi26–29 (2003).
Shalhoub, R. J. Pathogenesis of lipoid nephrosis: a disorder of T-cell function. Lancet 2, 556–560 (1974).
Shimada, M. et al. Minimal change disease: a ‘two-hit’ podocyte immune disorder? Pediatr Nephrol 26, 645–649, doi: 10.1007/s00467-010-1676-x (2011).
Sharpe, A. H. & Freeman, G. J. The B7-CD28 superfamily. Nat Rev Immunol 2, 116–126, doi: 10.1038/nri727 (2002).
Wang, S. & Chen, L. Co-signaling molecules of the B7-CD28 family in positive and negative regulation of T lymphocyte responses. Microbes Infect 6, 759–766, doi: 10.1016/j.micinf.2004.03.007 (2004).
Reiser, J. et al. Induction of B7-1 in podocytes is associated with nephrotic syndrome. J Clin Invest 113, 1390–1397, doi: 10.1172/JCI20402 (2004).
Garin, E. H. et al. Urinary CD80 excretion increases in idiopathic minimal-change disease. J Am Soc Nephrol 20, 260–266, doi: 10.1681/ASN.2007080836 (2009).
Garin, E. H. et al. Urinary CD80 is elevated in minimal change disease but not in focal segmental glomerulosclerosis. Kidney Int 78, 296–302, doi: 10.1038/ki.2010.143 (2010).
Navarro-Munoz, M. et al. Messenger RNA expression of B7-1 and NPHS1 in urinary sediment could be useful to differentiate between minimal-change disease and focal segmental glomerulosclerosis in adult patients. Nephrol Dial Transplant 26, 3914–3923, doi: 10.1093/ndt/gfr128 (2011).
Shimada, M. et al. Toll-like receptor 3 ligands induce CD80 expression in human podocytes via an NF-kappaB-dependent pathway. Nephrol Dial Transplant 27, 81–89, doi: 10.1093/ndt/gfr271 (2012).
Ishimoto, T. et al. Toll-like receptor 3 ligand, polyIC, induces proteinuria and glomerular CD80, and increases urinary CD80 in mice. Nephrol Dial Transplant 28, 1439–1446, doi: 10.1093/ndt/gfs543 (2013).
Yu, C. C. et al. Abatacept in B7-1-positive proteinuric kidney disease. N Engl J Med 369, 2416–2423, doi: 10.1056/NEJMoa1304572 (2013).
Fiorina, P. et al. Role of podocyte B7-1 in diabetic nephropathy. J Am Soc Nephrol 25, 1415–1429, doi: 10.1681/ASN.2013050518 (2014).
Larsen, C. P., Messias, N. C. & Walker, P. D. B7-1 immunostaining in proteinuric kidney disease. Am J Kidney Dis 64, 1001–1003, doi: 10.1053/j.ajkd.2014.07.023 (2014).
Benigni, A., Gagliardini, E. & Remuzzi, G. Abatacept in B7-1-positive proteinuric kidney disease. N Engl J Med 370, 1261–1263, doi: 10.1056/NEJMc1400502#SA1 (2014).
Gagliardini, E. et al. B7-1 is not induced in podocytes of human and experimental diabetic nephropathy. J Am Soc Nephrol 27, 999–1005, doi: 10.1681/ASN.2015030266 (2016).
Niemann-Masanek, U., Mueller, A., Yard, B. A., Waldherr, R. & van der Woude, F. J. B7-1 (CD80) and B7-2 (CD 86) expression in human tubular epithelial cells in vivo and in vitro . Nephron 92, 542–556, doi: 64084 (2002).
Wu, Q., Jinde, K., Endoh, M. & Sakai, H. Costimulatory molecules CD80 and CD86 in human crescentic glomerulonephritis. Am J Kidney Dis 41, 950–961 (2003).
Cara-Fuentes, G., Wasserfall, C. H., Wang, H., Johnson, R. J. & Garin, E. H. Minimal change disease: a dysregulation of the podocyte CD80-CTLA-4 axis? Pediatr Nephrol 29, 2333–2340, doi: 10.1007/s00467-014-2874-8 (2014).
Cara-Fuentes, G. et al. CD80 and suPAR in patients with minimal change disease and focal segmental glomerulosclerosis: diagnostic and pathogenic significance. Pediatr Nephrol 29, 1363–1371, doi: 10.1007/s00467-013-2679-1 (2014).
Novelli, R., Gagliardini, E., Ruggiero, B., Benigni, A. & Remuzzi, G. Any value of podocyte B7-1 as a biomarker in human MCD and FSGS? Am J Physiol Renal Physiol 310, F335–341, doi: 10.1152/ajprenal.00510.2015 (2016).
Mundel, P. & Greka, A. Developing therapeutic ‘arrows’ with the precision of William Tell: the time has come for targeted therapies in kidney disease. Curr Opin Nephrol Hypertens 24, 388–392, doi: 10.1097/MNH.0000000000000137 (2015).
Banu, N. & Meyers, C. M. IFN-gamma and LPS differentially modulate class II MHC and B7-1 expression on murine renal tubular epithelial cells. Kidney Int 55, 2250–2263, doi: 10.1046/j.1523-1755.1999.00495.x (1999).
Zoja, C. et al. Protein overload stimulates RANTES production by proximal tubular cells depending on NF-kappa B activation. Kidney Int 53, 1608–1615, doi: 10.1046/j.1523-1755.1998.00905.x (1998).
Ling, C. et al. Urinary CD80 levels as a diagnostic biomarker of minimal change disease. Pediatric Nephrol 30, 309–316, doi: 10.1007/s00467-014-2915-3 (2015).
Kidney Disease: Improving Global Outcomes (KDIGO) Acute Kidney Injury Work Group. KDIGO clinical practice guideline for acute kidney injury. Kidney Int Suppl 2, 1–138, doi: 10.1038/kisup.2011.32 (2012).
Zhou, X. J. et al. The aging kidney. Kidney Int 74, 710–720, doi: 10.1038/ki.2008.319 (2008).
Oh, S. W. et al. Clinical implications of pathologic diagnosis and classification for diabetic nephropathy. Diabetes Res Clin Pract. 97, 418–424, doi: 10.1016/j.diabres.2012.03.016 (2012).
Author information
Authors and Affiliations
Contributions
S.W.L. and H.J.C. conceptualised and designed the study. S.W.L. and J.H.P. performed the pathologic evaluation. S.H.B., S.K., K.Y.N., and D.-W.C. analysed the data. S.W.L. and H.J.C. wrote the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Lee, S., Baek, S., Paik, J. et al. Tubular B7-1 expression parallels proteinuria levels, but not clinical outcomes in adult minimal change disease patients. Sci Rep 7, 41859 (2017). https://doi.org/10.1038/srep41859
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep41859
This article is cited by
-
B7-1 mediates podocyte injury and glomerulosclerosis through communication with Hsp90ab1-LRP5-β-catenin pathway
Cell Death & Differentiation (2022)
-
Biomarkers in pediatric glomerulonephritis and nephrotic syndrome
Pediatric Nephrology (2021)
-
Glomerular endothelial cells and podocytes can express CD80 in patients with minimal change disease during relapse
Pediatric Nephrology (2020)
-
The role of B7-1 in proteinuria of glomerular origin
Nature Reviews Nephrology (2018)
-
Urinary CD80: a biomarker for a favorable response to corticosteroids in minimal change disease
Pediatric Nephrology (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.