Abstract
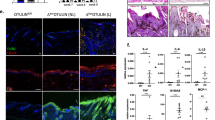
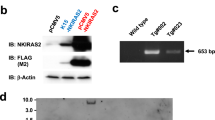
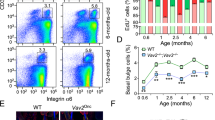
Keratinocyte-derived TNF-α acts as an endogenous tumour promoter and can also regulate AP-1 activity in mouse epidermis. To gain further insight into TNF-α signalling during skin tumour formation, mice deficient in TNFR1 (TNFR1−/− mice) or TNFR2 (TNFR2−/− mice) were subjected to chemical carcinogenesis. Tumour multiplicity was significantly reduced in TNFR1−/− and TNFR2−/− mice compared to wild-type (wt) mice, suggesting that both receptors have protumour activity. However, TNFR1−/− mice were markedly more resistant to tumour development than TNFR2−/− mice indicating that TNFR1 is the major mediator of TNF-α-induced tumour formation. TNFR1 and TNFR2 were both expressed in wt epidermis during tumour promotion and by primary keratinocytes in vitro. TPA-induced c-Jun expression was transient in TNFR1−/− and TNFR2−/− compared to wt epidermis and this was reflected by reduced induction of the AP-1-responsive genes granulocyte/macrophage-colony stimulating factor, matrix metalloproteinase-9 and matrix metalloproteinase-3. These genes were differentially regulated in TNFR1−/− compared to TNFR2−/− epidermis, suggesting that the TNF-α receptors act independently via different AP-1 complexes to transduce TNF-α signals during tumour promotion. In addition, TNFR2 cooperated with TNFR1 to optimise TNFR1-mediated TNF-α bioactivity on keratinocytes in vitro. Our data provide further insight into TNF-α signalling in malignancy and provide some rationale for the use of TNF-α antagonists in the treatment of cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Andrews NC and Faller DV . (1991). Nucleic Acids Res., 19, 2499.
Arnott CH, Scott KA, Moore RJ, Hewer A, Phillips DH, Parker P, Balkwill FR and Owens DM . (2002). Oncogene, 21, 4728–4738.
Behrens A, Jochum W, Sibilia M and Wagner EF . (2000). Oncogene, 19, 2657–2663.
Bradford MM . (1976). Anal. Biochem., 72, 248–254.
Chan FK and Lenardo MJ . (2000). Eur. J. Immunol., 30, 652–660.
Chomczynski P and Sacchi N . (1987). Anal. Biochem., 162, 156–159.
Coussens LM, Tinkle CL, Hanahan D and Werb Z . (2000). Cell, 103, 481–490.
DiGiovanni J . (1992). Pharmacol. Ther., 54, 63–128.
Frame S, Crombie R, Liddell J, Stuart D, Linardopoulos S, Nagase H, Portella G, Brown K, Street A, Akhurst R and Balmain A . (1998). Philos. Trans. R. Soc. Lond. B. Biol. Sci., 353, 839–845.
Grell M, Douni E, Wajant H, Lohden M, Clauss M, Maxeiner B, Georgopoulos S, Lesslauer W, Kollias G, Pfizenmaier K and Scheurich P . (1995). Cell, 83, 793–802.
Higuchi M and Aggarwal BB . (1992). J. Biol. Chem., 267, 20892–20899.
Hulboy DL, Matrisian LM and Crawford HC . (2001). Mol. Cell. Biol., 21, 5478–5487.
Jochum W, Passegue E and Wagner EF . (2001). Oncogene, 20, 2401–2412.
Kitakata H, Nemoto SY, Takahashi Y, Kondo T, Mai M and Mukaida N . (2002). Cancer Res., 62, 6682–6687.
Knight B, Yeoh GC, Husk KL, Ly T, Abraham LJ, Yu C, Rhim JA and Fausto N . (2000). J. Exp. Med., 192, 1809–1818.
Kohno T, Brewer MT, Baker SL, Schwartz PE, King MW, Hale KK, Squires CH, Thompson RC and Vannice JL . (1990). Proc. Natl. Acad. Sci. USA, 87, 8331–8335.
Leber TM and Balkwill FR . (1997). Anal. Biochem., 249, 24–28.
Littlejohn AF, Tucker SJ, Mohamed AA, McKay S, Helms MJ, Vandenabeele P and MacEwan DJ . (2003). Biochem. Pharmacol., 65, 91–99.
Locksley RM, Killeen N and Lenardo MJ . (2001). Cell, 104, 487–501.
MacEwan DJ . (2002). Cell Signal, 14, 477–492.
Mann A, Breuhahn K, Schirmacher P, Wilhelmi A, Beyer C, Rosenau A, Ozbek S, Rose-John S and Blessing M . (2001). Cancer Res., 61, 2311–2319.
Medvedev AE, Espevik T, Ranges G and Sundan A . (1996). J. Biol. Chem., 271, 9778–9784.
Moore RJ, Owens DM, Stamp G, Arnott C, Burke F, East N, Holdsworth H, Turner L, Rollins B, Pasparakis M, Kollias G and Balkwill F . (1999). Nat. Med., 5, 828–831.
Mori R, Kondo T, Ohshima T, Ishida Y and Mukaida N . (2002). FASEB J., 16, 963–974.
Morris RJ . (2000). J. Clin. Invest., 106, 3–8.
Mukhopadhyay A, Suttles J, Stout RD and Aggarwal BB . (2001). J. Biol. Chem., 276, 31906–31912.
Papathoma AS, Zoumpourlis V, Balmain A and Pintzas A . (2001). Mol. Carcinogen., 31, 74–82.
Peschon JJ, Torrance DS, Stocking KL, Glaccum MB, Otten C, Willis CR, Charrier K, Morrissey PJ, Ware CB and Mohler KM . (1998). J. Immunol., 160, 943–952.
Pinckard JK, Sheehan KC and Schreiber RD . (1997). J. Biol. Chem., 272, 10784–10789.
Roper E, Weinberg W, Watt FM and Land H . (2001). EMBO Rep., 2, 145–150.
Rutberg SE, Saez E, Glick A, Dlugosz AA, Spiegelman BM and Yuspa SH . (1996). Oncogene, 13, 167–176.
Scott KA, Moore RJ, Arnott CH, East N, Thompson RG, Scallon BJ, Shealy DJ and Balkwill FR . (2003). Mol. Cancer Ther., 2, 445–451.
Sternlicht MD, Lochter A, Sympson CJ, Huey B, Rougler JP, Gray JW, Pinkel D, Bissell MJ and Werb Z . (1999). Cell, 98, 137–146.
Suganuma M, Okabe S, Marino MW, Sakai A, Sueoka E and Fujiki H . (1999). Cancer Res., 59, 4516–4518.
Szabowski A, Maas-Szabowski N, Andrecht S, Kolbus A, Schorpp-Kistner M, Fusenig NE and Angel P . (2000). Cell, 103, 745–755.
Tartaglia LA, Pennica D and Goeddel DV . (1993). J. Biol. Chem., 268, 18542–18548.
Vandenabeele P, Declercq W, Beyaert R and Fiers W . (1995). Trends Cell Biol., 5, 392–399.
Vogt PK . (2001). Oncogene, 20, 2365–2377.
Yamada Y and Fausto N . (1998). Am. J. Pathol., 152, 1577–1589.
Yamada Y, Kirillova I, Peschon JJ and Fausto N . (1997). Proc. Natl. Acad. Sci. USA, 94, 1441–1446.
Young MR, Li JJ, Rincon M, Flavell RA, Sathyanarayana BK, Hunziker R and Colburn N . (1999). Proc. Natl. Acad. Sci. USA, 96, 9827–9832.
Zhong S, Quealy JA, Bode AM, Nomura M, Kaji A, Ma WY and Dong Z . (2001). Cancer Res., 61, 4084–4091.
Zhuang L, Wang B, Shinder GA, Shivji GM, Mak TW and Sauder DN . (1999). J. Immunol., 162, 1440–1447.
Acknowledgements
The authors thank Nick East for technical assistance; Marian Nakada, Fiona Watt, Frances Burke, Caroline Barton and David Owens for helpful comments on the manuscript; Centocor and Cancer Research UK for funding this research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Arnott, C., Scott, K., Moore, R. et al. Expression of both TNF-α receptor subtypes is essential for optimal skin tumour development. Oncogene 23, 1902–1910 (2004). https://doi.org/10.1038/sj.onc.1207317
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1207317
Keywords
This article is cited by
-
A TNFR1–UBCH10 axis drives lung squamous cell carcinoma dedifferentiation and metastasis through a cell-autonomous signaling loop
Cell Death & Disease (2022)
-
MultiVERSE: a multiplex and multiplex-heterogeneous network embedding approach
Scientific Reports (2021)
-
Anti-inflammation activity of brazilin in TNF-α induced human psoriasis dermatitis skin model
Applied Biological Chemistry (2019)
-
Pre-clinical study of a TNFR1-targeted 18F probe for PET imaging of breast cancer
Amino Acids (2018)
-
Innate sensing of microbial products promotes wound-induced skin cancer
Nature Communications (2015)