Abstract
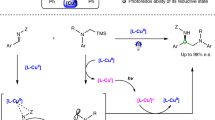
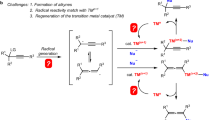
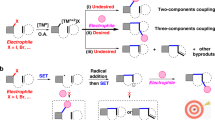
The 3d metals have been identified as economic and sustainable alternatives to palladium, the frequently used metal in transition-metal-catalysed cross-couplings. However, cobalt has long stood behind its neighbouring elements, nickel and copper, in asymmetric radical couplings owing to its high catalytic activity in the absence of ligands resulting in unfavourable un-asymmetric background reactions. Here we disclose an asymmetric metallaphotoredox catalysis (AMPC) strategy for the dynamic kinetic asymmetric transformation of racemic heterobiaryls, which represents a visible-light-induced, asymmetric radical coupling for the construction of axial chirality. This success can also be extended to the reductive cross-coupling variant featuring the use of more easily available organic halide feedstocks. The keys to these achievements are the rational design of a sustainable AMPC system that merges asymmetric cobalt catalysis with organic photoredox catalysis in combination with the identification of an efficient chiral polydentate ligand.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
Crystallographic data for the structures reported in this article have been deposited at the Cambridge Crystallographic Data Centre, under deposition numbers CCDC 2124717 (3g) and 2124714 (Me-3k). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/. Data related to materials and methods, optimization of conditions, experimental procedures, mechanistic experiments, DFT calculations, HPLC spectra and spectra are provided in the Supplementary information. All data are available from the corresponding authors upon reasonable request.
References
Schultz, D. M. & Yoon, T. P. Solar synthesis: prospects in visible light photocatalysis. Science 343, 1239176 (2014).
Marzo, L., Pagire, S. K., Reiser, O. & König, B. Visible-light photocatalysis: does it make a difference in organic synthesis? Angew. Chem. Int. Ed. 57, 10034–10072 (2018).
Chan, A. Y. et al. Metallaphotoredox: the merger of photoredox and transition-metal catalysis. Chem. Rev. 122, 1485–1542 (2022).
Brimioulle, R., Lenhart, D., Maturi, M. M. & Bach, T. Enantioselective catalysis of photochemical reactions. Angew. Chem. Int. Ed. 54, 3872–3890 (2015).
Silvil, M. & Melchiorre, P. Enhancing the potential of enantioselective organocatalysis with light. Nature 554, 41–49 (2018).
Lipp, A., Badir, S. O. & Molander, G. A. Stereoinduction in metallaphotoredox catalysis. Angew. Chem. Int. Ed. 60, 1714–1726 (2021).
Schmermund, L. et al. Photo-biocatalysis: biotransformations in the presence of light. ACS Catal. 9, 4115–4144 (2019).
Bauer, A., Westkämper, F., Grimme, S. & Bach, T. Catalytic enantioselective reactions driven by photoinduced electron transfer. Nature 436, 1139–1140 (2005).
Nicewicz, D. A. & MacMillan, D. W. C. Merging photoredox catalysis with organocatalysis: the direct asymmetric alkylation of aldehydes. Science 322, 77–80 (2008).
Arceo, E., Jurberg, I. D., Álvarez-Fernaández, A. & Melchiorre, P. Photochemical activity of a key donor–acceptor complex can drive stereoselective catalytic α-alkylation of aldehydes. Nat. Chem. 5, 750–756 (2013).
Tellis, J. C., Primer, D. N. & Molander, G. A. Single-electron transmetalation in organoboron cross-coupling by photoredox/nickel dual catalysis. Science 345, 433–436 (2014).
Du, J., Skubi, K. L., Schultz, D. M. & Yoon, T. P. A dual-catalysis approach to enantioselective [2+2] photocycloadditions using visible light. Science 344, 392–396 (2014).
Huo, H. et al. Asymmetric photoredox transition-metal catalysis activated by visible light. Nature 515, 100–103 (2014).
Kainz, Q. M. et al. Asymmetric copper-catalyzed C–N cross-couplings induced by visible light. Science 351, 681–684 (2016).
Emmanuel, M. A., Greenberg, N. R., Oblinsky, D. G. & Hyster, T. K. Accessing non-natural reactivity by irradiating nicotinamide-dependent enzymes with light. Nature 540, 414–417 (2016).
Litman, Z. C., Wang, Y.-J., Zhao, H.-M. & Hartwig, J. F. Cooperative asymmetric reactions combining photocatalysis and enzymatic catalysis. Nature 560, 355–359 (2018).
Shin, N. Y., Ryss, J. M., Zhang, X., Miller, S. J. & Knowles, R. R. Light-driven deracemization enabled by excited-state electron transfer. Science 366, 364–369 (2019).
Cheng, J. K., Xiang, S.-H., Li, S., Ye, L. & Tan, B. Recent advances in catalytic asymmetric construction of atropisomers. Chem. Rev. 121, 4805–4902 (2021).
Hölzl-Hobmeier, A. et al. Catalytic deracemization of chiral allenes by sensitized excitation with visible light. Nature 564, 240–243 (2018).
Plaza, M., Großkopf, J., Breitenlechner, S., Bannwarth, C. & Bach, T. Photochemical deracemization of primary allene amides by triplet energy transfer: a combined synthetic and theoretical study. J. Am. Chem. Soc. 143, 11209–11217 (2021).
Hapke, M. & Hilt, G. (eds) Cobalt Catalysis in Organic Synthesis (Wiley-VCH, 2020).
Mao, J. et al. Cobalt−bisoxazoline-catalyzed asymmetric Kumada cross-coupling of racemic α-bromo esters with aryl Grignard reagents. J. Am. Chem. Soc. 136, 17662–17668 (2014).
Li, Z. et al. A cobalt-catalyzed enantioconvergent radical Negishi C(sp3)-C(sp2) cross-coupling with chiral multidentate N,N,P ligand. Organometallics 40, 2215–2219 (2021).
Li, Y. et al. Cobalt-catalysed enantioselective C(sp3)-C(sp3) coupling. Nat. Catal. 4, 901–911 (2021).
Fu, G. C. Transition-metal catalysis of nucleophilic substitution reactions: a radical alternative to SN1 and SN2 processes. ACS Cent. Sci. 3, 692–700 (2017).
Wang, F., Chen, P. & Liu, G. Copper-catalyzed radical relay for asymmetric radical transformations. Acc. Chem. Res. 51, 2036–2046 (2018).
Gu, Q.-S., Li, Z.-L. & Liu, X.-Y. Copper(I)-catalyzed asymmetric reactions involving radicals. Acc. Chem. Res. 53, 170–181 (2020).
Twilton, J. et al. The merger of transition metal and photocatalysis. Nat. Rev. Chem. 1, 0052 (2017).
Kojima, M. & Matsunaga, S. The merger of photoredox and cobalt catalysis. Trends Chem. 2, 410–426 (2020).
Ding, W. et al. Bifunctional photocatalysts for enantioselective aerobic oxidation of β-ketoesters. J. Am. Chem. Soc. 139, 63–66 (2017).
Lu, F.-D. et al. Asymmetric propargylic radical cyanation enabled by dual organophotoredox and copper catalysis. J. Am. Chem. Soc. 141, 6167–6172 (2019).
Chen, J. et al. Photoinduced copper-catalyzed asymmetric C−O cross-coupling. J. Am. Chem. Soc. 143, 13382–13392 (2021).
Kakiuchi, F., Gendre, P. L., Yamada, A., Ohtaki, H. & Murai, S. Atropselective alkylation of biaryl compounds by means of transition-metal-catalyzed C–H/olefin coupling. Tetrahedron Asymmetry 11, 2647–265 (2000).
Ros, A. et al. Dynamic kinetic cross-coupling strategy for the asymmetric synthesis of axially chiral heterobiaryls. J. Am. Chem. Soc. 135, 15730–15733 (2013).
Bhat, V., Wang, S., Stoltz, B. M. & Virgil, S. C. Asymmetric synthesis of QUINAP via dynamic kinetic resolution. J. Am. Chem. Soc. 135, 16829–16832 (2013).
Ramírez-López, P. et al. A dynamic kinetic C–P cross–coupling for the asymmetric synthesis of axially chiral P,N ligands. ACS Catal. 6, 3955–3964 (2016).
Ramírez-López, P. et al. Synthesis of IAN-type N,N-ligands via dynamic kinetic asymmetric Buchwald–Hartwig amination. J. Am. Chem. Soc. 138, 12053–12056 (2016).
Hornillos, V. et al. Synthesis of axially chiral heterobiaryl alkynes via dynamic kinetic asymmetric alkynylation. Chem. Commun. 52, 14121–14124 (2016).
Carmona, J. A. et al. Dynamic kinetic asymmetric Heck reaction for the simultaneous generation of central and axial chirality. J. Am. Chem. Soc. 140, 11067–11075 (2018).
Han, S.-J., Bhat, V., Stoltz, B. M. & Virgil, S. C. Atroposelective synthesis of PINAP via dynamic kinetic asymmetric transformation. Adv. Synth. Catal. 361, 441–444 (2019).
Wang, Q., Cai, Z.-J., Liu, C.-X., Gu, Q. & You, S.-L. Rhodium-catalyzed atroposelective C−H arylation: efficient synthesis of axially chiral heterobiaryls. J. Am. Chem. Soc. 141, 9504–9510 (2019).
Romero-Arenas, A. et al. Ir-catalyzed atroposelective desymmetrization of heterobiaryls: hydroarylation of vinyl ethers and bicycloalkenes. J. Am. Chem. Soc. 142, 2628–2639 (2020).
Carmona, J. A., Rodríguez-Franco, C., Fernández, R., Hornillos, V. & Lassaletta, J. M. Atroposelective transformation of axially chiral (hetero)biaryls. From desymmetrization to modern resolution strategies. Chem. Soc. Rev. 50, 2968–2983 (2021).
Jin, M.-Y. & Yoshikai, N. Cobalt-xantphos-catalyzed, LiCl-mediated preparation of arylzinc reagents from aryl iodides, bromides, and chlorides. J. Org. Chem. 76, 1972–1978 (2011).
Zhou, W. et al. Chiral sulfinamide bisphosphine catalysts: design, synthesis, and application in highly enantioselective intermolecular cross-Rauhut–Currier reactions. Angew. Chem. Int. Ed. 54, 14853–14857 (2015).
Hu, M., Feng, H.-T., Yuan, Y.-X., Zheng, Y.-S. & Tang, B. Z. Chiral AIEgens–chiral recognition, CPL materials and other chiral applications. Coord. Chem. Rev. 416, 213329 (2020).
Han, J. et al. Enhanced circularly polarized luminescence in emissive charge-transfer complexes. Angew. Chem. Int. Ed. 58, 7013–7019 (2019).
Speckmeier, E., Fischer, T. G. & Zeitler, K. A toolbox approach to construct broadly applicable metal-free catalysts for photoredox chemistry: deliberate tuning of redox potentials and importance of halogens in donor−acceptor cyanoarenes. J. Am. Chem. Soc. 140, 15353–15365 (2018).
Gandolfo, E., Tang, X., Roy, S. R. & Melchiorre, P. Photochemical asymmetric nickel-catalyzed acyl cross-coupling. Angew. Chem. Int. Ed. 58, 16854–168587 (2019).
Acknowledgements
We thank J. Zhang and Soochow Chirally-Chem Co., Ltd for providing ligands, M.-Y. Liu for determination of circularly polarized luminescence signal (|glum|) and F.-F. Pan for the X-ray single-crystal diffraction analysis, and J.-R. Chen and Y. Cheng for helpful discussions. X.Q. acknowledges the supercomputing system in the Supercomputing Center of Wuhan University. This work was supported by the National Natural Science Foundation of China (grant nos. 91956201, 21820102003 and 21772053 to W.-J.X. and 21822103 and 21772052 to L.-Q.L.), the Natural Science Foundation of Hubei Province (grant No. 2017AHB047 to W.-J.X.) and the International Joint Research Center for Intelligent Biosensing Technology and Health.
Author information
Authors and Affiliations
Contributions
L.-Q.L. and W.-J.X. directed the project and wrote the manuscript with input from all other authors. Under the guidance of L.-Q.L. and W.-J.X., X.J. and W.X. developed the methods and designed and performed the synthetic and mechanistic experiments with the help of F.-D.L, Y.J., Q.Y. and L.-Y.X. In addition, S.D. performed the DFT calculations under the guidance of X.Q. J.A.T. gave many helpful suggestions and helped to revise this manuscript. All the authors participated in the discussion and preparation of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Catalysis thanks Dong Wang, Pedro Merino and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Methods, Discussion, references, Tables 1–15 and Figs. 1–24.
Supplementary Data 1
Computational data for Cartesian coordinates of optimized structures.
Supplementary Data 2
Crystallographic data for compound 3g.
Supplementary Data 3
Crystallographic data for compound Me-3k.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jiang, X., Xiong, W., Deng, S. et al. Construction of axial chirality via asymmetric radical trapping by cobalt under visible light. Nat Catal 5, 788–797 (2022). https://doi.org/10.1038/s41929-022-00831-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-022-00831-1
This article is cited by
-
Catalytic atroposelective synthesis
Nature Catalysis (2024)
-
Reduction of unactivated alkyl chlorides enabled by light-induced single electron transfer
Science China Chemistry (2024)
-
Photoredox cobalt-catalyzed regio-, diastereo- and enantioselective propargylation of aldehydes via propargyl radicals
Nature Communications (2023)
-
Construction of C-B axial chirality via dynamic kinetic asymmetric cross-coupling mediated by tetracoordinate boron
Nature Communications (2023)