Abstract
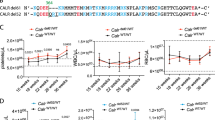
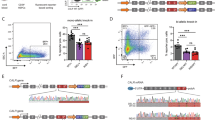
Frameshift mutations in the calreticulin (CALR) gene are present in 30% of essential thrombocythemia and myelofibrosis patients. The two most frequent mutations are CALR del52 (type 1, approximately 60%) and CALR ins5 (type 2, around 30%), but many other rarer mutations exist accounting each for less than 2% of all CALR mutations. Most of them are structurally classified as type 1-like and type 2-like CALR mutations according to the absence or presence of a residual wild-type calcium-binding motif and the modification of the alpha-helix structure. Yet, several key questions remain unanswered, especially the reason of such low frequencies of these other mutations. In an attempt to investigate specific pathogenic differences between type 1-like and type 2-like CALR mutations and del52 and ins5, we modeled two type 1-like (del34 and del46) and one type 2-like (del19) mutations in cell lines and in mice. All CALR mutants constitutively activate JAK2 and STAT5/3/1 in a similar way in the presence of the thrombopoietin receptor (MPL) and induced cytokine-independent cell growth but to a lesser extent with rare mutants over time. This correlates with reduced expression levels of rare CALR mutants compared to del52 and ins5. Lethally irradiated mice that were engrafted with bone marrow transduced with the different CALR mutations developed thrombocytosis, but to a much lesser extent with ins5 and the type 2-like CALR mutation. In contrast to type 2-like mice, type 1-like mice developed marked myelofibrosis and splenomegaly 10 months after engraftment. Similar to del52, type 1-like CALR mutations induced an expansion at an early stage of hematopoiesis compared to ins5 and type 2-like mutation. Thus, type 1-like and type 2-like CALR mutants structurally and functionally resemble del52 and ins5 mutants, respectively.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Barbui T, Thiele J, Gisslinger H, Finazzi G, Vannucchi AM, Tefferi A. The 2016 revision of WHO classification of myeloproliferative neoplasms: clinical and molecular advances. Blood Rev. 2016;30:453–9.
Vainchenker W, Kralovics R. Genetic basis and molecular pathophysiology of classical myeloproliferative neoplasms. Blood. 2017;129:667–79.
Nangalia J, Massie CE, Baxter EJ, Nice FL, Gundem G, Wedge DC, et al. Somatic CALR mutations in myeloproliferative neoplasms with nonmutated JAK2. N Engl J Med. 2013;369:2391–405.
Klampfl T, Gisslinger H, Harutyunyan AS, Nivarthi H, Rumi E, Milosevic JD, et al. Somatic mutations of calreticulin in myeloproliferative neoplasms. N Engl J Med. 2013;369:2379–90.
Chachoua I, Pecquet C, El-Khoury M, Nivarthi H, Albu RI, Marty C, et al. Thrombopoietin receptor activation by myeloproliferative neoplasm associated calreticulin mutants. Blood. 2016;127:1325–35.
Elf S, Abdelfattah NS, Chen E, Perales-Paton J, Rosen EA, Ko A, et al. Mutant calreticulin requires both its mutant C-terminus and the thrombopoietin receptor for oncogenic transformation. Cancer Discov. 2016;6:368–81.
Araki M, Yang Y, Masubuchi N, Hironaka Y, Takei H, Morishita S, et al. Activation of the thrombopoietin receptor by mutant calreticulin in CALR-mutant myeloproliferative neoplasms. Blood. 2016;127:1307–16.
Marty C, Pecquet C, Nivarthi H, El-Khoury M, Chachoua I, Tulliez M, et al. Calreticulin mutants in mice induce an MPL-dependent thrombocytosis with frequent progression to myelofibrosis. Blood. 2016;127:1317–24.
Elala YC, Lasho TL, Gangat N, Finke C, Barraco D, Haider M, et al. Calreticulin variant stratified driver mutational status and prognosis in essential thrombocythemia. Am J Hematol. 2016;91:503–6.
Pietra D, Rumi E, Ferretti VV, Di Buduo CA, Milanesi C, Cavalloni C, et al. Differential clinical effects of different mutation subtypes in CALR-mutant myeloproliferative neoplasms. Leukemia. 2016;30:431–8.
Cabagnols X, Defour JP, Ugo V, Ianotto JC, Mossuz P, Mondet J, et al. Differential association of calreticulin type 1 and type 2 mutations with myelofibrosis and essential thrombocytemia: relevance for disease evolution. Leukemia. 2015;29:249–52.
Tefferi A, Wassie EA, Guglielmelli P, Gangat N, Belachew AA, Lasho TL, et al. Type 1 versus Type 2 calreticulin mutations in essential thrombocythemia: a collaborative study of 1027 patients. Am J Hematol. 2014;89:E121–4.
Eder-Azanza L, Navarro D, Aranaz P, Novo FJ, Cross NC, Vizmanos JL. Bioinformatic analyses of CALR mutations in myeloproliferative neoplasms support a role in signaling. Leukemia. 2014;28:2106–9.
Tefferi A, Lasho TL, Tischer A, Wassie EA, Finke CM, Belachew AA, et al. The prognostic advantage of calreticulin mutations in myelofibrosis might be confined to type 1 or type 1-like CALR variants. Blood. 2014;124:2465–6.
Balligand T, Achouri Y, Pecquet C, Chachoua I, Nivarthi H, Marty C, et al. Pathologic activation of thrombopoietin receptor and JAK2-STAT5 pathway by frameshift mutants of mouse calreticulin. Leukemia. 2016;30:1775–8.
Shide K, Kameda T, Yamaji T, Sekine M, Inada N, Kamiunten A, et al. Calreticulin mutant mice develop essential thrombocythemia that is ameliorated by the JAK inhibitor ruxolitinib. Leukemia. 2017;31:1136–44.
Li J, Prins D, Park HJ, Grinfeld J, Gonzalez-Arias C, Loughran S, et al. Mutant calreticulin knockin mice develop thrombocytosis and myelofibrosis without a stem cell self-renewal advantage. Blood. 2018;131:649–61.
Acknowledgements
This work was supported by grants from Ligue Nationale Contre le Cancer (“Equipe labellisée 2016”, H.R.), Institut National du Cancer (PLBIO-2015, I.P.), MPN Research Foundation (I.P.), Agence Nationale de la Recherche (ANR-DFG 2014, JAKPOT, J.L.V.) and Institut National de la Santé et de la Recherche Médicale (Inserm). B.P-S. was funded by Institut de Médecine Moléculaire Personnalisée du Cancer (MMO, ANR). O.B. was supported by Agence Nationale de la Recherche. Support to S.N.C. was from Ludwig Institute for Cancer Research, Salus Sanguinis, Fondation contre le Cancer, Action de Recherche Concertée and Belgian Genetic Medical Initiative Interuniversity Attraction Pole, Belspo. We thank O. Bawa for histology, the staff of the animal facilities of Gustave Roussy directed by P. Gonin, and P. Rameau and Y. Lecluse at the Imaging and Cytometry Platform.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Toppaldoddi, K.R., da Costa Cacemiro, M., Bluteau, O. et al. Rare type 1-like and type 2-like calreticulin mutants induce similar myeloproliferative neoplasms as prevalent type 1 and 2 mutants in mice. Oncogene 38, 1651–1660 (2019). https://doi.org/10.1038/s41388-018-0538-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-018-0538-z
This article is cited by
-
Calreticulin del52 and ins5 knock-in mice recapitulate different myeloproliferative phenotypes observed in patients with MPN
Nature Communications (2020)
-
The role of calreticulin mutations in myeloproliferative neoplasms
International Journal of Hematology (2020)