Abstract
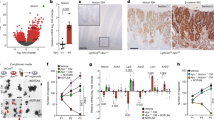
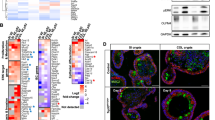
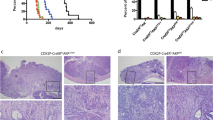
APC is considered a gatekeeper for colorectal cancer (CRC). Cells with heterozygous APC mutations have altered expression profiles suggesting that the first APC hit may help set the stage for subsequent transformation. Therefore, we measured transformation efficiency following what we have designated as ‘simultaneous’ versus ‘stepwise’ Apc loss. We combined a conditional Apc allele (ApcCKO) with a Cre reporter gene and an out-of-frame Cre allele (Pms2cre) that stochastically becomes functional by a frameshift mutation in single cells. Loss of one Apc allele (ApcCKO/+) had little consequence, whereas simultaneous loss of both Apc alleles (ApcCKO/CKO) resulted in increased clonal expansion (crypt fission), consistent with the gatekeeper function of Apc. Interestingly, our analyses showed that most of the Apc-deficient crypts in ApcCKO/CKO mice appeared normal, with morphological transformation, including β-catenin deregulation, occurring in only 17% of such crypts. To determine whether transformation efficiency was different following stepwise Apc loss, we combined ApcCKO with a germline mutant allele, either ApcMin or Apc1638N. Transformation efficiency following stepwise Apc loss (ApcMin/CKO or Apc1638N/CKO) was increased five-fold and essentially all of the Apc-deficient cells were dysplastic. In summary, our data suggest that the gatekeeper function of Apc consists of two roles, clonal expansion and morphological transformation, because simultaneous Apc loss frequently leads to occult clonal expansion without morphological transformation, whereas stepwise Apc loss more often results in visible neoplasia. Finally, that Apc-deficient cells in certain scenarios can retain a normal phenotype is unexpected and may have clinical implications for surveillance strategies to prevent CRC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Akyol A, Hinoi T, Feng Y, Bommer GT, Glaser TM, Fearon ER . (2008). Generating somatic mosaicism with a Cre recombinase-microsatellite sequence transgene. Nat Methods 5: 231–233.
Attolini CS, Cheng YK, Beroukhim R, Getz G, Abdel-Wahab O, Levine RL et al. (2010). A mathematical framework to determine the temporal sequence of somatic genetic events in cancer. Proc Natl Acad Sci USA 107: 17604–17609.
Barker N, Ridgway RA, van Es JH, van de Wetering M, Begthel H, van den Born M et al. (2009). Crypt stem cells as the cells-of-origin of intestinal cancer. Nature 457: 608–611.
Buchert M, Athineos D, Abud HE, Burke ZD, Faux MC, Samuel MS et al. (2010). Genetic dissection of differential signaling threshold requirements for the Wnt/beta-catenin pathway in vivo. PLoS Genet 6: e1000816.
Chan MF, van Amerongen R, Nijjar T, Cuppen E, Jones PA, Laird PW . (2001). Reduced rates of gene loss, gene silencing, and gene mutation in Dnmt1-deficient embryonic stem cells. Mol Cell Biol 21: 7587–7600.
Chen RZ, Pettersson U, Beard C, Jackson-Grusby L, Jaenisch R . (1998). DNA hypomethylation leads to elevated mutation rates. Nature 395: 89–93.
Cruz-Correa M, Hylind LM, Romans KE, Booker SV, Giardiello FM . (2002). Long-term treatment with sulindac in familial adenomatous polyposis: a prospective cohort study. Gastroenterology 122: 641–645.
Eads CA, Nickel AE, Laird PW . (2002). Complete genetic suppression of polyp formation and reduction of CpG-island hypermethylation in Apc(Min/+) Dnmt1-hypomorphic mice. Cancer Res 62: 1296–1299.
Fearon ER, Vogelstein B . (1990). A genetic model for colorectal tumorigenesis. Cell 61: 759–767.
Fodde R, Edelmann W, Yang K, van Leeuwen C, Carlson C, Renault B et al. (1994). A targeted chain-termination mutation in the mouse Apc gene results in multiple intestinal tumors. Proc Natl Acad Sci USA 91: 8969–8973.
Forbes SA, Tang G, Bindal N, Bamford S, Dawson E, Cole C et al. (2010). COSMIC (the Catalogue of Somatic Mutations in Cancer): a resource to investigate acquired mutations in human cancer. Nucleic Acids Res 38: D652–D657.
Hung KE, Maricevich MA, Richard LG, Chen WY, Richardson MP, Kunin A et al. (2010). Development of a mouse model for sporadic and metastatic colon tumors and its use in assessing drug treatment. Proc Natl Acad Sci USA 107: 1565–1570.
Kinzler KW, Vogelstein B . (1996). Lessons from hereditary colorectal cancer. Cell 87: 159–170.
Kuraguchi M, Wang XP, Bronson RT, Rothenberg R, Ohene-Baah NY, Lund JJ et al. (2006). Adenomatous polyposis coli (APC) is required for normal development of skin and thymus. PLoS Genet 2: e146.
Laird PW, Jackson-Grusby L, Fazeli A, Dickinson SL, Jung WE, Li E et al. (1995). Suppression of intestinal neoplasia by DNA hypomethylation. Cell 81: 197–205.
Lopez-Garcia C, Klein AM, Simons BD, Winton DJ . (2010). Intestinal stem cell replacement follows a pattern of neutral drift. Science 330: 822–825.
Lynch PM . (2010). Pharmacotherapy for inherited colorectal cancer. Expert Opin Pharmacother 11: 1101–1108.
Miller AJ, Dudley SD, Tsao JL, Shibata D, Liskay RM . (2008). Tractable Cre–lox system for stochastic alteration of genes in mice. Nat Methods 5: 227–229.
Moser AR, Pitot HC, Dove WF . (1990). A dominant mutation that predisposes to multiple intestinal neoplasia in the mouse. Science 247: 322–324.
Oshima M, Oshima H, Kitagawa K, Kobayashi M, Itakura C, Taketo M . (1995). Loss of Apc heterozygosity and abnormal tissue building in nascent intestinal polyps in mice carrying a truncated Apc gene. Proc Natl Acad Sci USA 92: 4482–4486.
Patel BB, Li XM, Dixon MP, Blagoi EL, Nicolas E, Seeholzer SH et al. (2011). APC+/− alters colonic fibroblast proteome in FAP. Oncotarget 2: 197–208.
Pleasance ED, Cheetham RK, Stephens PJ, McBride DJ, Humphray SJ, Greenman CD et al. (2010). A comprehensive catalogue of somatic mutations from a human cancer genome. Nature 463: 191–196.
Quante M, Tu SP, Tomita H, Gonda T, Wang SS, Takashi S et al. (2011). Bone marrow-derived myofibroblasts contribute to the mesenchymal stem cell niche and promote tumor growth. Cancer Cell 19: 257–272.
Quintana E, Shackleton M, Foster HR, Fullen DR, Sabel MS, Johnson TM et al. (2010). Phenotypic heterogeneity among tumorigenic melanoma cells from patients that is reversible and not hierarchically organized. Cancer Cell 18: 510–523.
Quyn AJ, Appleton PL, Carey FA, Steele RJ, Barker N, Clevers H et al. (2010). Spindle orientation bias in gut epithelial stem cell compartments is lost in precancerous tissue. Cell Stem Cell 6: 175–181.
Sansom OJ, Reed KR, Hayes AJ, Ireland H, Brinkmann H, Newton IP et al. (2004). Loss of Apc in vivo immediately perturbs Wnt signaling, differentiation, and migration. Genes Dev 18: 1385–1390.
Sasai H, Masaki M, Wakitani K . (2000). Suppression of polypogenesis in a new mouse strain with a truncated Apc(Delta474) by a novel COX-2 inhibitor, JTE-522. Carcinogenesis 21: 953–958.
Shibata H, Toyama K, Shioya H, Ito M, Hirota M, Hasegawa S et al. (1997). Rapid colorectal adenoma formation initiated by conditional targeting of the Apc gene. Science 278: 120–123.
Sjoblom T, Jones S, Wood LD, Parsons DW, Lin J, Barber TD et al. (2006). The consensus coding sequences of human breast and colorectal cancers. Science 314: 268–274.
Smits R, van der Houven van Oordt W, Luz A, Zurcher C, Jagmohan-Changur S, Breukel C et al. (1998). Apc1638N: a mouse model for familial adenomatous polyposis-associated desmoid tumors and cutaneous cysts. Gastroenterology 114: 275–283.
Snippert HJ, van der Flier LG, Sato T, van Es JH, van den Born M, Kroon-Veenboer C et al. (2010). Intestinal crypt homeostasis results from neutral competition between symmetrically dividing Lgr5 stem cells. Cell 143: 134–144.
Su LK, Kinzler KW, Vogelstein B, Preisinger AC, Moser AR, Luongo C et al. (1992). Multiple intestinal neoplasia caused by a mutation in the murine homolog of the APC gene. Science 256: 668–670.
Vermeulen L, De Sousa EMF, van der Heijden M, Cameron K, de Jong JH, Borovski T et al. (2010). Wnt activity defines colon cancer stem cells and is regulated by the microenvironment. Nat Cell Biol 12: 468–476.
Wang D, Pezo RC, Corner G, Sison C, Lesser ML, Shenoy SM et al. (2010). Altered dynamics of intestinal cell maturation in Apc1638N/+ mice. Cancer Res 70: 5348–5357.
Yeung AT, Patel BB, Li X-M, Seeholzer SH, Coudry RA, Cooper HS et al. (2008). One-hit effects in cancer: altered proteome of morphologically normal colon crypts in familial adenomatous polyposis. Cancer Res 68: 7579–7586.
Acknowledgements
We thank Drs James Stringer and Melissa Wong for critical reading of the manuscript. We also thank Drs Raju Kucherlapati and Winfried Edelmann for the ApcCKO and Apc1638N mice, respectively; John Swain and Dr Melissa Wong for ApcMin mice and Apc580S intestinal material; and Dan Lioy and Drs Gail Mandel and Paul Brehm for assistance with confocal microscopy. RML and DS were funded by NIH Grant 2R01GM032741-28. JMF was funded by NIH training Grant 5T32HD046420-05 and ACS postdoctoral fellow PF-11-067-01-TBE.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Fischer, J., Miller, A., Shibata, D. et al. Different phenotypic consequences of simultaneous versus stepwise Apc loss. Oncogene 31, 2028–2038 (2012). https://doi.org/10.1038/onc.2011.385
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.385
Keywords
This article is cited by
-
Apc-related models of intestinal neoplasia: a brief review for pathologists
Surgical and Experimental Pathology (2019)